Abstract
Subsoils contain more than half of soil organic carbon (SOC) and are expected to experience rapid warming in the coming decades. Yet our understanding of the stability of this vast carbon pool under global warming is uncertain. In particular, the fate of complex molecular structures (polymers) remains debated. Here we show that 4.5 years of whole-soil warming (+4 °C) resulted in less polymeric SOC (sum of specific polymers contributing to SOC) in the warmed subsoil (20–90 cm) relative to control, with no detectable change in topsoil. Warming stimulated the subsoil loss of lignin phenols (−17 ± 0%) derived from woody plant biomass, hydrolysable lipids cutin and suberin, derived from leaf and woody plant biomass (−28 ± 3%), and pyrogenic carbon (−37 ± 8%) produced during incomplete combustion. Given that these compounds have been proposed for long-term carbon sequestration, it is notable that they were rapidly lost in warmed soils. We conclude that complex polymeric carbon in subsoil is vulnerable to decomposition and propose that molecular structure alone may not protect compounds from degradation under future warming.
Similar content being viewed by others
Main
Despite evidence that the predominant response of soils to global warming is an increase in heterotrophic respiration1,2,3, it remains uncertain how global warming will affect soil carbon stocks4. One source of uncertainty is a lack of process understanding of soil organic carbon (SOC) dynamics in subsoils (here defined as below 20 cm depth; details in Methods). Subsoils store more than half of global SOC5, and climate models project that subsoils will warm in near synchrony with air and topsoils6. Despite their large stock size, subsoils remain understudied and are largely underrepresented in warming studies.
Carbon dynamics in soils are regulated by multiple biotic and abiotic factors, many of which can differ between topsoil and subsoil7. For example, whereas above-ground litter may be an important carbon input to topsoil, roots become more important inputs with increasing soil depth8. Depending on biotic and abiotic factors, specific molecular structures might preferentially degrade or accumulate9, and some complex molecular structures have been proposed to remain in the soil on centennial timescales10,11,12,13. A potential climatic control on the molecular composition of SOC is temperature since temperature may affect relative decomposition rates. Simple kinetic theory predicts that decomposition of a complex substrate (higher activation energy) is more temperature sensitive compared with simple substrates14, if other biotic and abiotic processes are excluded15. A meta-analysis on enzymatic activity in warmed soils indicated that in situ warming stimulates enzymes responsible for the decomposition of complex substrates (ligninase) more than it does enzymes responsible for the decomposition of simple substrates (cellulase)16. If this temperature effect dominates the varying influences of biotic and abiotic factors on decomposition under future warming, complex molecular structures that are to date predicted to remain in soils for long periods could be especially vulnerable to decomposition.
One polymeric compound that is considered stable in most soils due to its polyaromatic chemical structure is pyrogenic carbon (PyC), derived from incomplete combustion processes17. However, recent findings challenge the millennial persistence of PyC in soils, showing that PyC turns over on a timescale of centuries13—with a mean residence time about 1.5 times that of bulk SOC11. The wood-derived polymer lignin, which is made up of phenolic structures, has been described and modelled as part of the recalcitrant carbon pool, but it too has been shown to readily degrade in soils12,18. Finally, plant-derived hydrolysable lipids cutin, derived from cuticular tissues of vascular plants19,20, and suberin, derived from woody tissues20, are being considered useful for carbon sequestration21 due to their chemical structure22 and the assumption that they have longer turnover times than does bulk SOC10. It is crucial to better understand how polymeric subsoil SOC is affected by increasing temperatures, more so when such polymeric compounds are being considered for carbon sequestration purposes17,21.
In this Article, we made use of one of the first multi-year, whole-soil-profile warming experiments, located at the University of California Blodgett Experimental Forest, to assess warming effects on the composition and degradation of SOC pools at different depths. Specifically, we assessed the fate of plant polymers and PyC after 4.5 years of continuous warming using molecular proxies (lignin, hydrolysable lipids and benzene polycarboxylic acids). The experimental set-up consists of three blocks of paired circular plots (detailed description by ref. 23). In warmed plots, vertical heating rods inserted to 2.4 m soil depth provided continuous warming of the soil profile to 1 m depth, in concert with diurnal and seasonal temperature variations24. The target warming magnitude was +4 °C based on Representative Concentration Pathway 8.5 predictions for the study region by 210025. This unique set-up allowed us to explore in situ soil carbon responses to warming. Previous work at the study site demonstrated substantial loss of bulk SOC stocks (by 33%) in the subsoil, and soil respiration was consistently increased at all depths over 4.5 years of warming26. Through polymer class-specific measurements in this experiment, we show that polymer classes that have been proposed as highly stable10,11,12,13 are in fact also vulnerable to loss with soil warming such as that expected over this century.
Large loss of subsoil polymeric SOC but not topsoil
We found large differences between topsoil and subsoil for the fate of polymeric SOC under experimental warming. In the topsoil between 0 and 20 cm depth, absolute concentrations of PyC, lignin and hydrolysable lipids (per gram soil) did not differ significantly between warmed and control plots (Fig. 1a–c). By contrast, in subsoil from 20 to 90 cm soil depth, soil warming led to a loss of polymeric carbon, highlighting the vulnerability of subsoil carbon to decomposition under climate change. After 4.5 years of warming, the absolute concentration of PyC (per gram soil) was 37 ± 8% lower in warmed subsoils (Fig. 1a; PnumDF1:denDF26 < 0.01). Similarly, the wood-derived compound lignin was 17 ± 0% lower (Fig. 1b; P1:26 < 0.01) in warmed compared with control subsoils. The observed losses of PyC and lignin were corroborated by spectroscopic observations from the same site, where aromatic compounds (indicative of PyC and lignin) were less abundant in warmed compared with control plots27. A loss of lignin is consistent with the observation that warming increases the activity of lignin-degrading enzymes16. Finally, hydrolysable lipids were 28 ± 3% lower (Fig. 1c; P1:24 = 0.01) in warmed compared with control subsoils. In subsoil, hydrolysable lipids can be composed predominantly of root-derived suberin8. Thus, the decreasing root mass with warming at our site27 parallels the decrease of hydrolysable lipids.
a–c, Concentrations of PyC (a), lignin (b) and hydrolysable lipids (c) per gram soil were not significantly different in the topsoil (0–20 cm) but were significantly lower in warmed compared with control subsoils (20–90 cm). Values shown are the differences between the warmed and control plots expressed as a percentage ((warmed – control)/control × 100%), with negative values in red and positive values in blue. Black squares show the mean (n = 3), and grey circles indicate the single data points. Error bars represent the standard error of the mean (s.e.m.).
No preferential preservation or loss of polymeric SOC
Our results show that polymeric SOC was lost at a similar order of magnitude as were bulk SOC stocks (by 33% (ref. 26)) due to warming. Concentrations of investigated polymers were strongly correlated with bulk SOC concentrations in both control and warmed subsoils (Fig. 2). Compound carbon concentrations normalized to SOC concentrations did not differ between warmed and control subsoils for PyC (Fig. 2a; P1:26 = 0.71), lignin (Fig. 2b; P1:26 = 0.18) or hydrolysable lipids (Fig. 2c; P1:26 = 0.62). Thus, our results suggest that there was no preferential loss or preservation of any of these polymers.
a, PyC (control: r = 0.91, P < 0.001, n = 21; warmed: r = 0.88, P < 0.001, n = 21). b, Lignin (control: r = 0.79, P < 0.001, n = 21; warmed: r = 0.80, P < 0.001, n = 21). c, Hydrolysable lipids (control: r = 0.84, P < 0.001, n = 21; warmed: r = 0.91, P < 0.001, n = 21). Lines show the two-tailed linear regression of correlation for individual non-transformed data points (n = 21), and the grey shaded areas are the confidence intervals (0.95). Regression coefficients and P values are given in the respective figures. The blue colours represent information for control subsoil; red represents the warmed subsoil.
Different SOC dynamics in topsoils and subsoils under warming
We report a rapid decrease in concentrations of PyC, lignin and the hydrolysable lipids in subsoils due to in situ experimental warming. These polymeric substances of SOC have been assumed to persist and degrade slower than bulk SOC10,11,12,13. However, this might vary between ecosystems and soil properties9. Importantly, whereas our results show that polymeric C is not preserved preferentially in subsoil with warming, we did not observe a significant effect in topsoil. Thus, the key strength of our method is the uniform warming of the whole profile, which better reflects predictions of whole-soil warming6. Nonetheless, experimental warming usually entails limitations due to the experimental set-up, such as effects of temperature magnitude, duration of warming or concomitant drying28,29. Some of these limitations will probably also apply for our experimental set-up. For example, at our experimental site, warming caused a slight drying of the topsoil only26, which might confound the observed effect of warming.
To our knowledge, only one study has reported changes in polymeric carbon (lignin, suberin and cutin) in warmed subsoil, using infrared heaters18. This study on the Tibetan plateau, China, showed an accumulation of suberin and a loss of lignin, potentially due to a shorter period of soil freezing, which allowed for deeper root penetration and enhanced lignin degradation18. At our experimental site, root mass was lower in the warmed plots27, which could indicate fewer root inputs or faster root turnover. If warming-induced losses of subsoil SOC26 are not replaced by, for example, more root growth18, then low SOC concentrations in the subsoil might trigger microorganisms to utilize or assimilate complex substrates that might not have been degraded otherwise; alternatively, the stimulation of decomposition enzymes by warming could increase microbial decomposition of all substrates. Likewise, increased fluxes of dissolved organic carbon in warmed plots at our site26 might have stimulated the decomposition of polymeric carbon by flushing fresh substrate to the subsoil and alleviating previous resource limitations30. Our results highlight that not only simple and labile compounds but also complex polymeric SOC in subsoil might be lost in a warming world due to the higher microbial activity.
Some experiments have investigated the fate of lignin and hydrolysable lipid concentrations under warming in topsoil. Lignin was lost from warmed topsoils in two experiments31,32,33 in contrast to our results of no significant effect. At another experimental site, lignin in topsoil decreased only after more than 20 years of soil warming; this was attributed to changing substrate availability34. It remains to be investigated how lignin in topsoil is affected at our site in the long term. The results for hydrolysable lipids are more contrasting. In a hardwood forest, suberin was not affected by 14 months of soil warming, whereas cutin concentrations increased31, leading to cutin being considered a ‘heat-proof compound’35. In a mixed hardwood forest experiment, concentrations of cutin and suberin significantly decreased after 4 years of soil warming32, but these trends were transient and no longer significant after 10 years of warming33. Differences in topsoil responses among ecosystems could be related to differences in litter properties. For example, differences in vegetation, among deciduous31, mixed hardwood32,33 and coniferous forests (our site), result in litter inputs to organic horizons with very different properties and thus degradability. Suseela et al. 36 showed that the litter type affects how decomposition responds to warming, which in turn will affect inputs from the litter horizon to topsoil. Besides explaining differences among ecosystems, increasing inputs from the litter horizon to the topsoil at our site could be partially responsible for the difference in how warming affected carbon cycling in the topsoil versus the subsoil.
Polymeric SOC as an available pool under climate warming
We observed a rapid loss of complex polymers in subsoil due to experimental warming, and our observations indicate that warming stimulated SOC loss at the same rate for PyC, lignin and hydrolysable lipids as for bulk SOC (Fig. 2). The vulnerability to decomposition of SOC in the soil matrix is controlled by a range of biotic and abiotic ecosystem properties37 and not only kinetics of individual enzymatic reactions. For example, the physical state of soil organic matter, as free particulate organic matter (POM), occluded POM or mineral-associated organic matter, affects decomposition rates38,39, but little is known about temperature sensitivity. At the experimental site, primarily free POM in subsoils was lost, whereas stocks of occluded and mineral-associated carbon did not change with warming26. Complex plant-derived compounds might dominantly contribute to POM12,40; thus, the fire- and plant-derived polymers lost from these warmed subsoils might have been part of the POM fraction. In the first years of warming, the respired carbon was primarily of young 14C age, which aligns well with POM as a primary source23. We hypothesize that polymeric SOC will be vulnerable to enhanced decomposition in warmer subsoil because, or if, it is largely found in the unprotected POM.
We highlight the importance of separately considering topsoils and subsoils in predictions of SOC dynamics under warming because the temperature sensitivity of different SOC compounds varies with soil depth. None of the polymeric compounds analysed here proved heat-proof in warmed subsoil, and these compounds decomposed in synchrony with bulk SOC. These findings agree with the paradigm that there is no inherently stable SOC but that the stability depends on ecosystem properties37, which also change with soil depth. This study provides crucial knowledge for understanding the long-term fate of compounds that are being considered in carbon sequestration approaches.
Methods
Experimental set-up and sampling
The whole-soil warming experiment at the University of California Blodgett Experimental Forest (120°39ʹ40″ W; 38°54ʹ43″ N) was sampled after 4.5 years of whole-soil warming. The experiment was designed to study changes in biogeochemical cycles in a warming climate23. The climate is characterized as Mediterranean with mean annual precipitation of 1,660 mm and mean annual air temperature of 12.5 °C (ref. 26). The experiment is located on an Alfisol of granitic origin, and the soil is covered by an organic horizon. The vegetation in the coniferous forest is dominated by ponderosa pine (Pinus ponderosa), sugar pine (Pinus lambertiana), incense cedar (Calodefrus decurrens), white fir (Abies concolor) and Douglas fir (Pseudotsuga menziesii), with understory vegetation consisting of shrubs (mainly Notholithocarpus densiflorus), grasses and some other annual plants (for example, Gallium triflorum). The experimental set-up has been previously described in detail (ref. 23). Briefly, three blocks of paired circular plots (n = 3) were set up in October 2013. Plots measure 3 m in diameter, and temperature was elevated in the warmed compared with control plots by 4 °C from 0.2 to 1 m and 2.6 °C above 0.2 m due to surface heat loss. The experimental design preserved diurnal and seasonal temperature variations depth-specifically. Samples were collected in April 2018, after 4.5 years of warming, using a manual soil corer of 4.78 cm diameter. The organic horizons were removed before soil sampling. The mineral soil was sampled in increments of 10 cm to a soil depth of 90 cm. After recovery, the samples were stored cool for transportation and freeze-dried within a week after sampling. Roots were manually recovered from the cores, and the soil was sieved to 2 mm. Ground subsamples were used to measure carbon concentrations on an elemental analyser coupled to an isotope ratio mass spectrometer (EA-IRMS; FLASH 2000 HT Plus, linked by ConFlo IV to DELTA V Plus IRMS; Thermo Fisher Scientific).
The differentiation between topsoil and subsoil at a depth of 20 cm is based on the observation by ref. 26 that carbon stocks are significantly different between depth increments from 0 to 20 cm and 20 to 90 cm. Furthermore, using principal component analysis with the R function prcomp (54 data points with 4 variables, centred and scaled), depth increments from 0 to 20 cm were clearly separated from the ones from 20 to 90 cm (Extended data Fig. 1).
Biomarker analysis
Pyrogenic carbon
Benzene polycarboxylic acids were used as an approximation of PyC, and the analysis was conducted following the protocol by Wiedemeier et al. (2016) (ref. 41) with some modifications. The dried and milled sample containing approximately 10 mg OC was weighed in digestion tubes and digested with nitric acid (65%) at 170 °C for 8 h. A glass fibre filter was used for filtration of the resulting solution. In a next step, the sample was passed over a cation exchange resin for further cleaning and subsequently freeze-dried. The sample was then redissolved in MeOH/H2O (1/1), passed over a solid-phase extraction cartridge (C18 SPE tube, Supelco), dried again and dissolved in deionized water for the transfer to a high-performance liquid chromatography vial. Separation and quantification were conducted on an Agilent 1290 Infinity high-performance liquid chromatography system (Santa Clara) equipped with an Agilent InfinityLab Poroshell 120 SB-C18 column (100 mm × 4.6 mm × 2.7 mm) and coupled to a photo diode array detector. Ortho-phosphoric acid (Honeywell) dissolved in water (pH 1.2–1.3) was used as mobile phase A, and pure acetonitrile (Scharlau) was used as mobile phase B. For quantification, all acids with the different numbers of carboxyl functions were summed (B3CA, B4CA, B5CA and B6CA, respectively).
Lignin
Lignin was analysed by alkaline CuO oxidation using microwave digestion to break down the lignin structure into monomers as described in refs. 42,43 with some modifications. Briefly, all dried and milled soil samples were analysed in duplicate, and an equivalent of 2–5 mg total organic carbon was weighed into the microwave tubes. After adding 500 mg CuO powder, 50 mg ferrous ammonium sulfate and 20 ml of 2 M NaOH solution, samples were oxidized in the microwave at 150 °C for 90 min. Once cooled, 500 μl of a mixture containing cinnamic acid and ethylvanillin in NaOH (50 mg l−1 each) was added as internal standard to each sample, and subsequently the pH was adjusted to pH 2.10 using HCl. Samples were then collected on preconditioned C18 columns (DISCOVERY DSC-18 SPE tube, 500 mg; Sigma-Aldrich), eluted 5 times with 500 μl ethyl acetate, dried under N2 and redissolved in 400 μl p-anisic acid in ethyl acetate (50 mg l–1). Derivatization was done by mixing 70 μl sample with 70 μl N,O-bis (trimethylsilyl) trifluoroacetamide/tetramethylchlorosilane (BSTFA + TCMS; 99/1; Sigma-Aldrich) and heating for 20 min at 60 °C. Compounds were then quantified on an Agilent 7890B gas chromatograph (GC) equipped with a 50 m J&W DB-5ms GC column, a multi-mode injector and a flame ionization detector. Compound identification was supported by external standard and sample measurements on an Agilent 6890 N GC equipped with a split/splitless injector coupled to an Agilent 5973 mass selective detector. Both instruments were equipped with DB-5MS column (50 m × 0.2 mm × 0.33 μm) and 1.5 m de-activated pre-column, with helium as the carrier gas (1 ml min−1). The temperature programme was starting at 80 °C and kept isothermal for 5 min, increasing to 110 °C at a rate of 2 °C min−1, increasing further to 170 °C at a rate of 0.5 °C min−1 and at a rate of 15 °C min−1 to 320 °C, where temperature was held again for 10 min. The total concentration of lignin was calculated by summing eight measured oxidation products (vanillin, acetovanillone, vanillic acid, syringaldehyde, acetosyringone, syringic acid, p-coumaric acid and ferulic acid).
Suberin and cutin
Cutin and suberin biomarkers were isolated by base hydrolysis to release bound lipids after removal of free lipids, as described in Mendez-Millan et al. (2011) (ref. 44) with some modifications. All samples were analysed in triplicate. Each sample was refluxed for 18 h at 85–88 °C in purified water: MeOH (1/9 v/v) with 6% KOH. On the same day, the sample was filtered and then stored overnight in the refrigerator. Thereafter, water was added and the pH adjusted to pH 2.0 with HCl. Target compounds were then extracted a total of five times with dichloromethane by phase separation, combining the organic phase with the target compounds in a round-bottom flask. To ensure complete removal of remaining water, the sample was passed over Na2SO4 and dried under N2. Before GC analysis, deuterated eicosanoic acid (D39C20; Cambridge Isotope Laboratories, Inc.) was added to the sample as an internal standard, followed by silylation at 80 °C for 1 h using N,O-Bis-(trimethylsilyl)-acetamid (BSA; Merck) as described in ref. 45. Compounds were then quantified using the same GC system as for lignin. The temperature programme was starting at 50 °C and kept isothermal for 4 min, increasing to 150 °C at a rate of 4 °C min−1 and at a rate of 3 °C min−1 to 320 °C, where temperature was held again for 40 min. Differentiation of compounds specific for woody (suberin) and leaf (cutin) inputs was not possible in a meaningful way. On the one hand, the above-ground grass biomass contained several compounds in high concentrations, which are usually used to characterize woody inputs (C22diacid, ωOHC18-ωOHC24), leaving only two compounds for the characterization of below-ground inputs (C18 and C20 diacids). On the other hand, there were only two markers specific for leaf inputs, which were additionally of very low concentrations (9 or 10, ωdiOHC16 and ωOHC14). Thus, only the sum of all hydrolysable lipids is shown.
Data analysis
All statistical analyses were conducted using R version 4.1.146 on the RStudio interface47. Normality, homoscedasticity and model fit were assessed using residual plots, Shapiro–Wilk test and Levene test. Outliers were assessed using box plots, rosnerTest in the EnvStats package and residual plots. The effect of outlier exclusion was tested by comparing model fit with and without outliers. Data were log transformed to improve model fit. The warming effect was tested using mixed-effect models using gls and lme functions in the nlme package. Restricted maximum likelihood was used with the random-effect block (n = 3) and the fixed-effects treatment, depth and their interaction. As previous results26 indicated more-pronounced effects in the subsoil, models were run on a subset of depth increments (below 20 cm), which is specified when we report the results. We used a significance level of P < 0.05. Pearson correlation was calculated using cor.test function as a two-tailed correlation on non-transformed individual data points.
Data availability
The data used in this study are available from ESS-DIVE repository (https://doi.org/10.15485/1915661).
Code availability
We used no unreported custom computer code or algorithm.
References
Lu, M. et al. Responses of ecosysem carbon cycle to experimental warming: a meta-analysis. Ecology 94, 726–738 (2013).
Rustad, L. et al. A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126, 543–562 (2001).
Song, J. et al. A meta-analysis of 1,119 manipulative experiments on terrestrial carbon-cycling responses to global change. Nat. Ecol. Evol. 3, 1309–1320 (2019).
Bradford, M. A. et al. Managing uncertainty in soil carbon feedbacks to climate change. Nat. Clim. Change 6, 751–758 (2016).
Jobbagy, E. G. & Jackson, R. B. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol. Appl. 10, 423–436 (2000).
Soong, J. L., Phillips, C. L., Ledna, C., Koven, C. D. & Torn, M. S. CMIP5 models predict rapid and deep soil warming over the 21st century. J. Geophys. Res. Biogeosci. 125, e2019JG005266 (2020).
Rumpel, C. & Kögel-Knabner, I. Deep soil organic matter—a key but poorly understood component of terrestrial C cycle. Plant Soil 338, 143–158 (2011).
Spielvogel, S. et al. Distribution of cutin and suberin biomarkers under forest trees with different root systems. Plant Soil 381, 95–110 (2014).
Hall, S. J., Ye, C., Weintraub, S. R. & Hockaday, W. C. Molecular trade-offs in soil organic carbon composition at continental scale. Nat. Geosci. 13, 687–692 (2020).
Feng, X., Xu, Y., Jaffé, R., Schlesinger, W. H. & Simpson, M. J. Turnover rates of hydrolysable aliphatic lipids in Duke Forest soils determined by compound specific 13C isotopic analysis. Org. Geochem. 41, 573–579 (2010).
Lutfalla, S. et al. Pyrogenic carbon lacks long-term persistence in temperate arable soils. Front. Earth Sci. 5, 2017.00096 (2017).
Thevenot, M., Dignac, M. F. & Rumpel, C. Fate of lignins in soils: a review. Soil Biol. Biochem. 42, 1200–1211 (2010).
Wang, J., Xiong, Z. & Kuzyakov, Y. Biochar stability in soil: meta-analysis of decomposition and priming effects. Glob. Change Biol. Bioenergy 8, 512–523 (2016).
Arrhenius, S. Über die Reaktionsgeschwindigkeit bei der Inversion von Rohrzucker durch Säuren. Z. f.ür. Phys. Chem. 4, 226–248 (1889).
Davidson, E. A. & Janssens, I. A. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440, 165–173 (2006).
Chen, J. et al. Soil carbon loss with warming: new evidence from carbon‐degrading enzymes. Glob. Change Biol. https://doi.org/10.1111/gcb.14986 (2020).
Lehmann, J. et al. Biochar in climate change mitigation. Nat. Geosci. 14, 883–892 (2021).
Jia, J. et al. Climate warming alters subsoil but not topsoil carbon dynamics in alpine grassland. Glob. Change Biol. 25, 4383–4393 (2019).
Goñi, M. A. & Hedges, J. I. The diagenetic behavior of cutin acids in buried conifer needles and sediments from a coastal marine environment. Geochim. Cosmochim. Acta 54, 3083–3093 (1990).
Kolattukudy, P. E. Biopolyester membranes of plants: cutin and suberin. Science 208, 990–1000 (1980).
Harman-Ware, A. E., Sparks, S., Addison, B. & Kalluri, U. C. Importance of suberin biopolymer in plant function, contributions to soil organic carbon and in the production of bio-derived energy and materials. Biotechnol. Biofuels Bioprod. 14, 75 (2021).
Lorenz, K., Lal, R., Preston, C. M. & Nierop, K. G. J. Strengthening the soil organic carbon pool by increasing contributions from recalcitrant aliphatic bio(macro)molecules. Geoderma 142, 1–10 (2007).
Hicks Pries, C. E., Castanha, C., Porras, R. C. & Torn, M. S. The whole-soil carbon flux in response to warming. Science 355, 1420–1423 (2017).
Hanson, P. J. et al. A method for experimental heating of intact soil profiles for application to climate change experiments. Glob. Change Biol. 17, 1083–1096 (2011).
IPCC Climate Change 2021: The Physical Science Basis (eds Masson-Delmotte, V. et al.) (Cambridge Univ. Press, 2021).
Soong, J. L. et al. Five years of whole-soil warming led to loss of subsoil carbon stocks and increased CO2 efflux. Sci. Adv. 7, abd1343 (2021).
Ofiti, N. O. E. et al. Warming promotes loss of subsoil carbon through accelerated degradation of plant-derived organic matter. Soil Biol. Biochem. 156, 108185 (2021).
Romero-Olivares, A. L., Allison, S. D. & Treseder, K. K. Soil microbes and their response to experimental warming over time: A meta-analysis of field studies. Soil Biol. Biochem. 107, 32–40 (2017).
Chen, J. et al. Stronger warming effects on microbial abundances in colder regions. Sci. Rep. 5, 18032 (2015).
Fontaine, S. et al. Stability of organic carbon in deep soil layers controlled by fresh carbon supply. Nature 450, 277–280 (2007).
Feng, X., Simpson, A. J., Wilson, K. P., Dudley Williams, D. & Simpson, M. J. Increased cuticular carbon sequestration and lignin oxidation in response to soil warming. Nat. Geosci. 1, 836–839 (2008).
Pisani, O., Frey, S. D., Simpson, A. J. & Simpson, M. J. Soil warming and nitrogen deposition alter soil organic matter composition at the molecular-level. Biogeochemistry 123, 391–409 (2015).
VandenEnden, L., Anthony, M. A., Frey, S. D. & Simpson, M. J. Biogeochemical evolution of soil organic matter composition after a decade of warming and nitrogen addition. Biogeochemistry 156, 161–175 (2021).
Pold, G., Grandy, A. S., Melillo, J. M. & DeAngelis, K. M. Changes in substrate availability drive carbon cycle response to chronic warming. Soil Biol. Biochem. 110, 68–78 (2017).
Prescott, C. Heat-proof carbon compound. Nat. Geosci. 1, 815–816 (2008).
Suseela, V., Tharayil, N., Xing, B. & Dukes, J. S. Labile compounds in plant litter reduce the sensitivity of decomposition to warming and altered precipitation. N. Phytol. 200, 122–133 (2013).
Schmidt, M. W. I. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56 (2011).
Hartley, I. P., Hill, T. C., Chadburn, S. E. & Hugelius, G. Temperature effects on carbon storage are controlled by soil stabilisation capacities. Nat. Commun. 12, 6713 (2021).
Rocci, K. S., Lavallee, J. M., Stewart, C. E. & Cotrufo, M. F. Soil organic carbon response to global environmental change depends on its distribution between mineral-associated and particulate organic matter: a meta-analysis. Sci. Total Environ. 793, 148569 (2021).
Lavallee, J. M., Soong, J. L. & Cotrufo, M. F. Conceptualizing soil organic matter into particulate and mineral-associated forms to address global change in the 21st century. Glob. Change Biol. 26, 261–273 (2020).
Wiedemeier, D. B. et al. Characterization, quantification and compound-specific isotopic analysis of pyrogenic carbon using benzene polycarboxylic acids (BPCA). J. Vis. Exp. https://doi.org/10.3791/53922 (2016).
Goñi, M. A. & Montgomery, S. Alkaline CuO oxidation with a microwave digestion system: lignin analyses of geochemical samples. Anal. Chem. 72, 3116–3121 (2000).
Hedges, J. I. & Ertel, J. R. Characterization of lignin by gas capillary chromatography of cupric oxide oxidation products. Anal. Chem. 54, 174–178 (1982).
Mendez-Millan, M., Dignac, M. F., Rumpel, C. & Derenne, S. Can cutin and suberin biomarkers be used to trace shoot and root-derived organic matter? A molecular and isotopic approach. Biogeochemistry 106, 23–38 (2011).
Wiesenberg, G. L. B. & Gocke, M. I. in Hydrocarbon and Lipid Microbiology Protocols (eds McGenity, T. et al.) 61–91 (Springer, 2017); https://doi.org/10.1007/8623_2015_157
R Core Team. R: A Language and Environment for Statistical Computing, version 4.1.1 (R Core Team, 2021).
R Studio Team. RStudio: Integrated Development Environment for R, version 2021.09.0+351 (R Studio Team, 2021).
Acknowledgements
The research was supported by Swiss National Science Foundation (SNF) project 172744 (DEEP C) awarded to M.W.I.S. and US Department of Energy, Office of Science, Office of Biological and Environmental Research under contract number DE-AC02-05CH11231 awarded to M.S.T. We thank C. Castanha (LBL), J. Soong (LBL), and E. Solly (UZH) for field assistance and T. Speckert (UZH), G. Santilli (UZH) and Y. Brügger (UZH) for laboratory assistance. The University Research Priority Program Global Change and Biodiversity (URPP-GCB) at the University of Zurich offered opportunities for helpful discussions and exchange.
Funding
Open access funding provided by University of Zurich.
Author information
Authors and Affiliations
Contributions
M.S.T. designed and maintained the warming field experiment. M.W.I.S. conceived the DEEP C project. C.U.Z. carried out biogeochemical analyses, supervised by G.L.B.W. N.O.E.O. assisted with statistical analysis. All co-authors participated in the field campaign and contributed to data interpretation and to the manuscript written by C.U.Z.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Peer review
Peer review information
Nature Geoscience thanks Ji Chen and Bruce Hungate for their contribution to the peer review of this work. Primary Handling Editor: Xujia Jiang, in collaboration with the Nature Geoscience team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
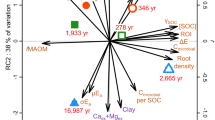
Extended Data Fig. 1 Principal component analysis conducted on SOC, lignin, hydrolysable lipids and pyrogenic carbon.
Principal component analysis conducted on SOC, lignin, hydrolysable lipids and pyrogenic carbon, showing a clear separation of the increments from 0–20 cm and 20–90 cm. (a) Score plot of the individual soil depth and treatments (n = 54) and (b) the corresponding loading plot of the variables (n = 54). PC1 and PC2 explained 85.4% and 8.1% of the variance, respectively.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zosso, C.U., Ofiti, N.O.E., Torn, M.S. et al. Rapid loss of complex polymers and pyrogenic carbon in subsoils under whole-soil warming. Nat. Geosci. 16, 344–348 (2023). https://doi.org/10.1038/s41561-023-01142-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-023-01142-1
This article is cited by
-
Subsoil carbon loss
Nature Geoscience (2023)