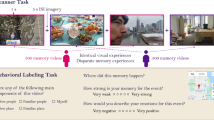
Abstract
It has long been hypothesized that individual recollection fits collective memory. To look for a collective schema, we analysed the content of 30 years of media coverage of World War II on French national television. We recorded human brain activity using functional magnetic resonance imaging as participants recalled World War II displays at the Caen Memorial Museum following an initial tour. We focused on the medial prefrontal cortex, a key region for social cognition and memory schemas. The organization of individual memories captured using the distribution of the functional magnetic resonance imaging signal in the dorsal part of the medial prefrontal cortex was more accurately predicted by the structure of the collective schema than by various control models of contextual or semantic memory. Collective memory, which exists outside and beyond individuals, can also organize individual memories and constitutes a common mental model that connects people’s memories across time and space.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All raw behavioural and imaging data are archived at the GIP Cyceron Centre in Caen. The collective memory corpus is archived at the INAthèque (Bibliothèque Nationale de France) in Paris, which has the legal deposit. This corpus was collected by the MATRICE project (http://www.matricememory.fr/?lang=en), a multidisciplinary and technological consortium of research units, whose aim is to provide tools and technological and theoretical background to understand the relationship between collective and individual memory. Scientists interested in the analysis of this corpus are welcome to submit a project to the MATRICE project and join the consortium. The tagged Wikipedia corpus used for these analyses is available at http://redac.univ-tlse2.fr/corpus/wikipedia.html.
Code availability
The text processing and topic analysis of the collective memory and Wikipedia corpora were conducted using TXM (http://textometrie.ens-lyon.fr/) and MALLET (http://mallet.cs.umass.edu/topics.php) software. The MATLAB implementation based on SPM12 (https://www.fil.ion.ucl.ac.uk/spm/) and the RSA toolbox (https://github.com/rsagroup/rsatoolbox) of the first level and RSAs are available from the corresponding author on request.
References
Halbwachs, M. Les Cadres Sociaux de la Mémoire (Presses Univ. de France, 1925).
Bartlett, F. C. Remembering: A Study in Experimental and Social Psychology (Cambridge Univ. Press, 1932).
Wertsch, J. V. & Roediger, H. L. III Collective memory: conceptual foundations and theoretical approaches. Memory 16, 318–326 (2008).
Hirst, W. & Manier, D. Towards a psychology of collective memory. Memory 16, 183–200 (2008).
García-Gavilanes, R., Mollgaard, A., Tsvetkova, M. & Yasseri, T. The memory remains: understanding collective memory in the digital age. Sci. Adv. 3, e1602368 (2017).
Olick, J. K. Collective memory: the two cultures. Sociol. Theory 7, 333–348 (1999).
Coman, A., Momennejad, I., Drach, R. D. & Geana, A. Mnemonic convergence in social networks: the emergent properties of cognition at a collective level. Proc. Natl Acad. Sci. USA 113, 8171–8176 (2016).
Hirst, W. & Echterhoff, G. Remembering in conversations: the social sharing and reshaping of memories. Annu. Rev. Psychol. 63, 55–79 (2012).
Hirst, W., Yamashiro, J. K. & Coman, A. Collective memory from a psychological perspective. Trends Cogn. Sci. 22, 438–451 (2018).
Gilboa, A. & Malartte, H. Neurobiology of schemas and schema-mediated memory. Trends Cogn. Sci. 21, 618–631 (2019).
Mar, R. M. The neural bases of social cognition and story comprehension. Annu. Rev. Psychol. 62, 103–134 (2011).
Krueger, F., Barbey, A. K. & Grafman, J. The medial prefrontal cortex mediates social event knowledge. Trends Cogn. Sci. 13, 103–109 (2009).
van Kesteren, M. T., Fernández, G., Norris, D. G. & Hermans, E. J. Persistent schema-dependent hippocampal-neocortical connectivity during memory encoding and postencoding rest in humans. Proc. Natl Acad. Sci. USA 107, 7550–7555 (2010).
Benoit, R. G., Szpunar, K. K. & Schacter, D. L. Ventromedial prefrontal cortex supports affective future simulation by integrating distributed knowledge. Proc. Natl Acad. Sci. USA 111, 16550–16555 (2014).
Barron, H. C., Dolan, R. J. & Behrens, T. E. Online evaluation of novel choices by simultaneous representation of multiple memories. Nat. Neurosci. 16, 1492–1498 (2013).
Euston, D. R., Gruber, A. J. & McNaughton, B. L. The role of medial prefrontal cortex in memory and decision making. Neuron 76, 1057–1070 (2012).
Wagner, D., Haxby, J. V. & Heatherton, T. F. The representation of self and person knowledge in the medial prefrontal cortex. WIRES Cogn. Sci. 3, 451–470 (2012).
Wagner, D., Kelley, W. M., Haxby, J. V. & Heatherton, T. F. The dorsal medial prefrontal cortex responds preferentially to social interactions during natural viewing. J. Neurosci. 36, 6917–6925 (2016).
Van Overwalle, F. Social cognition and the brain: a meta-analysis. Hum. Brain Mapp. 30, 829–858 (2009).
Powers, K. E., Chavez, R. S. & Heatherton, T. F. Individual differences in response of dorsomedial prefrontal cortex predict daily social behavior. Soc. Cogn. Affect. Neurosci. 11, 121–126 (2016).
Contreras, J. M., Schirmer, J., Banaji, M. R. & Mitchell, J. P. Common brain regions with distinct patterns of neural responses during mentalizing about groups and individuals. J. Cogn. Neurosci. 25, 1406–1417 (2013).
Chen, J. et al. Shared memories reveal shared structure in neural activity across individuals. Nat. Neurosci. 20, 115–125 (2017).
Foltz, P. W. & Wells, A. D. Automatically deriving readers’ knowledge structures from texts. Behav. Res. Methods Instrum. Comput. 31, 208–214 (1999).
Manning, J. R., Sperling, M. R., Sharan, A., Rosenberg, E. A. & Kahana, M. J. Spontaneously reactivated patterns in frontal and temporal lobe predict semantic clustering during memory search. J. Neurosci. 32, 8871–8878 (2012).
Kenett, Y. N., Levi, E., Anaki, D. & Faust, M. The semantic distance task: quantifying semantic distance with semantic network path length. J. Exp. Psychol. Learn. Mem. Cogn. 43, 1470–1489 (2017).
Griffiths, T. L., Steyvers, M. & Tenenbaum, J. B. Topics in semantic representation. Psychol. Rev. 114, 211–244 (2007).
Pereira, F., Detre, G. & Botvinick, M. Generating text from functional brain images. Front. Hum. Neurosci. 5, 72 (2011).
Huth, A. G., de Heer, W. A., Griffiths, T. L., Theunissen, F. E. & Gallant, J. L. Natural speech reveals the semantic maps that tile human cerebral cortex. Nature 532, 453–458 (2016).
Blei, D. M., Ng, A. Y. & Jordan, M. I. Latent Dirichlet allocation. J. Mach. Learn. Res. 3, 993–1022 (2003).
Steyvers, M. & Griffiths, T. L. in Handbook of Latent Semantic Analysis (eds Landauer, T. et al.) 427–448 (Erlbaum, 2007).
Coman, A., Brown, A. D., Koppel, J. & Hirst, W. Collective memory from a psychological perspective. Int. J. Polit. Cult. Soc. 22, 125–141 (2009).
Kriegeskorte, N. & Mur, M. Inverse MDS: inferring dissimilarity structure from multiple item arrangements. Front. Psychol. 3, 245 (2012).
Abdi, H., Williams, L. J., Valentin, D. & Bennani-Dosse, M. STATIS and DISTATIS: optimum multi-table principal component analysis and three way metric multidimensional scaling. WIRES Comput. Stat. 4, 124–167 (2012).
Nili, H. et al. Toolbox for representational similarity analysis. PLoS Comput. Biol. 10, e1003553 (2014).
Ferron, M. & Massa, P. Beyond the encyclopedia: collective memories in Wikipedia. Mem. Stud. 7, 22–45 (2014).
Pentzold, C. Fixing the floating gap: the online encyclopaedia Wikipedia as a global memory place. Mem. Stud. 2, 255–272 (2009).
Chikazoe, J., Lee, D. H., Kriegeskorte, N. & Anderson, A. K. Population coding of affect across stimuli, modalities and individuals. Nat. Neurosci. 17, 1114–1122 (2014).
Bracci, S., Caramazza, A. & Peelen, M. V. Representational similarity of body parts in human occipitotemporal cortex. J. Neurosci. 35, 12977–12985 (2015).
Brod, G., Lindenberger, U., Werkle-Bergner, M. & Shing, Y. L. Differences in the neural signature of remembering schema-congruent and schema-incongruent events. Neuroimage 117, 358–366 (2015).
Pernet, C. R., Wilcox, R. & Rousselet, G. A. Robust correlation analyses: false positive and power validation using a new open source MATLAB toolbox. Front. Psychol. 3, 606 (2013).
Kumaran, D., Summerfield, J. J., Hassabis, D. & Maguire, E. A. Tracking the emergence of conceptual knowledge during human decision making. Neuron 63, 889–901 (2009).
Edelson, M., Sharot, T., Dolan, R. J. & Dudai, Y. Following the crowd: brain substrates of long-term memory conformity. Science 333, 108–111 (2011).
Echterhoff, G., Higgins, E. T. & Levine, J. M. Shared reality: experiencing commonality with others’ inner states about the world. Psychol. Sci. 4, 496–521 (2009).
Nicolle, A. et al. An agent independent axis for executed and modeled choice in medial prefrontal cortex. Neuron 75, 1114–1121 (2012).
Friston, K. & Frith, C. A duet for one. Conscious. Cogn. 36, 390–405 (2015).
Mahr, J. & Csibra, G. Why do we remember? The communicative function of episodic memory. Behav. Brain Sci. e1, 1–63 (2017).
Constantinescu, A. O., O’Reilly, J. X. & Behrens, T. E. J. Organizing conceptual knowledge in humans with a gridlike code. Science 352, 1464–1468 (2016).
Yun, J., Lee, S. H. & Jeong, H. Early onset of structural inequality in the formation of collaborative knowledge in all Wikimedia projects. Nat. Hum. Behav. 3, 155–163 (2019).
Peschanski, D. in Mémoire et Oubli (ed. Eustache, F.) 71–91 (Le Pommier, 2014).
Turner, B. O., Mumford, J. A., Poldrack, R. A. & Ashby, F. G. Spatiotemporal activity estimation for multivoxel pattern analysis with rapid event-related designs. NeuroImage 62, 1429–1438 (2012).
Mumford, J. A., Turner, B. O., Ashby, F. G. & Poldrack, R. A. Deconvolving BOLD activation in event-related designs for multivoxel pattern classification analyses. NeuroImage 59, 2636–2643 (2012).
Tzourio-Mazoyer, N. et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage 15, 273–289 (2002).
Benjamini, Y. & Yekutieli, D. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 29, 1165–1188 (2001).
Dale, A. M., Fischl, B. & Sereno, M. I. Cortical surface-based analysis. I. Segmentation and surface reconstruction. NeuroImage 9, 179–194 (1999).
Fischl, B., Sereno, M. I. & Dale, A. M. Cortical surface-based analysis. II. Inflation, flattening, and a surface-based coordinate system. NeuroImage 9, 195–207 (1999).
Oosterhof, M. N., Wiesterl, T., Downing, P. E. & Diedrichsen, J. A comparison of volume-based and surface-based multi-voxel pattern analysis. NeuroImage 56, 593–600 (2011).
Winkler, A. M., Ridgway, G. R., Webster, M. A., Smith, S. & Nichols, T. E. Permutation inference for the general linear model. NeuroImage 92, 381–397 (2014).
Smith, S. M. & Nichols, T. E. Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage 44, 83–98 (2009).
Peschanski, D. Les Années Noires 387–402 (Hermann, 2012).
Heiden, S. The TXM platform: building open-source textual analysis software compatible with the TEI encoding scheme. In 24th Pacific Asia Conference on Language, Information and Computation (eds Otoguro, R. et al.) 389–398 (Institute for Digital Enhancement of Cognitive Development, Waseda University, 2010).
Schmid, H. Probabilistic part-of-speech tagging using decision trees. In International Conference on New Methods in Language Processing 44–49 (1994).
Griffiths, T. L. & Steyvers, M. Finding scientific topics. Proc. Natl Acad. Sci. USA 101, 5228–5235 (2004).
Acknowledgements
We thank R. Benoit and M. Wimber for helpful corrections and comments, as well as R. Henson for helpful discussions, on an earlier version of this manuscript. We thank B. Pincemin for her help in the early stages of this project, as well as for her final comments on this manuscript. We thank S. Grimaldi, head of the Caen Memorial, for giving us full access and clearance to proceed with our experiment. We thank “l’observatoire B2V des Mémoires” and M. Morel for help with data collection for the image arrangement task, as well as H. Abdi for guidance on data analysis. We thank D. Maréchal for his help with the selection of television news, as well as K. Dauchot and M. Dufossé for their help during the initial piloting of the experiment. We thank E. Portier and T. Chan for editing the English of the main text. We also thank all participants for volunteering in this study. The study was supported by an EQUIPEX (MATRICE) led by D.P. and funded by the French National Research Agency (grant no. 10-EQPX-21-01), and by a three-year postdoctoral fellowship from the Normandy region to P.G. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
P.G., D.P. and F.E. designed the experiment and wrote the article. P.G. analysed the data. T.V. designed and programmed the image arrangement task, constructed experimental material, processed the corpus and collected the data. S.H. and M.D. programmed the script embedded in TXM to lexically process the corpus. J.-L.G. and A.L. designed and programmed the speech-to-text conversion algorithm to transform the corpus of news into text. C.K.-P. and D.P. supervised the collection of the collective memory corpus and its storing, manual editing and trimming. F.V. supervised the MRI data collection.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Primary Handling Editor(s): Marike Schiffer.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
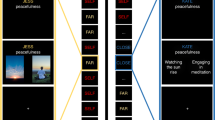
Extended Data Fig. 1 Results of searchlight analysis.
A searchlight analysis following the surface curvature of the brain was ran to test the effects of collective schema, and contextual and semantic memory. At each searchlight, brain RDM was extracted and compared to RDM models using a regression model, thus producing a beta map for each participant (n = 24) and RDM model. To correct for multiple comparisons, the group-level beta map was submitted to maximal permutation testing using threshold-free cluster enhancement (TFCE58). Blue regions indicate clusters surviving TFCE correction across the whole brain (P corrected < .05) for collective schema. There was no statistically significant evidence that brain regions were related to other RDM models using the searchlight approach.
Extended Data Fig. 2 Graph network of topic model.
To illustrate the semantic model and the connections between words derived from our topic models, we computed a Lemmas x Lemmas correlation matrix using the estimated distribution of probabilities over 10 topics. This correlation matrix was then thresholded and transformed into a binary adjacency matrix by keeping the top 10% of the strongest connections between lemmas. The adjacency matrix is visualized here using a force vector algorithm proposed with the Gephi software (https://gephi.org/). Each node represents one of the 6,240 lemmas. The color and the size of the node is determined by its maximal topic assignment and probability, respectively. Amongst the 6,240 nodes, only key words describing Memorial pictures and specifically translated into English, are displayed here for visualization purpose. The size of the label is proportional to its topic probability. The distribution of topic probabilities for each Memorial pictures is directly derived from the topic probabilities associated with these key words.
Supplementary information
Supplementary Information
Supplementary Tables 1–2.
Source data
Source Data Fig. 3
Statistical Source Data
Source Data Fig. 4
Statistical Source Data
Rights and permissions
About this article
Cite this article
Gagnepain, P., Vallée, T., Heiden, S. et al. Collective memory shapes the organization of individual memories in the medial prefrontal cortex. Nat Hum Behav 4, 189–200 (2020). https://doi.org/10.1038/s41562-019-0779-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-019-0779-z
This article is cited by
-
The anterior thalamic nuclei: core components of a tripartite episodic memory system
Nature Reviews Neuroscience (2022)
-
Geometric models reveal behavioural and neural signatures of transforming experiences into memories
Nature Human Behaviour (2021)
-
Decoding individual identity from brain activity elicited in imagining common experiences
Nature Communications (2020)
-
Remembering together
Nature Human Behaviour (2019)