Abstract
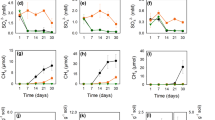
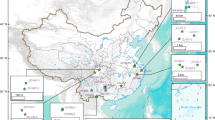
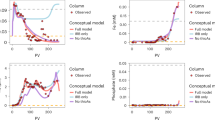
The accumulation of carcinogenic arsenic in rice, the world’s main staple crop, represents a health threat to millions of people. The speciation of arsenic controls its mobility and bioavailability and therefore its entry into the food chain. Inorganic and methylated oxyarsenic species have been a focus of research, but arsenic characterization in the field has largely ignored thioarsenates, in which sulfur takes the place of oxygen. Here, on the basis of field, mesocosm and soil incubation studies across multiple paddy soils from rice cultivation areas in Italy, France and China, we find that thioarsenates are important arsenic species in paddy-soil pore waters. We observed thioarsenates throughout the cropping season, with concentrations comparable to the much-better-investigated methylated oxyarsenates. Anaerobic soil incubations confirmed a large potential for thiolation across a wide diversity of paddy soil types in different climate zones and with different parent materials. In these incubations, inorganic thioarsenates occurred predominantly where soil pH exceeded 6.5 and in the presence of zero-valent sulfur. Methylated thioarsenates occurred predominantly at soil pH below 7 and in the presence of their precursors, methylated oxyarsenates. High concentrations of dissolved iron limited arsenic thiolation. Sulfate fertilization increased thioarsenate formation. It is currently unclear whether thiolation is good or bad for rice consumption safety. Nevertheless, we highlight thiolation as an important factor to arsenic biogeochemistry in rice paddies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are also available from the corresponding author on reasonable request.
References
Stone, R. Arsenic and paddy rice: a neglected cancer risk? Science 321, 184–185 (2008).
Ma, J. F. et al. Transporters of arsenite in rice and their role in arsenic accumulation in rice grain. Proc. Natl Acad. Sci. 105, 9931–9935 (2008).
Wang, P., Zhang, W., Mao, C., Xu, G. & Zhao, F.-J. The role of OsPT8 in arsenate uptake and varietal difference in arsenate tolerance in rice. J. Exp. Bot. 67, 6051–6059 (2016).
Ye, Y. et al. OsPT4 contributes to arsenate uptake and transport in rice. Front. Plant Sci. 8, 2197 (2017).
Xu, X. Y., McGrath, S. P., Meharg, A. A. & Zhao, F. J. Growing rice aerobically markedly decreases arsenic accumulation. Environ. Sci. Technol. 42, 5574–5579 (2008).
Jia, Y. et al. Microbial arsenic methylation in soil and rice rhizosphere. Environ. Sci. Technol. 47, 3141–3148 (2013).
Lomax, C. et al. Methylated arsenic species in plants originate from soil microorganisms. N. Phytol. 193, 665–672 (2012).
Zhao, F.-J., Zhu, Y.-G. & Meharg, A. A. Methylated arsenic species in rice: geographical variation, origin, and uptake mechanisms. Environ. Sci. Technol. 47, 3957–3966 (2013).
Meharg, A. A. & Zhao, F.-J. (eds) in Arsenic & Rice 71–101 (Springer, 2012).
Besold, J. et al. Monothioarsenate transformation kinetics determining arsenic sequestration by sulfhydryl groups of peat. Environ. Sci. Technol. 52, 7317–7326 (2018).
Wallschläger, D. & London, J. Determination of methylated arsenic–sulfur compounds in groundwater. Environ. Sci. Technol. 42, 228–234 (2007).
Conklin, S. D., Fricke, M. W., Creed, P. A. & Creed, J. T. Investigation of the pH effects on the formation of methylated thio-arsenicals, and the effects of pH and temperature on their stability. J. Anal. At. Spectrom. 23, 711–716 (2008).
Planer-Friedrich, B., London, J., McCleskey, R. B., Nordstrom, D. K. & Wallschläger, D. Thioarsenates in geothermal waters of Yellowstone National Park: determination, preservation, and geochemical importance. Environ. Sci. Technol. 41, 5245–5251 (2007).
Planer-Friedrich, B., Schaller, J., Wismeth, F., Mehlhorn, J. & Hug, S. J. Monothioarsenate occurrence in Bangladesh groundwater and its removal by ferrous and zero-valent iron technologies. Environ. Sci. Technol. 52, 5931–5939 (2018).
Planer-Friedrich, B. & Wallschläger, D. A critical investigation of hydride generation-based arsenic speciation in sulfidic waters. Environ. Sci. Technol. 43, 5007–5013 (2009).
Smieja, J. A. & Wilkin, R. T. Preservation of sulfidic waters containing dissolved As(III). J. Environ. Monit. 5, 913–916 (2003).
Kögel-Knabner, I. et al. Biogeochemistry of paddy soils. Geoderma 157, 1–14 (2010).
Wind, T. & Conrad, R. Localization of sulfate reduction in planted and unplanted rice field soil. Biogeochemistry 37, 253–278 (1997).
Ayotade, K. A. Kinetics and reactions of hydrogen sulphide in solution of flooded rice soils. Plant Soil 46, 381–389 (1977).
Saalfield, S. L. & Bostick, B. C. Changes in iron, sulfur, and arsenic speciation associated with bacterial sulfate reduction in ferrihydrite-rich systems. Environ. Sci. Technol. 43, 8787–8793 (2009).
Burton, E. D., Johnston, S. G. & Kocar, B. D. Arsenic mobility during flooding of contaminated soil: the effect of microbial sulfate reduction. Environ. Sci. Technol. 48, 13660–13667 (2014).
Xu, L. Y. et al. Speciation change and redistribution of arsenic in soil under anaerobic microbial activities. J. Hazard. Mater. 301, 538–546 (2016).
Crusciol, C. A. C., Nascente, A. S., Soratto, R. P. & Rosolem, C. A. Upland rice growth and mineral nutrition as affected by cultivars and sulfur availability. Soil Sci. Soc. Am. J. 77, 328–335 (2013).
Schütz, H., Holzapfel-Pschorn, A., Conrad, R., Rennenberg, H. & Seiler, W. A 3-year continuous record on the influence of daytime, season, and fertilizer treatment on methane emission rates from an Italian rice paddy. J. Geophys. Res. Atmos. 94, 16405–16416 (1989).
Minamikawa, K., Sakai, N. & Hayashi, H. The effects of ammonium sulfate application on methane emission and soil carbon content of a paddy field in Japan. Agric. Ecosyst. Environ. 107, 371–379 (2005).
Fan, J., Xia, X., Hu, Z., Ziadi, N. & Liu, C. Excessive sulfur supply reduces arsenic accumulation in brown rice. Plant Soil Environ. 59, 169–174 (2013).
Zhang, J. et al. Influence of sulfur on transcription of genes involved in arsenic accumulation in rice grains. Plant Mol. Biol. Report. 34, 556–565 (2016).
Jia, Y. & Bao, P. Arsenic bioavailability to rice plant in paddy soil: influence of microbial sulfate reduction. J. Soil. Sediment. 15, 1960–1967 (2015).
Zeng, X. et al. Effects of sulfate application on inhibiting accumulation and alleviating toxicity of arsenic in panax notoginseng grown in arsenic-polluted soil. Water Air Soil Poll. 227, 148 (2016).
Baker, M., Inniss, W., Mayfield, C., Wong, P. & Chau, Y. Effect of pH on the methylation of mercury and arsenic by sediment microorganisms. Environ. Technol. Lett. 4, 89–100 (1983).
Cullen, W. R. et al. Methylated and thiolated arsenic species for environmental and health research—a review on synthesis and characterization. J. Environ. Sci. 49, 7–27 (2016).
Kim, Y.-T., Lee, H., Yoon, H.-O. & Woo, N. C. Kinetics of dimethylated thioarsenicals and the formation of highly toxic dimethylmonothioarsinic acid in environment. Environ. Sci. Technol. 50, 11637–11645 (2016).
Kerl, C. F., Rafferty, C., Clemens, S. & Planer-Friedrich, B. Monothioarsenate uptake, transformation, and translocation in rice plants. Environ. Sci. Technol. 52, 9154–9161 (2018).
Kerl, C. F. et al. Methylated thioarsenates and monothioarsenate differ in uptake, transformation, and contribution to total arsenic translocation in rice plants. Environ. Sci. Technol. 53, 5787–5796 (2019).
Ackerman, A. H. et al. Comparison of a chemical and enzymatic extraction of arsenic from rice and an assessment of the arsenic absorption from contaminated water by cooked rice. Environ. Sci. Technol. 39, 5241–5246 (2005).
Ayotade, K. A. Kinetics and reactions of hydrogen-sulfide in solution of flooded rice soils. Plant Soil 46, 381–389 (1977).
Tang, L., Yang, J. & Shen, X. Effects of additional iron-chelators on Fe 2+-initiated lipid peroxidation: evidence to support the Fe 2+… Fe 3+ complex as the initiator. J. Inorg. Biochem. 68, 265–272 (1997).
Colman, B. P. Understanding and eliminating iron interference in colorimetric nitrate and nitrite analysis. Environ. Monit. Assess. 165, 633–641 (2010).
Suess, E., Wallschläger, D. & Planer-Friedrich, B. Stabilization of thioarsenates in iron-rich waters. Chemosphere 83, 1524–1531 (2011).
Zang, V. & Van Eldik, R. Kinetics and mechanism of the autoxidation of iron (II) induced through chelation by ethylenediaminetetraacetate and related ligands. Inorg. Chem. 29, 1705–1711 (1990).
Suess, E. et al. Discrimination of thioarsenites and thioarsenates by X-ray absorption spectroscopy. Anal. Chem. 81, 8318–8326 (2009).
Ministry of Ecology and Environment of the People’s Republic of China Soil Environment Quality Risk Control Standard for Soil Contamination of Agricultural Land GB 15618-2018 (Ministry of Ecology and Environment, 2018).
Zhao, F.-J., Ma, Y., Zhu, Y.-G., Tang, Z. & McGrath, S. P. Soil contamination in China: current status and mitigation strategies. Environ. Sci. Technol. 49, 750–759 (2014).
Ratering, S. & Schnell, S. Localization of iron-reducing activity in paddy soil by profile studies. Biogeochemistry 48, 341–365 (2000).
Stroud, J. L. et al. Assessing the labile arsenic pool in contaminated paddy soils by isotopic dilution techniques and simple extractions. Environ. Sci. Technol. 45, 4262–4269 (2011).
Zhang, S.-Y. et al. Diversity and abundance of arsenic biotransformation genes in paddy soils from southern China. Environ. Sci. Technol. 49, 4138–4146 (2015).
Zhao, F.-J. et al. Arsenic methylation in soils and its relationship with microbial arsM abundance and diversity, and As speciation in rice. Environ. Sci. Technol. 47, 7147–7154 (2013).
Stookey, L. L. Ferrozine—a new spectrophotometric reagent for iron. Anal. Chem. 42, 779–781 (1970).
Lohmayer, R., Kappler, A., Lösekann-Behrens, T. & Planer-Friedrich, B. Sulfur species as redox partners and electron shuttles for ferrihydrite reduction by Sulfurospirillum deleyianum. Appl. Environ. Microb. 80, 3141–3149 (2014).
Grömping, U. Relative importance for linear regression in R: the package relaimpo. J. Stat. Softw. 17, 1–27 (2006).
De’Ath, G. Multivariate regression trees: a new technique for modeling species–environment relationships. Ecology 83, 1105–1117 (2002).
Acknowledgements
We acknowledge financial support for a PhD stipend to J. Wang from the China Scholarship Council (CSC), mobility grants within the Bavarian Funding Programme for travels to France (BFHZ FK-40-15), Italy (BayIntAn2016), China (BayCHINA2018), as well as from the National Key Research and Development Project of China (2016YFE0106400) and the National Natural Science Foundation of China (41325003) for paddy soils survey, sampling and determination in China. We acknowledge data support from the Soil Data Center, National Earth System Science Data Sharing Service Infrastructure, National Science & Technology Infrastructure of China (http://soil.geodata.cn) and Ente Regionale per i Servizi all’Agricoltura e alle Foreste—Regione Lombardia of Italy (https://www.ersaf.lombardia.it/). We are indebted to the staff at the Rice Research Centre in Castello d’Agogna (Pavia, Italy) for help with soil sampling and mesocosm rice cultivation, including S. Feccia, F. Mazza, U. Rolla, E. Miniotti, G. Beltarre, A. Iuzzolino, F. Bonassi, L. Pizzin, D. Tenni, A. Zanellato, F. Massara, E. Magnani, G. Bertone and M. Zini. We thank C. Thomas and A. Boisnard from the Centre Français du Riz (Arles, Italy) for assistance with soil sampling in France. We are grateful to M. T. F. Wagner, C. Lerda, S. Will, J. M. Leon, J. Besold, L. Wegner and S. Zhang for help with field sampling and laboratory assistance, to Y. Yang for help with map design, and to J. Mehlhorn for help with statistical analyses.
Author information
Authors and Affiliations
Contributions
J.W. initiated DTPA method development, conceived and performed all mesocosm and incubation experiments including analyses, evaluated the results and contributed to manuscript preparation; C.F.K. contributed to field survey, sample analyses, data evaluation and manuscript preparation; L.B. contributed to DTPA method development; P.H. and L.W. initiated the Chinese soil survey and advised on incubation experiments; P.H., T.M., G.W. and L.W. sampled and characterized the Chinese soils; M.R. assisted in design, setup and operation of mesocosms and sample collection; M.M. and D.S.-P. assisted in analyses of aqueous parameters from mesocosms; M.M. contributed to field survey and data discussion; B.P.-F. initiated and supervised the project, carried out the field survey, conceived experiments and wrote the manuscript; all authors contributed to revising the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Primary Handling Editors: Xujia Jiang; Rebecca Neely.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, Figs. 1–20 and Tables 1–9
Supplementary Table 1
Pore water chemistry including As speciation
Supplementary Table 2
Soil classification and basic chemical parameters
Supplementary Table 3
Pore water chemistry in the mesocosm experiments
Supplementary Table 4
Mean values for thiolation and methylation
Supplementary Table 5
Location, parent material, and soil classification
Supplementary Table 6
Spearman’s correlation analyses for soil and pore water parameters
Supplementary Table 7
Relative importance of predictor values
Supplementary Table 8
Quantitative Recovery of As species in a fresh model pore water
Supplementary Table 9
Characteristics of irrigation water for mesocosm experiments
Rights and permissions
About this article
Cite this article
Wang, J., Kerl, C.F., Hu, P. et al. Thiolated arsenic species observed in rice paddy pore waters. Nat. Geosci. 13, 282–287 (2020). https://doi.org/10.1038/s41561-020-0533-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-020-0533-1
This article is cited by
-
Fertilization Enhances Grain Inorganic Arsenic Assimilation in Rice
Exposure and Health (2024)
-
Sulfate reduction accelerates groundwater arsenic contamination even in aquifers with abundant iron oxides
Nature Water (2023)
-
Sulfur being an overlooked promoter of groundwater arsenic contamination
Nature Water (2023)
-
Effect of phosphate on arsenic species uptake in plants under hydroponic conditions
Journal of Plant Research (2023)
-
Altering the localization and toxicity of arsenic in rice grain
Scientific Reports (2022)