Abstract
Salinity-driven density stratification of the upper Arctic Ocean isolates sea-ice cover and cold, nutrient-poor surface waters from underlying warmer, nutrient-rich waters. Recently, stratification has strengthened in the western Arctic but has weakened in the eastern Arctic; it is unknown if these trends will continue. Here we present foraminifera-bound nitrogen isotopes from Arctic Ocean sediments since 35,000 years ago to reconstruct past changes in nutrient sources and the degree of nutrient consumption in surface waters, the latter reflecting stratification. During the last ice age and early deglaciation, the Arctic was dominated by Atlantic-sourced nitrate and incomplete nitrate consumption, indicating weaker stratification. Starting at 11,000 years ago in the western Arctic, there is a clear isotopic signal of Pacific-sourced nitrate and complete nitrate consumption associated with the flooding of the Bering Strait. These changes reveal that the strong stratification of the western Arctic relies on low-salinity inflow through the Bering Strait. In the central Arctic, nitrate consumption was complete during the early Holocene, then declined after 5,000 years ago as summer insolation decreased. This sequence suggests that precipitation and riverine freshwater fluxes control the stratification of the central Arctic Ocean. Based on these findings, ongoing warming will cause strong stratification to expand into the central Arctic, slowing the nutrient supply to surface waters and thus limiting future phytoplankton productivity.
Similar content being viewed by others
Main
The Arctic climate system is changing rapidly1, as exemplified by dramatic declines in sea-ice extent, concentration and thickness over the past four decades1,2. Resultant increases in the area and seasonal duration of open water are associated with increased Arctic Ocean biological productivity3. Climate model simulations suggest that the Arctic Ocean could be seasonally ice-free within decades4, with expected impacts on the biological productivity5,6,7,8 of the Arctic Ocean and climate1.
Density stratification from salinity is fundamental to Arctic Ocean circulation, sea-ice cover and productivity9. Cold and fresh surface waters of the polar mixed layer (PML) are separated from warmer and saltier subsurface waters of Atlantic and Pacific origin by the ‘halocline’9, which isolates the PML from subsurface heat and nutrients. Excess precipitation relative to evaporation, river discharge and the advection of low-salinity ocean waters across the Bering Strait are each thought to contribute to the Arctic Ocean’s salinity stratification10,11,12. Over recent decades, this stratification has weakened in the eastern Arctic (Eurasian Basin) and strengthened in the western Arctic (Amerasian Basin)13,14. Whether or not these trends will continue into the future—and how sea-ice coverage and productivity will be impacted—is uncertain5,6,7,8,15,16.
To gain insight into past Arctic Ocean stratification, we reconstructed the sources and degree of biological consumption of nitrogen (N), the limiting nutrient for summertime Arctic productivity5,17, at three locations in the open (that is, off-shelf) western and central Arctic Ocean over the past 35,000 years (Fig. 1a). We analysed the N isotopic composition of organic matter (expressed as δ15N = [(15N/14N)sample/(15N/14N)air − 1] × 1,000) within the shell walls of the planktonic foraminifer Neogloboquadrina pachyderma (hereafter, δ15NN.p.). The δ15NN.p. value reflects the δ15N of organic matter produced in surface waters, which itself depends on the δ15N of the subsurface nitrogen supply (which is as nitrate, NO3−) and the degree of nitrate consumption, that is, the summertime drawdown of nitrate as a proportion of the annual nitrate supply (Methods). In contrast to bulk-sediment δ15N, δ15NN.p. is protected from early bacterial diagenesis and terrestrial organic matter contamination (Extended Data Fig. 1).
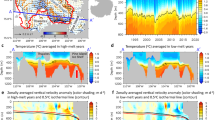
a, Schematic circulation of AHW (in blue) and PHW (in pale red)18. The coloured circles and diamonds indicate sediment cores and water-column nitrate δ15N profiles, respectively. The white asterisk denotes the location of the NGRIP ice core45; the white diamond, square and triangle and the black cross denote the locations of Holocene sea-ice reconstructions (Extended Data Fig. 2). b, Cross-section along the grey dashed line in a of nitrate concentration (coloured) and salinity S (black contours, interval 0.5 psu) from AOS94 data19. Vertical dashed lines indicate the locations of nitrate isotope data from ARC01 stations 10, 14, 19, 26, 30 and 32 (in c). c, Arctic Ocean nitrate δ15N from ARC01 (for colour code see a). Vertical blue and red shading indicates the average δ15N values for AHW and PHW nitrate, respectively. The error bar is the average measurement 1 s.d. (Methods). Panels a and b are plotted using ODV51.
Arctic hydrography and nitrogen isotopes
Water and nitrate are supplied to the upper Arctic Ocean from distinct Atlantic and Pacific sources (Fig. 1a). Atlantic-sourced halocline water (AHW) originates from inflow through the Fram Strait and the Barents Sea and dominates the eastern and central Arctic Ocean up to the Mendeleev Ridge in the Amerasian Basin5,18,19,20. In the far western Arctic Ocean, AHW is covered by fresher Pacific-sourced halocline water (PHW), which forms in the Chukchi Sea from inflow across the approximately 50-m-deep Bering Strait5,21. The northern boundary of PHW is at the Mendeleev Ridge today18 (Fig. 1a) but appears to shift with the Arctic Oscillation, having reached the Lomonosov Ridge in the 1980s22.
Salinity-driven density stratification determines two critical aspects of Arctic Ocean biological nitrate consumption. First, the stratification constrains the upward flux of nitrate to the PML, which occurs mostly in the winter23. Second, the stratification sets the depth of the summertime PML24 relative to the depth of the euphotic zone, which impacts the light conditions for phytoplankton. These features are illustrated by comparing the summertime PML characteristics between the Amerasian and Eurasian basins (Fig. 1b). In the Amerasian Basin, a thin (≤10 m)24, low-salinity PML with near-zero nitrate concentration overlies the PHW, reflecting the weakness of wintertime nitrate resupply across the strong salinity gradient5,17,20,21,23. By contrast, the salinity gradient in the Eurasian Basin is less extreme. Accordingly, the Eurasian Basin summertime PML is deeper (~20 m)24, and PML nitrate concentrations overlying the AHW are higher (3–10 µM), reflecting the higher nitrate flux across the weaker halocline17,23.
AHW and PHW differ in their nitrate characteristics. In AHW, nitrate occurs at a concentration of ~10 µM and with a δ15N of 5‰ (Fig. 1c), equivalent to high-latitude North Atlantic subsurface source waters25,26. By contrast, PHW exhibits a higher nitrate concentration of ~15 µM and a δ15N of 8‰ (Fig. 1c). The PHW nitrate δ15N reflects that of North Pacific nitrate (~6.5‰)27, with further isotopic enrichment by organic matter regeneration and benthic nitrogen loss on the Bering, Chukchi and East Siberian sea shelves26,27,28,29.
The origin of the nitrate (Atlantic versus Pacific) and the degree of nitrate consumption in the PML both affect the δ15NN.p.. Based on previous work, the δ15NN.p. is 2‰ higher than the δ15N of nitrate consumed in the euphotic zone30. This implies a δ15NN.p. value of 7 ± 0.3‰ for complete consumption of AHW nitrate and 10 ± 0.3‰ for complete consumption of PHW nitrate (errors are 1 s.d.; Methods). In the central Arctic, the coretop δ15NN.p. value of 6.1‰ at site B28 on the Lomonosov Ridge is lower than the value expected for complete consumption of AHW nitrate (blue line, Fig. 2), consistent with incomplete nitrate consumption in the central Arctic today17,23 (Fig. 1b). In the western Arctic, the coretop δ15NN.p. value of 8.3‰ at the southerly site B8 and 8.0‰ at the northerly site B17 are intermediate between the values expected for complete consumption of AHW and PHW nitrate (Fig. 2). Given complete western Arctic nitrate consumption today17,23 (Fig. 1b), we infer that these δ15NN.p. values indicate complete consumption of a mixture of PHW and AHW nitrate sources at the core sites. Mixing of AHW and PHW is probable at these sites on the Mendeleev Ridge, as they are overlain by the PHW–AHW boundary today18 (Fig. 2a). Moreover, mutidecadal shifts in PHW–AHW boundary position18,22 will change the influence of the PHW and AHW at these locations, and the coretop δ15NN.p. integrates over ~1,000 years.
a, Close-up bathymetric map of core locations, plotted using ODV51. The black dashed line denotes the boundary between the central and western Arctic Ocean along the Lomonosov Ridge. Blue and pale red lines trace the AHW and PHW pathways18 as in Fig. 1a. b, Horizontal blue and pale red bars indicate the expected δ15NN.p. from complete consumption of AHW and PHW nitrate, respectively, with the δ15NN.p. elevated by 2‰ above nitrate δ15N (Methods). The intermediate western Arctic δ15NN.p. between AHW and PHW nitrate indicates complete consumption of a mixture of AHW and PHW nitrate at these locations. The central Arctic δ15NN.p. is lower than AHW nitrate δ15N, consistent with incomplete consumption of nitrate in the modern central Arctic surface.
The downcore δ15NN.p. shows three distinct intervals since 35,000 years ago (35 ka; Fig. 3a) corresponding to the late Holocene (5–0 ka), the early and middle Holocene (11–5 ka) and the last glacial period and deglaciation (35–11 ka) (see age models in Extended Data Fig. 3). During the last glacial period and deglaciation, the western Arctic δ15NN.p. values (5.9 ± 0.7‰ at B17; 5.1 ± 0.6‰ at B8) were equal to or lower than the central Arctic δ15NN.p. (6.0 ± 0.5‰ at B28). At the end of the deglaciation, the δ15NN.p. increased significantly at all locations (statistical tests in Supplementary Table 1), but the western Arctic δ15NN.p. increase was around three times greater than in the central Arctic. During the Holocene, the western Arctic δ15NN.p. values of 7.7–8.7‰ were around 1–3‰ higher than the central Arctic δ15NN.p. values. Whereas there is no apparent Holocene trend in the western Arctic δ15NN.p., the central Arctic δ15NN.p. declined significantly after 5 ka (Fig. 3a and Supplementary Table 1).
a, Downcore δ15NN.p.; colours are as in Fig. 2. The horizontal grey bar is the expected δ15NN.p. from complete consumption of AHW nitrate. Error bars are ±1 s.d. from sample replicates (average ±0.3‰, 1 s.d.). b, Global ice volume equivalent sea level (ESL)31 (light red) and relative sea level (RSL) reconstructed at the Bering Strait33 (dark red). The horizontal dashed line indicates the modern depth of the Bering Strait. c, 75° N peak summer (21st June) insolation52. d, Ice δ18O, a climate proxy, at the NGRIP ice core45 (Fig. 1a). The vertical blue and red bars denote, respectively, the Younger Dryas (YD) event and the proposed timing of postglacial Bering Strait flooding (BSF)33,34.
Following modern observations, we interpret past δ15NN.p. values to reflect changes in both nitrate sources and the completeness of nitrate consumption, with the latter modulated by the strength of salinity-driven density stratification.
Past Arctic Ocean nitrate sources
The expanded ice sheets of the last glacial period drove a lower global mean sea level31, exposing the Arctic shelves and the Bering Strait32,33 (Fig. 3b). Under this configuration, Pacific waters could not flow into the Arctic, and the lower western Arctic δ15NN.p. value in part reflected the replacement of PHW nitrate with AHW nitrate.
The postglacial flooding of the Bering Strait has been dated to 13–11 ka, with complete inundation occurring after 11.5 ka (refs. 33,34) (Fig. 3b). Accordingly, the rapid increase in the western Arctic δ15NN.p. values at ~11 ka probably records the resumption of Pacific water inflow due to Bering Strait flooding. Furthermore, the highest Holocene western Arctic δ15NN.p. values occurred within around 1,000 years following flooding of the Bering Strait. This suggests that benthic nitrogen loss on the Bering, Chukchi and East Siberian sea shelves, which contributes to the high δ15N of modern PHW nitrate26,27,28,29, resumed rapidly upon Bering Strait flooding and did not require full inundation of the Arctic shelves, which was not completed until around 5 ka (ref. 35).
The modern central Arctic is not notably influenced by PHW nitrate (Fig. 1c) and could not have been during the last ice age when the Bering Strait was exposed. Moreover, we assume that the δ15N of AHW nitrate has not changed over the study period, as supported by records from the North Atlantic (Methods; Extended Data Fig. 4). Thus, the δ15NN.p. of the central Arctic site B28 was probably not substantially affected by source nitrate changes.
Completeness of nitrate consumption
In the case of nitrate supply from AHW, a δ15NN.p. value of less than 7‰ indicates incomplete nitrate consumption (Fig. 2). During the last ice age, when AHW nitrate was the only nitrate source to the Arctic Ocean, the δ15NN.p. of 5–6‰ at all sites (Fig. 3a) indicates incomplete consumption throughout the Arctic Ocean. During the deglaciation, complete nitrate consumption first commenced at sites B17 and B28 at around 13 ka, as indicated by δ15NN.p. values of approximately 7‰. This timing corresponds to a deglacial meltwater pulse from the Mackenzie River into the Arctic Ocean36,37 (Extended Data Fig. 2). The event was not captured at site B8. At site B8, complete nitrate consumption commenced by around 11 ka, coincident with flooding of the Bering Strait (Fig. 3).
Over the Holocene, the δ15NN.p. values at sites B8 and B17 suggest that complete nitrate consumption persisted in the western Arctic; we propose that the ~1‰ variations in δ15NN.p. indicate changes in the relative proportions of PHW and AHW at these locations instead of consumption changes (Methods; Extended Data Fig. 5). In the central Arctic, δ15NN.p. at site B28 follows boreal summer insolation across the Holocene (Fig. 3c). These data indicate that central Arctic nitrate consumption was complete in the early and middle Holocene during peak summer insolation but became incomplete after 5 ka as the summer insolation decreased.
The reconstructed decline in the degree of nitrate consumption could represent reduced nitrate demand in the PML due to less favourable conditions for phytoplankton growth, or to greater nitrate supply to the PML due to weaker stratification. Today, incomplete nitrate consumption in the Eurasian Basin is attributed to light limitation of primary production by sea ice5, although grazing pressure20 or iron limitation38 may also contribute locally. However, none of these demand-based mechanisms appear to explain the past changes in nitrate consumption indicated by δ15NN.p. values. While the sea-ice extent was reduced in the Arctic marginal seas in the early and middle Holocene (Extended Data Fig. 2), central Arctic site B28 was probably perennially ice covered throughout the Holocene39, suggesting that higher early and middle Holocene nitrate consumption occurred without corresponding alleviation of light limitation. Moreover, the abrupt change from incomplete to complete nitrate consumption in the western Arctic at around 11 ka occurs without an apparent change in sea-ice coverage40, arguing against alleviation of light limitation as the cause of the increased nitrate consumption at that time. Thus, the changes in the degree of nitrate consumption are best attributed to changes in nitrate supply, as controlled by the density stratification of the Arctic upper water column23,24.
Density stratification and freshwater input
The changes in nitrate supply indicated by δ15NN.p. values require weaker stratification throughout the Arctic Ocean during the ice age and early deglaciation, pervasively stronger stratification in the early and middle Holocene, and a bifurcation in the late Holocene, with continued strong stratification in the western Arctic but weaker stratification in the central Arctic (Fig. 4). This stratification history can be explained by changes in freshwater input to the Arctic Ocean. In both simple conceptual41 and coupled ice–ocean models42, the strength of Arctic stratification scales with freshwater input, with greater freshwater input shoaling the PML and strengthening the halocline.
The colour indicates the nitrate source (blue is AHW, red is PHW, purple is a mixture); the colour intensity indicates the nitrate concentration, with hatching indicating complete nitrate consumption and deeper shades representing higher nitrate concentrations. White lines and numbers indicate the transport of nitrate and its δ15N; black lines and numbers denote sinking organic matter and its δ15N. Italicized numbers are inferred on the basis of modern observations. For simplicity, only cores B8 and B28 are shown. a, Late Holocene (5–0 ka): PHW and complete nitrate consumption in the western Arctic, and AHW and incomplete nitrate consumption in the central Arctic. b, Holocene Thermal Maximum47,48,49 (~10–5 ka): the same PHW/AHW configuration as a, but with complete nitrate consumption across the Arctic Ocean. c, Closed Bering Strait (before 11 ka): AHW only and incomplete nitrate consumption across the Arctic Ocean. Note that central Arctic changes between 13 and 11 ka are not shown.
During the last glacial period and early deglaciation (Fig. 4c), freshwater input to the Arctic was reduced due to the closure of the Bering Strait, which today accounts for around 30% of total freshwater inflows to the Arctic Ocean10, alongside lower precipitation in the Arctic region under a colder regional climate and ice-damming of major Arctic rivers43. The δ15NN.p. data show that incomplete nitrate consumption characterized the upper Arctic Ocean, consistent with weaker stratification in model simulations with reduced freshwater input41,42. Thus, the halocline was probably weaker and the PML was probably deeper during the last glacial period than today, leading to a greater nitrate supply and light limitation of phytoplankton growth, analogous to (but more extreme than) the Eurasian Basin today. This inference is supported by subsurface temperature reconstructions, which suggest a deeper halocline at the time44.
After 15 ka, our records indicate a sequence of deglacial events that enhanced freshwater inputs to, and stratification of, the Arctic Ocean. Oxygen isotopes in the NGRIP ice core45 (Fig. 3d) show that regional deglacial warming was punctuated by millennial-scale warming in the Bølling–Allerød period (14.7–12.9 ka) and cooling in the Younger Dryas period (12.9–11.7 ka). Although riverine input to the Arctic Ocean increased during the Bølling–Allerød period46, nitrate consumption remained incomplete throughout the Arctic at this time (Fig. 3a), suggesting that freshwater input was not yet sufficient to substantially intensify stratification. However, at 13 ka, meltwater discharge from the Mackenzie River36,37 appears to have strengthened Arctic Ocean stratification, explaining the more complete nitrate consumption at sites B17 and B28 (Fig. 3a and Extended Data Fig. 2). The lack of change at the most southerly site B8 may have been due to north and eastward routing of the meltwater36 (Fig. 1a). After 13 ka, the δ15NN.p. decline at site B28 (and possibly also at B17) suggests a brief return to weaker stratification, which may reflect the short duration (~700 years36) of the meltwater event as well as lower rates of precipitation and riverine input46 during the cooler Younger Dryas period.
At 11 ka, δ15NN.p. of ≥7‰ indicates complete nitrate consumption in both the western and central Arctic Ocean and hence strong stratification. The postglacial flooding of the Bering Strait at around 11 ka would have abruptly increased freshwater input to the western Arctic Ocean, leading to more intense stratification that, in turn, drove complete nitrate consumption. Together, the increase in nitrate consumption and coincident introduction of PHW nitrate to the western Arctic account for the large δ15NN.p. increase at sites B8 and B17 (Fig. 4b,c).
In the central Arctic Ocean, the δ15NN.p. at site B28 also rose to ~7‰ around 11 ka. While this timing is coeval with the western Arctic δ15NN.p. increase and the postglacial flooding of the Bering Strait, the δ15NN.p. at site B28 has many other changes that are unrelated to the flooding of the Bering Strait. Moreover, PHW does not reach the central Arctic Ocean today, and the lack of δ15NN.p. measurements above 7‰ at site B28 suggests that PHW was absent from the central Arctic Ocean throughout our record (Fig. 4). Accordingly, we attribute the central Arctic δ15NN.p. rise at 11 ka to stratification by other local freshwater sources, such as greater riverine input46 from ice sheet discharge and the breakup of ice-dammed Arctic rivers43. It is possible that the Bering Strait flooding played a role in triggering these additional freshwater sources to the central Arctic Ocean, explaining the coincidence of the δ15NN.p. rise at all sites.
During the Holocene, the δ15NN.p. data indicate diverging nitrate consumption trajectories in the western and central Arctic Ocean. In the western Arctic, complete nitrate consumption and thus strong stratification persisted throughout the Holocene (Fig. 4a,b). We propose that this reflects robust stratification in the western Arctic maintained by the continuous input of low-salinity water across the Bering Strait.
In the central Arctic, complete nitrate consumption continued from 10 to 5 ka, indicating strong central Arctic Ocean stratification through the early and middle Holocene (Fig. 4b). This period, referred to as the Holocene Thermal Maximum (HTM), is characterized by a warmer-than-preindustrial summer climate in the Arctic region and sea-ice retreat on the Arctic shelves39,46 due to high summer insolation47,48,49 (Extended Data Fig. 2). A strongly stratified central Arctic during the HTM suggests a sustained period of elevated freshwater input—riverine discharge46 and/or precipitation—associated with the warmer summers (Extended Data Fig. 2). After 5 ka, central Arctic Ocean δ15NN.p. declined in concert with summer insolation (Fig. 3c), indicating that central Arctic Ocean stratification weakened in the late Holocene (Fig. 4a). This probably resulted from a decrease in the same freshwater sources that maintained the earlier stratification.
Implications for the future Arctic Ocean
The rapid increase in nitrate consumption during the reconnection of the North Pacific and Arctic oceans by 11 ka demonstrates that the high degree of nitrate consumption in the modern western Arctic Ocean, occurring despite extensive sea-ice cover, is largely a result of strong stratification. In turn, the development of strong stratification in the western Arctic Ocean by 11 ka indicates that this stratification results from low-salinity inflow across the Bering Strait. In the central Arctic Ocean, greater nitrate consumption during the HTM indicates that stronger stratification characterized past warmer regional Arctic climates, which we suggest arose from greater freshwater input to the central Arctic Ocean in response to the warmer climate.
With regard to the future, climate models find that continued warming will lead to increased freshwater delivery to the Arctic Ocean10 and enhanced stratification, with limited nutrient supply triggering oligotrophy6,7,8. By contrast, satellite-based estimates of Arctic Ocean productivity have increased during the past two decades3,50. An important caveat is that observed productivity increases have largely occurred on the Arctic shelves3,50 where Pacific and North Atlantic inflows and weaker stratification have enhanced the nutrient supply16,50. Our results provide insight into the factors controlling the nutrient supply to the extensive open Arctic Ocean, which is underrepresented in satellite-based estimates due to sea-ice cover3,50.
The HTM provides a useful point of comparison for future Arctic change39,49. A more strongly stratified central Arctic Ocean during the HTM (Fig. 4b) implies that stratification will probably increase as ongoing Arctic warming enhances freshwater input to the central Arctic Ocean10. Such stronger future central Arctic Ocean stratification would be a reversal from the recently observed weakening of stratification13,14 and a return to conditions most recently experienced during the HTM. This enhanced stratification will limit the nutrient supply even as sea-ice retreat alleviates light limitation. Thus, our reconstructions imply that future productivity in the central Arctic Ocean will become more strongly constrained by nutrient supply, as predicted by climate models6,7,8. This dynamic should reduce or prevent any productivity rise that might otherwise occur due to improved light availability with sea-ice loss.
In the western Arctic Ocean, the effect of global warming on water-column stratification may be less consequential. Global-warming-driven strengthening of the stratification would simply ensure that the western Arctic Ocean maintains the stratified, nutrient-limited conditions that have persisted since the flooding of the Bering Strait. In this sense, in the face of continued global warming, the open western basin will probably be the most biogeochemically stable region of the Arctic Ocean.
Methods
Sediment cores and chronology
The 1994 Arctic Ocean Section (or AOS94) collected Mark III box cores (40 × 40 × 60 cm) along a transect from the Mendeleev Ridge to the Lomonosov Ridge53. Each box core was subsampled with 10 cm diameter plastic tubes. Our samples are sourced from the ‘B’ tube at site B8 (78.13° N, 176.74° W, 1,031 m water depth) and site B17 (81.27° N, 178.97° E, 2,217 m water depth) from the Mendeleev Ridge, and at site B28 (88.87° N, 140.18° E, 1,990 m water depth) from the Lomonosov Ridge. Sediment samples were taken every 1 cm. Approximately 1,500 N. pachyderma sinistral (Ehrenberg) tests (~5–7 mg) were picked from the 212–300 µm size fraction using a binocular microscope.
Age models were created from existing 14C dates on N. pachyderma from B8, B17 and B28 (refs. 53,54,55). All existing dates were recalibrated using Marine20 (ref. 56) and no local reservoir correction was applied (that is, ∆R = 0 years). This choice of calibration scheme reflects several considerations:
-
1.
While high-latitude oceans have sluggish air–sea 14C equilibration due to upwelling and/or sea-ice coverage, the upper Arctic Ocean is primarily ventilated along horizontal (advective) pathways and not local vertical mixing. The extremely old marine reservoir ages for the Arctic Ocean (>1,500 years) suggested by coarse-resolution ocean-circulation models57 are not consistent with the prevalence of well-ventilated Atlantic water inflow to the Arctic over the timespan of this study36,44, probably because such simulations do not capture these advective ventilation pathways.
-
2.
Higher prebomb marine reservoir ages (of ~500 years) observed in near-shore western Arctic waters reflect the input of older Pacific-sourced waters transiting across the Bering Strait. Thus, there is an expectation that Arctic marine reservoir ages may have been younger when the Bering Strait was exposed36, in contrast with previous expectations for older marine reservoir ages at this time58.
-
3.
We conservatively choose not to assume a particular ∆R history given the few available constraints of past Arctic marine reservoir ages. Instead, we allow that our calculated ages may be imprecise due to past changes in ∆R and interpret our data on millennial or longer timescales.
Age models (Extended Data Fig. 3) were created using Bayesian age–depth modelling of calibrated calendar ages in ‘Bacon’59 using an accumulation rate prior of 500 yr cm−1, an accumulation shape prior of 1.5, a default section thickness of 5 cm and default autocorrelation priors of memory strength 4 and memory mean 0.7. For B28, all available 14C dates (number of samples, N = 15) from ref. 53 were incorporated into the age–depth model. For B8 and B17, combining 14C datasets from different studies resulted in poor mixing of the Markov Chain Monte Carlo age–depth iterations, as indicated by a Gelman and Rubin reduction factor60 of >1.05. This was addressed by using only 14C dates from ref. 54 in our age–depth models for B8 and B17. Disagreement between 14C ages is most pronounced for dates before 20 ka and may reflect that dates were obtained from different subsampled ‘push cores’ with 1–2 cm misalignments in the sample depths53. Nonetheless, our resulting age models show coherent Arctic Ocean sedimentation-rate patterns, with higher sedimentation rates of 0.5–2 cm per kyr during the Holocene and late Marine Isotope Stage 3, and lower sedimentation rates of 0.2–0.4 cm per kyr during Marine Isotope Stage 2, as observed previously44,53,55,61. Note that the higher sedimentation-rate uncertainty before 15 ka reflects the few available 14C dates and the low sedimentation rates during peak glacial conditions55. This uncertainty does not materially affect our interpretations as we do not interpret millennial-scale patterns before 15 ka.
Seawater nitrate isotope analyses
Seawater samples for nitrate isotope analyses were collected along the western Arctic Ocean GEOTRACES transect (ARC01). Collected samples were filtered through a 0.2 µm polyethersulfone membrane into pre-rinsed 60 ml high-density polyethylene bottles and were stored frozen until analysis. Nitrite was removed by sulfamic acid addition62 before analysis in samples in which nitrite was detected. Twenty nanomoles of nitrate were reduced to N2O gas via the denitrifier method63. Nitrogen and oxygen isotopes were measured on the N2O by continuous-flow isotope ratio mass spectrometry using a Thermo Delta V Advantage spectrometer with a purpose-built, gas chromatography-based device for N2O extraction, concentration and purification64,65 at the University of Connecticut. Samples were analysed in duplicate, yielding an average δ15N standard deviation of 0.2‰.
Foraminifera-bound nitrogen isotope analyses
N. pachyderma samples were gently crushed between glass slides and subjected to clay removal, reduction with buffered sodium dithionite to remove the oxide coatings, and oxidation with buffered potassium persulfate to remove non-bound organic material, following standard protocols66,67. Cleaned foraminiferal carbonate was dissolved in 4 M HCl to release the bound organic nitrogen, which was then oxidized to nitrate using basic potassium persulfate66,67,68. Nitrate was converted to N2O gas via the denitrifier method63, and the δ15N of the N2O was measured using a purpose-built, automated, helium continuous-flow-based extraction and purification system at Princeton University69. Analytical precision based on long-term replication internal carbonate-bound organic N standards is ≤±0.30‰ (1 s.d.). The reported analytical precision is 1 s.d. for duplicate δ15N analyses on sample splits that underwent separate cleaning, oxidation and bacterial conversion to N2O, and averaged ±0.31‰ (1 s.d., N = 18).
Comparison of N. pachyderma δ 15N with bulk-sediment δ 15N
Bulk-sediment δ15N is vulnerable to diagenetic alteration and contamination by allochthonous organic matter (for example, ref. 70). These vulnerabilities encouraged the development of the δ15N of foraminifera-bound organic matter as a palaeoceanographic proxy. The organic matter in foraminifera tests is purely marine in origin (deriving from the host organism) and is protected from diagenetic alteration by the calcite test71. Alteration and contamination of bulk-sediment δ15N are pronounced in the Arctic Ocean72, as further demonstrated by comparing δ15NN.p. with bulk-sediment δ15N. Lacking matching bulk-sediment and foraminifera-bound δ15N data from the same core, we compare the δ15NN.p. values from central Arctic Ocean site B28 with a bulk-sediment δ15N record at site PS2185-4 (87.5° N, 144.5° E), which is also located on the Lomonosov Ridge approximately 150 km to the south of site B28 73. An age model for PS2185-4 was constructed from 15 accelerator mass spectrometry 14C dates on N. pachyderma74 using the above parameters (see ‘Sediment cores and chronology’). Both sites are >800 km from the nearest shelf area and should record comparable open central Arctic Ocean conditions.
Extended Data Fig. 1 shows that, while site δ15NN.p. data from site B28 and PS2185-4 bulk sediment δ15N data have similar values in the late Holocene and both increase between the last glacial period and the late Holocene, the two records otherwise have a very different structure. The differences may be explained by changes in the ratio of terrigenous to marine organic N in the sediment, which biases bulk sediment δ15N but does not affect δ15NN.p.. Terrigenous organic material typically has a low δ15N, as do Mackenzie River sediments (1.4–2.8‰, ref. 75) and riverine dissolved organic nitrogen along the Laptev Sea shelf (2.1‰, ref. 76). Thus, lower values of bulk sediment δ15N compared with δ15NN.p. before 5 ka may reflect a greater proportion of terrigenous nitrogen relative to marine organic nitrogen in the sediment (see ref. 73).
Reconstructing nutrient consumption from δ 15NN.p.
Phytoplankton preferentially assimilate 14N-bearing nitrate, leaving the residual nitrate pool enriched in 15N (ref. 77,78). As heterotrophic zooplankton, foraminifera consume (and source their nitrogen from) phytoplankton. Consequently, the δ15N of phytoplankton biomass consumed by foraminifera decreases with lower degrees of nitrate consumption and increases as consumption increases to completeness. With seasonally complete nitrate consumption, the δ15N of integrated net biomass production must equal the δ15N of the underlying nitrate supply, that is, the δ15N of AHW or PHW nitrate.
The δ15N of biomass produced from a given nitrate source can be simulated with the Rayleigh model, which assumes that nitrate consumption occurs in a closed system with constant isotopic fractionation79,80,81. Over the course of the growing season, the accumulated biomass δ15N is given by
where δ15Nnitrate is the δ15N of the reactant nitrate source, f is the degree of seasonal nitrate consumption given by the concentration ratio between the remaining nitrate and the nitrate supply, and εassimilation is the isotope effect of nitrate assimilation, which is set to 5‰ given available constraints81,82.
The δ15N of organic matter preserved in N. pachyderma (δ15NN.p. as in the main text) is observed to be ~2‰ elevated over δ15Naccumulated in the North Atlantic30, a region of complete nitrate consumption today (where f ~ 0, so equation (1) simplifies to δ15Naccumulated = δ15Nnitrate). The 15N enrichment in non-spinose, symbiont-barren N. pachyderma is consistent with δ15N elevation observed in non-spinose, symbiont-barren foraminifera over thermocline nitrate δ15N in oligotrophic subtropical gyres with complete nitrate consumption83,84. Given that N. pachyderma shows a strong seasonal flux bias to peak summer at high latitudes85, we assume that the diet of N. pachyderma reflects the biomass accumulated throughout the growing season30. This allows for calculating the degree of nitrate consumption (f) from δ15NN.p. using the Rayleigh accumulated product model (equation (1))
where δ15Nnitrate is 5‰ for AHW (Fig. 1c). This calculation is more challenging in the case of mixed AHW and PHW sources, a situation that only occurs after 11 ka in the western Arctic Ocean. For this period, we assume complete nitrate consumption (f ≈ 0) and that δ15NN.p. variations reflect the changing proportions of AHW and PHW at this location, as supported by palaeoceanographic data (see below).
Stability of AHW δ 15Nnitrate since 35 ka
We assume that AHW nitrate has maintained a constant δ15N value of 5‰ over the past 35 ka (for example, Fig. 3a) based on several lines of evidence. Today, the δ15N of AHW nitrate is a function of the contributing water masses to high-latitude North Atlantic subsurface waters plus any isotopic imprint from local N2 fixation or water-column denitrification; the latter two are negligible for AHW given the absence of these processes in cold, well-oxygenated high-latitude North Atlantic waters25,26,86. Instead, the δ15N of AHW nitrate is primarily inherited from water masses that contribute to high-latitude North Atlantic subsurface waters. These include subtropical North Atlantic central waters (NACW), with low δ15Nnitrate from regional N2 fixation87, and Southern Ocean mode water (SAMW) and Antarctic intermediate water, which have higher nitrate δ15N due to partial nitrate consumption in the surface Southern Ocean86. The resultant mixture of these source water masses imparts high-latitude North Atlantic subsurface waters and ultimately AHW with a δ15Nnitrate of ~5‰, which is approximately equal to the mean deep ocean nitrate δ15N (refs. 25,86).
Past changes in AHW nitrate δ15N could result from changing contributions of, or the δ15N of nitrate in, these source water masses. Foraminifera-bound δ15N records from the Atlantic basin inform us as to the nitrate δ15N of these water masses in the past (Extended Data Fig. 4a,b). Foraminifera-bound δ15N from the subtropical North Atlantic is elevated before 11 ka (refs. 71,88), indicating reduced regional N2 fixation and thus a higher δ15Nnitrate of NACW (Extended Data Fig. 4a). In isolation, higher δ15Nnitrate in NACW would tend to increase the AHW nitrate δ15N before 11 ka, the opposite sense of change that would explain our results. With regard to Southern Ocean contributions, nitrate consumption was more complete within the Southern Ocean source regions of mode and intermediate waters before 11 ka (ref. 89) (Extended Data Fig. 4b). However, high degrees of consumption in the Southern Ocean would have meant that any high-δ15N residual nitrate that was input into the SAMW was at a lower nitrate concentration. As a consequence, this nitrate δ15N in Southern Ocean-sourced thermocline waters would have been quickly moderated downwards by mixing with underlying nitrate-rich deep water, with its nitrate δ15N of ~5‰ (ref. 89). Thus, the reconstructed Southern Ocean nutrient consumption changes probably had a limited capacity to change nitrate δ15N in SAMW or downstream in AHW.
Although direct reconstructions of past AHW δ15Nnitrate are not currently possible given the confounding influence of changing nitrate consumption (for example, equation (2)), proximal data suggest relatively constant nitrate δ15N in the high latitude North Atlantic source region of AHW (Extended Data Fig. 4c,d). A δ15NN.p. record from 57° N shows values of ~7‰ during the Holocene that decline to around 4–6‰ during the last glacial period30 (Extended Data Fig. 4c). These declines are interpreted to represent incomplete nitrate consumption in the subpolar North Atlantic Ocean. Two exceptions are δ15NN.p. values of ~7‰ during Heinrich Events 2 and 3, which are argued to reflect intervals of complete surface nitrate consumption due to strong stratification30. Given the 2‰ elevation of δ15NN.p over nitrate δ15N, these results from intervals of putatively complete consumption (the Holocene and Heinrich events) imply a generally constant δ15Nnitrate of around 5‰ in the North Atlantic Ocean upstream of the Arctic Ocean. Additionally, a foraminifera-bound δ15N record from the oligotrophic subtropical North Atlantic Ocean (31° N) suggests a nitrate δ15N of 5–5.5‰ during the last glacial period90. Northward transport of this nitrate is also consistent with an AHW nitrate δ15N of around 5‰.
In summary, the above arguments indicate that high-latitude North Atlantic Ocean and AHW δ15Nnitrate over the past 35,000 years was probably not less than the modern value of 5‰, and, if anything, may have been slightly higher than 5‰ before the Holocene due to reduced subtropical North Atlantic N2 fixation71,88. Any such increase in AHW δ15Nnitrate before the Holocene is in the opposite sense of the dominant change in our δ15NN.p. records, in which δ15NN.p. rises from Marine Isotope Stage 2 to the Holocene (Fig. 3).
Holocene δ 15NN.p. and Pacific water inflow
At western Arctic sites B8 and B17, δ15NN.p. varies between 7.7 and 8.7‰ from 11 ka to the coretop (Fig. 3a). These variations, despite being modest, may indicate changes in the position of the PHW–AHW front associated with varying input of Pacific water to the Arctic. At site B8, which presumably is most sensitive to Pacific water input due to its proximity to the Bering Strait (Fig. 1a), two intervals of elevated δ15NN.p. values occurred between 8–7 ka and 6–5 ka. These times correspond to higher reconstructed phytoplankton productivity from opal and brassicasterol mass-accumulation rates, and to reduced sea-ice extent as demonstrated by reduced values of the phytoplankton marker-IP25 index (PIP25), at a core location proximal to the Bering Strait in the Chukchi Sea91. These biomarker charges are argued to reflect two intervals of enhanced Pacific water inflow to the Arctic Ocean91. We propose that enhanced Pacific water input during these intervals shifted the position of the Pacific–Atlantic halocline front18, leading to an increased presence of PHW at site B8, higher δ15Nnitrate and thus higher δ15NN.p. under conditions of complete nitrate consumption. A late Holocene reduction of Pacific water input is further supported by the coeval δ15NN.p. decline at site B17 after 5 ka (Fig. 3).
Data availability
Source data are provided with this paper. Foraminifera-bound nitrogen isotope data are available from the NCEI Paleoceanography database (https://www.ncdc.noaa.gov/paleo-search/study/33272). Arctic Ocean seawater nitrate isotope data are available from BCO-DMO (https://www.bco-dmo.org/dataset/733109/data).
Change history
29 October 2021
In the version of this article initially published online, the following metadata was omitted and has now been included: “Open access funding provided by Max Planck Institute for Chemistry (2)”.
References
Snow, Water, Ice and Permafrost in the Arctic (SWIPA) 2017 (Arctic Monitoring and Assessment Programme, 2017).
Meier, W. N. et al. Arctic sea ice in transformation: a review of recent observed changes and impacts on biology and human activity. Rev. Geophys. 52, 185–217 (2014).
Arrigo, K. R. & van Dijken, G. L. Continued increases in Arctic Ocean primary production. Prog. Oceanogr. 136, 60–70 (2015).
SIMIP Community, Arctic sea ice in CMIP6. Geophys. Res. Lett. 47, e2019GL086749 (2020).
Tremblay, J.-É. et al. Global and regional drivers of nutrient supply, primary production and CO2 drawdown in the changing Arctic Ocean. Prog. Oceanogr. 139, 171–196 (2015).
Vancoppenolle, M. et al. Future Arctic Ocean primary productivity from CMIP5 simulations: uncertain outcome, but consistent mechanisms. Global Biogeochem. Cycles 27, 605–619 (2013).
Slagstad, D., Wassmann, P. F. J. & Ellingsen, I. Physical constrains and productivity in the future Arctic Ocean. Front. Mar. Sci. 2, 85 (2015).
Fu, W., Keith Moore, J., Primeau, F. W., Lindsay, K. & Randerson, J. T. A growing freshwater lens in the Arctic Ocean with sustained climate warming disrupts marine ecosystem function. J. Geophys. Res. Biogeosci. 125, e2020JG005693 (2020).
Aagaard, K., Coachman, L. & Carmack, E. On the halocline of the Arctic Ocean. Deep Sea Res. A 28, 529–545 (1981).
Haine, T. W. N. et al. Arctic freshwater export: status, mechanisms, and prospects. Global Planet. Change 125, 13–35 (2015).
Carmack, E. C. et al. Freshwater and its role in the Arctic marine system: sources, disposition, storage, export, and physical and biogeochemical consequences in the Arctic and global oceans. J. Geophys. Res. Biogeosci. 121, 675–717 (2016).
Woodgate, R. A. Increases in the Pacific inflow to the Arctic from 1990 to 2015, and insights into seasonal trends and driving mechanisms from year-round Bering Strait mooring data. Prog. Oceanogr. 160, 124–154 (2018).
Polyakov, I. V. et al. Greater role for Atlantic inflows on sea-ice loss in the Eurasian Basin of the Arctic Ocean. Science 356, 285–291 (2017).
Polyakov, I. V., Pnyushkov, A. V. & Carmack, E. C. Stability of the arctic halocline: a new indicator of arctic climate change. Environ. Res. Lett. 13, 125008 (2018).
Metzner, E. P., Salzmann, M. & Gerdes, R. Arctic Ocean surface energy flux and the cold halocline in future climate projections. J. Geophys. Res. Oceans 125, e2019JC015554 (2020).
Polyakov, I. V. et al. Borealization of the Arctic Ocean in response to anomalous advection from sub-Arctic seas. Front. Mar. Sci. 7, 491 (2020).
Codispoti, L. et al. Synthesis of primary production in the Arctic Ocean: III. Nitrate and phosphate based estimates of net community production. Prog. Oceanogr. 110, 126–150 (2013).
McLaughlin, F., Carmack, E., Macdonald, R., Weaver, A. J. & Smith, J. The Canada Basin, 1989–1995: upstream events and far-field effects of the Barents Sea. J. Geophys. Res. 107, 3082 (2002).
Swift, J. et al. Waters of the Makarov and Canada basins. Deep Sea Res. 2 44, 1503–1529 (1997).
Bluhm, B., Kosobokova, K. & Carmack, E. A tale of two basins: an integrated physical and biological perspective of the deep Arctic Ocean. Prog. Oceanogr. 139, 89–121 (2015).
Woodgate, R. A., Aagaard, K. & Weingartner, T. J. A year in the physical oceanography of the Chukchi Sea: moored measurements from autumn 1990–1991. Deep-Sea Res. Pt. II 52, 3116–3149 (2005).
Morison, J. et al. Changing Arctic Ocean freshwater pathways. Nature 481, 66–70 (2012).
Randelhoff, A. et al. Pan-Arctic Ocean primary production constrained by turbulent nitrate fluxes. Front. Mar. Sci. 7, 150 (2020).
Peralta-Ferriz, C. & Woodgate, R. A. Seasonal and interannual variability of pan-Arctic surface mixed layer properties from 1979 to 2012 from hydrographic data, and the dominance of stratification for multiyear mixed layer depth shoaling. Prog. Oceanogr. 134, 19–53 (2015).
Van Oostende, N. et al. Variation of summer phytoplankton community composition and its relationship to nitrate and regenerated nitrogen assimilation across the North Atlantic Ocean. Deep Sea Res. 1 121, 79–94 (2017).
Fripiat, F. et al. Influence of the bordering shelves on nutrient distribution in the Arctic halocline inferred from water column nitrate isotopes. Limnol. Oceanogr. 63, 2154–2170 (2018).
Granger, J. et al. Coupled nitrification-denitrification in sediment of the eastern Bering Sea shelf leads to 15N enrichment of fixed N in shelf waters. J. Geophys. Res. Oceans 116, C11006 (2011).
Granger, J., Sigman, D. M., Gagnon, J. G., Tremblay, J.-E. & Mucci, A. On the properties of the Arctic halocline and deep water masses of the Canada Basin from nitrate isotope ratios. J. Geophys. Res. Oceans 123, 5443–5458 (2018).
Brown, Z. W., Casciotti, K. L., Pickart, R. S., Swift, J. H. & Arrigo, K. R. Aspects of the marine nitrogen cycle of the Chukchi Sea shelf and Canada Basin. Deep Sea Res. 2 118, 73–87 (2015).
Straub, M. et al. Nutrient conditions in the subpolar North Atlantic during the last glacial period reconstructed from foraminifera-bound nitrogen isotopes. Paleoceanography 28, 79–90 (2013).
Lambeck, K., Rouby, H., Purcell, A., Sun, Y. & Sambridge, M. Sea level and global ice volumes from the Last Glacial Maximum to the Holocene. Proc. Natl Acad. Sci. USA 111, 15296–15303 (2014).
Jakobsson, M. et al. Arctic Ocean glacial history. Quat. Sci. Rev. 92, 40–67 (2014).
Pico, T., Mitrovica, J. X. & Mix, A. C. Sea level fingerprinting of the Bering Strait flooding history detects the source of the Younger Dryas climate event. Sci. Adv. 6, eaay2935 (2020).
Jakobsson, M. et al. Post-glacial flooding of the Bering Land Bridge dated to 11 cal ka BP based on new geophysical and sediment records. Clim. Past 13, 991–1005 (2017).
Bauch, H. A. et al. Chronology of the Holocene transgression at the North Siberian margin. Global Planet. Change 31, 125–139 (2001).
Keigwin, L. D. et al. Deglacial floods in the Beaufort Sea preceded Younger Dryas cooling. Nat. Geosci. 11, 599–604 (2018).
Tarasov, L. & Peltier, W. R. Arctic freshwater forcing of the Younger Dryas cold reversal. Nature 435, 662–665 (2005).
Rijkenberg, M. J. A., Slagter, H. A., van der Loeff, M. R., van Ooijen, J. & Gerringa, L. J. A. Dissolved Fe in the deep and upper Arctic Ocean with a focus on Fe limitation in the Nansen Basin. Front. Mar. Sci. 5, 88 (2018).
de Vernal, A. et al. Natural variability of the Arctic Ocean sea ice during the present interglacial. Proc. Natl Acad. Sci. USA 117, 26069–26075 (2020).
de Vernal, A., Hillaire-Marcel, C. & Darby, D. A. Variability of sea ice cover in the Chukchi Sea (western Arctic Ocean) during the Holocene. Paleoceanography 20, PA4018 (2005).
Nilsson, J. & Walin, G. Salinity-dominated thermohaline circulation in sill basins: can two stable equilibria exist? Tellus A 62, 123–133 (2010).
Pemberton, P. & Nilsson, J. The response of the central Arctic Ocean stratification to freshwater perturbations. J. Geophys. Res. Oceans 121, 792–817 (2016).
Polyak, L., Niessen, F., Gataullin, V. & Gainanov, V. The eastern extent of the Barents–Kara ice sheet during the Last Glacial Maximum based on seismic-reflection data from the eastern Kara Sea. Polar Res. 27, 162–172 (2008).
Cronin, T. M. et al. Deep Arctic Ocean warming during the last glacial cycle. Nat. Geosci. 5, 631–634 (2012).
North Greenland Ice Core Project members, High-resolution record of Northern Hemisphere climate extending into the last interglacial period. Nature 431, 147–151 (2004).
Hörner, T., Stein, R., Fahl, K. & Birgel, D. Post-glacial variability of sea ice cover, river run-off and biological production in the western Laptev Sea (Arctic Ocean)—a high-resolution biomarker study. Quat. Sci. Rev. 143, 133–149 (2016).
Kaufman, D. S. et al. Holocene thermal maximum in the western Arctic (0–180° W). Quat. Sci. Rev. 23, 529–560 (2004).
Park, H.-S., Kim, S.-J., Stewart, A. L., Son, S.-W. & Seo, K.-Y. Mid-Holocene Northern Hemisphere warming driven by Arctic amplification. Sci. Adv. 5, eaax8203 (2019).
Axford, Y., de Vernal, A. & Osterberg, E. C. Past warmth and its impacts during the holocene thermal maximum in greenland. Annu. Rev. Earth. Planet. Sci. 49, 279–307 (2021).
Lewis, K. M., van Dijken, G. L. & Arrigo, K. R. Changes in phytoplankton concentration now drive increased Arctic Ocean primary production. Science 369, 198–202 (2020).
Schlitzer, R. Ocean Data View (ODV Forum, 2021); http://odv.awi.de
Laskar, J. et al. A long-term numerical solution for the insolation quantities of the Earth. Astron. Astrophys. 428, 261–285 (2004).
Darby, D. A., Bischof, J. F. & Jones, G. A. Radiocarbon chronology of depositional regimes in the western Arctic Ocean. Deep Sea Res. 2 44, 1745–1757 (1997).
Poore, R. Z., Ostermann, D. R. & McGeehin, J. Stable Isotope Data and AMS 14C Dates from Arctic Ocean Section 1994 Surface Sediment Transect and Box Core Samples from the Mendeleyev Ridge Area (US Geological Survey, 1999).
Poirier, R. K., Cronin, T. M., Briggs, W. M. Jr. & Lockwood, R. Central Arctic paleoceanography for the last 50 kyr based on ostracode faunal assemblages. Mar. Micropaleontol. 88–89, 65–76 (2012).
Heaton, T. J. et al. Marine20—the marine radiocarbon age calibration curve (0–55,000 cal BP). Radiocarbon 62, 779–820 (2020).
Butzin, M., Köhler, P. & Lohmann, G. Marine radiocarbon reservoir age simulations for the past 50,000 years. Geophys. Res. Lett. 44, 8473–8480 (2017).
Hanslik, D. et al. Quaternary Arctic Ocean sea ice variations and radiocarbon reservoir age corrections. Quat. Sci. Rev. 29, 3430–3441 (2010).
Blaauw, M. & Christen, J. A. Flexible paleoclimate age-depth models using an autoregressive gamma process. Bayesian Anal. 6, 457–474 (2011).
Brooks, S. P. & Gelman, A. General methods for monitoring convergence of iterative simulations. J. Comput. Graph. Stat. 7, 434–455 (1998).
Polyak, L. et al. Late Quaternary stratigraphy and sedimentation patterns in the western Arctic Ocean. Global Planet. Change 68, 5–17 (2009).
Granger, J. & Sigman, D. M. Removal of nitrate with sulfamic acid for nitrate N and O isotope analysis with the denitrifier method. Rapid Commun. Mass Spectrom. 23, 3753–3762 (2009).
Sigman, D. M. et al. A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater. Anal. Chem. 73, 4145–4153 (2001).
Casciotti, K. L., Sigman, D. M., Hastings, M. G., Böhlke, J. K. & Hilkert, A. Measurement of the oxygen isotopic composition of nitrate in seawater and freshwater using the denitrifier method. Anal. Chem. 74, 4905–4912 (2002).
McIlvin, M. R. & Casciotti, K. L. Technical updates to the bacterial method for nitrate isotope analyses. Anal. Chem. 83, 1850–1856 (2011).
Ren, H. et al. Impact of glacial/interglacial sea level change on the ocean nitrogen cycle. Proc. Natl Acad. Sci. USA 114, E6759–E6766 (2017).
Kast, E. R. et al. Nitrogen isotope evidence for expanded ocean suboxia in the early Cenozoic. Science 364, 386–389 (2019).
Knapp, A. N., Sigman, D. M. & Lipschultz, F. N isotopic composition of dissolved organic nitrogen and nitrate at the Bermuda Atlantic time-series study site. Global Biogeochem. Cycles 19, GB1018 (2005).
Weigand, M. A., Foriel, J., Barnett, B., Oleynik, S. & Sigman, D. M. Updates to instrumentation and protocols for isotopic analysis of nitrate by the denitrifier method. Rapid Commun. Mass Spectrom. 30, 1365–1383 (2016).
Robinson, R. S. et al. A review of nitrogen isotopic alteration in marine sediments. Paleoceanography 27, PA4203 (2012).
Ren, H. et al. Foraminiferal isotope evidence of reduced nitrogen fixation in the ice age Atlantic. Ocean Sci. 323, 244–248 (2009).
Schubert, C. J. & Calvert, S. E. Nitrogen and carbon isotopic composition of marine and terrestrial organic matter in Arctic Ocean sediments: implications for nutrient utilization and organic matter composition. Deep Sea Res. 1 48, 789–810 (2001).
Schubert, C. J., Stein, R. & Calvert, S. E. Tracking nutrient and productivity variations over the last deglaciation in the Arctic Ocean. Paleoceanography 16, 199–211 (2001).
Nørgaard-Pedersen, N., Spielhagen, R. F., Thiede, J. & Kassens, H. Central Arctic surface ocean environment during the past 80,000 years. Paleoceanography 13, 193–204 (1998).
Naidu, A. S. et al. Organic carbon isotope ratios of Arctic Amerasian continental shelf sediments. Int. J. Earth Sci. 89, 522–532 (2000).
Thibodeau, B., Bauch, D. & Voss, M. Nitrogen dynamic in Eurasian coastal Arctic ecosystem: insight from nitrogen isotope. Global Biogeochem. Cycles 31, 836–849 (2017).
Wada, E. & Hittori, A. Nitrogen isotope effects in the assimilation of inorganic nitrogenous compounds by marine diatoms. Geomicrobiol. J. 1, 85–101 (1978).
Sigman, D. M., Altabet, M. A., McCorkle, D. C., Francois, R. & Fischer, G. The δ15N of nitrate in the Southern Ocean: nitrate consumption in surface waters. Global Biogeochem. Cycles 13, 1149–1166 (1999).
Mariotti, A. et al. Experimental determination of nitrogen kinetic isotope fractionation: some principles; illustration for the denitrification and nitrification processes. Plant Soil 62, 413–430 (1981).
Ren, H. et al. Glacial-to-interglacial changes in nitrate supply and consumption in the subarctic North Pacific from microfossil-bound N isotopes at two trophic levels. Paleoceanography 30, 1217–1232 (2015).
Sigman, D. M. & Fripiat, F. in Encyclopedia of Ocean Sciences 3rd edn, Vol. 1 (eds Cochran, J. K. et al.) 263–278 (Elsevier, 2019).
Fripiat, F. et al. The isotope effect of nitrate assimilation in the Antarctic Zone: improved estimates and paleoceanographic implications. Geochim. Cosmochim. Acta 247, 261–279 (2019).
Ren, H., Sigman, D. M., Thunell, R. C. & Prokopenko, M. G. Nitrogen isotopic composition of planktonic foraminifera from the modern ocean and recent sediments. Limnol. Oceanogr. 57, 1011–1024 (2012).
Smart, S. et al. Ground-truthing the planktic foraminifer-bound nitrogen isotope paleo-proxy in the Sargasso Sea. Geochim. Cosmochim. Acta 235, 463–482 (2018).
Kohfeld, K. E., Fairbanks, R. G., Smith, S. L. & Walsh, I. D. Neogloboquadrina pachyderma (sinistral coiling) as paleoceanographic tracers in polar oceans: evidence from northeast water polynya plankton tows, sediment traps, and surface sediments. Paleoceanography 11, 679–699 (1996).
Marconi, D. et al. Nitrate isotope distributions on the US GEOTRACES North Atlantic cross-basin section: signals of polar nitrate sources and low latitude nitrogen cycling. Mar. Chem. 177, 143–156 (2015).
Knapp, A. N., DiFiore, P. J., Deutsch, C., Sigman, D. M. & Lipschultz, F. Nitrate isotopic composition between Bermuda and Puerto Rico: implications for N2 fixation in the Atlantic Ocean. Global Biogeochem. Cycles 22, GB3014 (2008).
Straub, M. et al. Changes in North Atlantic nitrogen fixation controlled by ocean circulation. Nature 501, 200–203 (2013).
Martínez-García, A. et al. Iron fertilization of the subantarctic ocean during the Last Ice Age. Science 343, 1347 (2014).
Auderset, A. Nutrient Cycling in the Oligotrophic Ocean Over the Past 65 Million Years Ch. 3 PhD thesis, ETH Zurich (2020).
Stein, R. et al. Holocene variability in sea ice cover, primary production, and Pacific-Water inflow and climate change in the Chukchi and East Siberian Seas (Arctic Ocean). J. Quat. Sci. 32, 362–379 (2017).
Müller, J. et al. Holocene cooling culminates in sea ice oscillations in Fram Strait. Quat. Sci. Rev. 47, 1–14 (2012).
Cronin, T. M. et al. Quaternary sea-ice history in the Arctic Ocean based on a new ostracode sea-ice proxy. Quat. Sci. Rev. 29, 3415–3429 (2010).
Acknowledgements
We thank S. Oleynik, F. Rubach and L. Gemery for laboratory assistance, and L. Keigwin, A. de Vernal and T. Pico for sharing data. J.R.F., F.F., A.M.-G. and G.H.H. were funded by the Max Planck Society. J.R.F., D.M.S. and O.M.U. also received support from US National Science Foundation grants OPP-0612198 and OCE-1536368, the Tuttle Fund of the Department of Geosciences of Princeton University, the Grand Challenges Program of the Princeton Environmental Institute and from ExxonMobil through the Andlinger Center for Energy and the Environment of Princeton University. J.G. was funded by US National Science Foundation grant OCE-1535002. T.M.C. was funded by the USGS Land Change Program. Any use of trade, firm or product names is for descriptive purposes only and does not imply endorsement by the US Government.
Funding
Open access funding provided by Max Planck Institute for Chemistry (2).
Author information
Authors and Affiliations
Contributions
J.R.F., D.M.S. and J.G. designed the study; J.R.F. and O.M.U. performed the δ15NN.p. analyses; J.G. performed the seawater nitrate δ15N analyses; T.M.C. provided the sediment samples; J.R.F., O.M.U. and T.M.C. provided chronology; J.R.F. and D.M.S. drafted the first version of the manuscript; all authors contributed to the interpretation of the data and preparation of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Geoscience thanks Brian Haley, Sofia Ribeiro and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: James Super.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Comparison of δ15NN.p. and N content from Site B28 (blue circles, this study) and bulk sediment δ15N and %TOC from Site PS2185-4 (brown triangles, ref. 73).
a, Prior to 15 ka, bulk sediment δ15N averages 4.1 ‰, which is 1.5 to 2 ‰ lower than δ15NN.p.. Between 15 and 10 ka, bulk sediment δ15N increases by 1 ‰ while δ15NN.p. increases by 2 ‰; the high δ15NN.p. at 13 ka is absent in bulk sediment δ15N. Between 10 and 5 ka, bulk sediment δ15N continues to increase, including a rapid 1.5 ‰ increase between 6 and 5 ka; in contrast, δ15NN.p. is constant through this time. After 5 ka, δ15NN.p. declines by ~1 ‰ while bulk sediment δ15N is constant. b, N. pachyderma N content is constant whereas PS2185-4 percent total organic carbon (%TOC) declines downcore. The decline in bulk sediment %TOC might reflect progressive diagenetic organic matter loss with depth, with terrestrial organic carbon being more recalcitrant.
Extended Data Fig. 2 Comparison to meltwater discharge and sea ice proxies.
a, Arctic δ15NN.p. (colors as in Fig. 3), (b) N. pachyderma δ18O from the Beaufort Slope indicating freshwater flooding events in the Arctic Ocean36; greater freshwater discharge is upward. (c)–(f) Relative sea ice extent from c-e, PIP25 biomarker index in the (c) Chukchi Sea91, (d) Fram Strait92, (e) Laptev Sea45, and (f) open western Arctic Ocean sea ice coverage (months/year >50%) from dinocyst assemblages40 (locations in Fig. 1a). More extensive sea ice is downward. Vertical brown bar denotes the Younger Dryas; vertical blue bar indicates proposed timing of postglacial Bering Strait flooding33,34.
Extended Data Fig. 3 Age models.
Age-depth models for sediment cores B8 (a), B17 (b) and B28 (c). Black line in each panel is Bayesian age-depth model from Bacon59; gray shading indicates 95% confidence interval. Symbols indicate calibrated 14C dates on N. pachyderma. Data sources: a) dark diamonds54, light diamonds55; b) diamonds54, triangles53, circles93; c) diamonds53.
Extended Data Fig. 4 Foraminifera-bound δ15N constraints on AHW nitrate δ15N over the last 35,000 years.
a, δ15N from the tropical Atlantic, with higher values indicating weaker N2 fixation during the last ice age71,88. b, δ15N from the Subantarctic Southern Ocean, with higher values indicating more complete surface ocean nitrate consumption during the last ice age89. c, δ15NN.p. from subpolar North Atlantic, interpreted in terms of changing nitrate consumption (dark gray bars at bottom); the data imply relatively constant δ15Nnitrate if the Heinrich Stadial δ15NN.p. peaks reached complete consumption30 (light gray bars for Heinrich Stadials 2 and 3, compared with light gray bar for the Holocene).
Extended Data Fig. 5 Comparison to Holocene western Arctic productivity and sea ice proxies.
a, Site B8 δ15NN.p. (orange circles), (b) Chukchi Sea phytoplankton productivity from brassicasterol (green) and opal (blue) mass accumulation rates and (c) Chukchi Sea sea ice extent from PIP25 (blue) (ref. 91). Higher brassicasterol and opal MARs indicate higher phytoplankton productivity; lower PIP25 (plotted upward) indicates reduced sea ice extent. Gray shading denotes two intervals of higher Chukchi Sea productivity and reduced sea ice that have been attributed to increased Pacific water input to the Arctic Ocean91.
Supplementary information
Supplementary Data 1
Seawater nitrate and foraminifera-bound δ15N data generated in this study.
Supplementary Table 1
Mean δ15NN.p. values for time intervals and statistical comparisons using two-tailed, heteroscedastic t-tests. Significant differences at α = 0.05 are bolded.
Source data
Source Data Fig. 1
Datasets used and references in Fig. 1.
Source Data Fig. 2
Datasets used and references in Fig. 2.
Source Data Fig. 3
Datasets used and references in Fig. 3.
Source Data Extended Data Fig. 1
Datasets used and references in Extended Data Fig. 1.
Source Data Extended Data Fig. 2
Datasets used and references in Extended Data Fig. 2.
Source Data Extended Data Fig. 3
Datasets used and references in Extended Data Fig. 3.
Source Data Extended Data Fig. 4
Datasets used and references in Extended Data Fig. 4.
Source Data Extended Data Fig. 5
Datasets used and references in Extended Data Fig. 5.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Farmer, J.R., Sigman, D.M., Granger, J. et al. Arctic Ocean stratification set by sea level and freshwater inputs since the last ice age. Nat. Geosci. 14, 684–689 (2021). https://doi.org/10.1038/s41561-021-00789-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-021-00789-y
This article is cited by
-
Omnipresent authigenic calcite distorts Arctic radiocarbon chronology
Communications Earth & Environment (2023)
-
Arctic deep-water anoxia and its potential role for ocean carbon sink during glacial periods
Communications Earth & Environment (2023)
-
Stochastic Processes Dominate in the Water Mass–Based Segregation of Diazotrophs in a High Arctic Fjord (Svalbard)
Microbial Ecology (2023)