Abstract
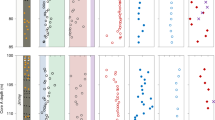
The first of the ‘Big Five’ Phanerozoic mass extinctions occurred in tandem with an episode of glaciation during the Hirnantian Age of the Late Ordovician. The mechanism or change in the carbon cycle that promoted this glaciation, thereby resulting in the extinction, is still debated. Here we report new, coupled nitrogen isotope analyses of bulk sediments and chlorophyll degradation products (porphyrins) from the Vinini Creek section (Vinini Formation, Nevada, USA) to show that eukaryotes increasingly dominated marine export production in the lead-up to the Hirnantian extinction. We then use these findings to evaluate changes in the carbon cycle by incorporating them into a biogeochemical model in which production is increased in response to an elevated phosphorus inventory, potentially caused by enhanced continental weathering in response to the activity of land plants and/or an episode of volcanism. The results suggest that expanded eukaryotic algal production may have increased the community average cell size, leading to higher export efficiency during the Late Katian. The coincidence of this community shift with a large-scale marine transgression increased organic carbon burial, drawing down CO2 and triggering the Hirnantian glaciation. This episode may mark an early Palaeozoic strengthening of the biological pump, which, for a short while, may have made eukaryotic algae indirect killers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Sepkoski, J. J. in Global Events and Event Stratigraphy in the Phanerozoic: Results of the International Interdisciplinary Cooperation in the IGCP-Project 216 ‘ Global Biological Events in Earth History’ (ed. Walliser, O. H.) 35–51 (Springer, Berlin Heidelberg, 1996).
Finnegan, S. et al. The magnitude and duration of Late Ordovician–Early Silurian glaciation. Science 331, 903–906 (2011).
Brenchley, P. et al. High-resolution stable isotope stratigraphy of Upper Ordovician sequences: constraints on the timing of bioevents and environmental changes associated with mass extinction and glaciation. Geol. Soc. Am. Bull. 115, 89–104 (2003).
Sheehan, P. M. The Late Ordovician mass extinction. Annu. Rev. Earth Planet. Sci. Lett. 29, 331–364 (2001).
Yapp, C. J. & Poths, H. Ancient atmospheric CO2 pressures inferred from natural goethites. Nature 355, 342 (1992).
Berner, R. A. GEOCARBSULF: a combined model for Phanerozoic atmospheric O2 and CO2. Geochim. Cosmochim. Acta 70, 5653–5664 (2006).
Tobin, K. J., Bergström, S. M. & De La Garza, P. A mid-Caradocian (453 Ma) drawdown in atmospheric pCO2 without ice sheet development? Palaeogeogr. Palaeoclimatol. Palaeoecol. 226, 187–204 (2005).
Pancost, R. D. et al. Reconstructing Late Ordovician carbon cycle variations. Geochim. Cosmochim. Acta 105, 433–454 (2013).
Kump, L. et al. A weathering hypothesis for glaciation at high atmospheric pCO2 during the Late Ordovician. Palaeogeogr. Palaeoclimatol. Palaeoecol. 152, 173–187 (1999).
Buggisch, W. et al. Did intense volcanism trigger the first Late Ordovician icehouse? Geology 38, 327–330 (2010).
Melchin, M. J., Mitchell, C. E., Holmden, C. & Štorch, P. Environmental changes in the Late Ordovician–early Silurian: review and new insights from black shales and nitrogen isotopes. Geol. Soc. Am. Bull. 125, 1635–1670 (2013).
Brenchley, P. et al. Bathymetric and isotopic evidence for a short-lived Late Ordovician glaciation in a greenhouse period. Geology 22, 295–298 (1994).
Brenchley, P., Carden, G. & Marshall, J. Environmental changes associated with the ‘first strike’ of the Late Ordovician mass extinction. Mod. Geol. 20, 69–82 (1995).
LaPorte, D. et al. Local and global perspectives on carbon and nitrogen cycling during the Hirnantian glaciation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 276, 182–195 (2009).
Luo, G. et al. Perturbation of the marine nitrogen cycle during the Late Ordovician glaciation and mass extinction. Palaeogeogr. Palaeoclimatol. Palaeoecol. 448, 339–348 (2016).
Rohrssen, M., Love, G. D., Fischer, W., Finnegan, S. & Fike, D. A. Lipid biomarkers record fundamental changes in the microbial community structure of tropical seas during the Late Ordovician Hirnantian glaciation. Geology 41, 127–130 (2013).
Newman, D. K., Neubauer, C., Ricci, J. N., Wu, C.-H. & Pearson, A. Cellular and molecular biological approaches to interpreting ancient biomarkers. Annu. Rev. Earth Planet. Sci. 44, 493–522 (2016).
Higgins, M. B., Robinson, R. S., Husson, J. M., Carter, S. J. & Pearson, A. Dominant eukaryotic export production during ocean anoxic events reflects the importance of recycled NH4 +. Proc. Natl Acad. Sci. USA 109, 2269–2274 (2012).
Kaljo, D., Männik, P., Martma, T. & Nõlvak, J. More about the Ordovician-Silurian transition beds at Mirny Creek, Omulev Mountains, NE Russia: carbon isotopes and conodonts Eston. J. Earth Sci. 61, 277–294 (2012).
Finney, S. C. et al. Late Ordovician mass extinction: a new perspective from stratigraphic sections in central Nevada. Geology 27, 215–218 (1999).
Finney, S. C., Berry, W. B. & Cooper, J. D. The influence of denitrifying seawater on graptolite extinction and diversification during the Hirnantian (latest Ordovician) mass extinction event. Lethaia 40, 281–291 (2007).
Higgins, M. B. et al. Paleoenvironmental implications of taxonomic variation among δ15N values of chloropigments. Geochim. Cosmochim. Acta 75, 7351–7363 (2011).
Sachs, J. P., Repeta, D. J. & Goericke, R. Nitrogen and carbon isotopic ratios of chlorophyll from marine phytoplankton. Geochim. Cosmochim. Acta 63, 1431–1441 (1999).
Beaumont, V. I., Jahnke, L. L. & Des Marais, D. J. Nitrogen isotopic fractionation in the synthesis of photosynthetic pigments in Rhodobacter capsulatus and Anabaena cylindrica. Org. Geochem. 31, 1075–1085 (2000).
Ahm, A.-S. C., Bjerrum, C. J. & Hammarlund, E. U. Disentangling the record of diagenesis, local redox conditions, and global seawater chemistry during the latest Ordovician glaciation. Earth Planet. Sci. Lett. 459, 145–156 (2017).
French, K., Rocher, D., Zumberge, J. & Summons, R. Assessing the distribution of sedimentary C40 carotenoids through time. Geobiology 13, 139–151 (2015).
Junium, C. K., Freeman, K. H. & Arthur, M. A. Controls on the stratigraphic distribution and nitrogen isotopic composition of zinc, vanadyl and free base porphyrins through Oceanic Anoxic Event 2 at Demerara Rise. Org. Geochem. 80, 60–71 (2015).
Delabroye, A. et al. Phytoplankton dynamics across the Ordovician/Silurian boundary at low palaeolatitudes: correlations with carbon isotopic and glacial events. Palaeogeogr. Palaeoclimatol. Palaeoecol. 312, 79–97 (2011).
Lenton, T. M., Crouch, M., Johnson, M., Pires, N. & Dolan, L. First plants cooled the Ordovician. Nat. Geosci. 5, 86–89 (2012).
Nodder, S. D. et al. Particle transformations and export flux during an in situ iron‐stimulated algal bloom in the Southern Ocean. Geophys. Res. Lett. 28, 2409–2412 (2001).
Kwon, E. Y., Primeau, F. & Sarmiento, J. L. The impact of remineralization depth on the air–sea carbon balance. Nat. Geosci. 2, 630–635 (2009).
Lomas, M. et al. Increased ocean carbon export in the Sargasso Sea linked to climate variability is countered by its enhanced mesopelagic attenuation. Biogeosciences 7, 57–70 (2010).
Bao, R. et al. Widespread dispersal and aging of organic carbon in shallow marginal seas. Geology 44, 791–794 (2016).
Hilligsøe, K. M. et al. Linking phytoplankton community size composition with temperature, plankton food web structure and sea–air CO2 flux. Deep Sea Res. Part I: Oceanogr. Res. Pap. 58, 826–838 (2011).
Michaels, A. F. & Silver, M. W. Primary production, sinking fluxes and the microbial food web. Deep Sea Res. Part A: Oceanogr. Res. Pap. 35, 473–490 (1988).
Armstrong, H. A., Baldini, J., Challands, T. J., Gröcke, D. R. & Owen, A. W. Response of the inter-tropical convergence zone to Southern Hemisphere cooling during upper Ordovician glaciation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 284, 227–236 (2009).
Bjerrum, C. J., Bendtsen, J. & Legarth, J. J. F. Modeling organic carbon burial during sea level rise with reference to the Cretaceous. Geochem. Geophys. Geosyst. 7, Q05008 (2006).
Falkowski, P. G., Barber, R. T. & Smetacek, V. Biogeochemical controls and feedbacks on ocean primary production. Science 281, 200–206 (1998).
Bergman, N. M., Lenton, T. M. & Watson, A. J. COPSE: a new model of biogeochemical cycling over Phanerozoic time. Am. J. Sci. 304, 397–437 (2004).
Pohl, A., Donnadieu, Y., Le Hir, G., Buoncristiani, J.-F. & Vennin, E. Effect of the Ordovician paleogeography on the (in)stability of the climate. Clim. Past. 10, 2053 (2014).
Chen, B. & Liu, H. Relationships between phytoplankton growth and cell size in surface oceans: interactive effects of temperature, nutrients, and grazing. Limnol. Oceanogr. 55, 965–972 (2010).
Fan, J., Peng, Pa & Melchin, M. Carbon isotopes and event stratigraphy near the Ordovician–Silurian boundary, Yichang, South China. Palaeogeogr. Palaeoclimatol. Palaeoecol. 276, 160–169 (2009).
Holmden, C. et al. Nd isotope records of late Ordovician sea-level change—implications for glaciation frequency and global stratigraphic correlation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 386, 131–144 (2013).
Wilde, P. in Advances in Ordovician Geology Paper 90-9 (eds Barnes, C. R. & Williams, S. H.) 283–298 (Geological Survey of Canada, Ottawa, 1991).
Berry, W. B. Black shales: An Ordovician perspective. Geol. Soc. Am. Spec. Pap. 466, 141–147 (2010).
Higgins, M. B., Robinson, R. S., Casciotti, K. L., McIlvin, M. R. & Pearson, A. A method for determining the nitrogen isotopic composition of porphyrins. Anal. Chem. 81, 184–192 (2009).
Braman, R. S. & Hendrix, S. A. Nanogram nitrite and nitrate determination in environmental and biological materials by vanadium (III) reduction with chemiluminescence detection. Anal. Chem. 61, 2715–2718 (1989).
Sigman, D. et al. A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater. Anal. Chem. 73, 4145–4153 (2001).
Hayes, J. M. Fractionation of carbon and hydrogen isotopes in biosynthetic processes. Rev. Mineral. Geochem. 43, 225–277 (2001).
Berry, W. B. & Wilde, P. Progressive ventilation of the oceans; an explanation for the distribution of the lower Paleozoic black shales. Am. J. Sci. 278, 257–275 (1978).
Leggett, J. British Lower Palaeozoic black shales and their palaeo-oceanographic significance. J. Geol. Soc. 137, 139–156 (1980).
Berner, R. A. The Phanerozoic Carbon Cycle: CO 2 and O 2 (Oxford Univ. Press, Oxford, 2004).
Reinhard, C. T. et al. Evolution of the global phosphorus cycle. Nature 541, 386–389 (2017).
Haq, B. U. & Schutter, S. R. A chronology of Paleozoic sea-level changes. Science 322, 64–68 (2008).
Klaas, C. & Archer, D. E. Association of sinking organic matter with various types of mineral ballast in the deep sea: implications for the rain ratio. Global Biogeochem. Cycles 16, 1116 (2002).
Kostadinov, T., Siegel, D. & Maritorena, S. Global variability of phytoplankton functional types from space: assessment via the particle size distribution. Biogeosciences 7, 3239–3257 (2010).
Acknowledgements
We gratefully acknowledge X. Wang (Johns Hopkins University) for valuable discussions on the execution and C++ code modification of the COPSE model. J.S. acknowledges financial support from Z. T. Guo and the China Scholarship Council. We thank D. T. Johnston and J. J. Kharbush for helpful discussions, and B. D. A. Naafs, S. J. Hurley and S. J. Carter for assistance with sample preparation and machine use. We thank A. J. Kaufman for his help with bulk N-isotopic analysis of Vinini Creek samples. We thank D. Hu for the bulk N-isotope analysis of XY-5 samples. This study was supported by the National Natural Science Foundation of China (grant numbers 41520104007 and 41330102). A.P. received support from the NASA Astrobiology Institute and the Gordon and Betty Moore Foundation.
Author information
Authors and Affiliations
Contributions
J.S. and A.P. designed the paper. J.S., G.A.H. and S.D.W. performed the biomarker analysis. J.S. and K.C. performed the bulk sediment analysis. J.S., A.P. and Y.G.Z. contributed to the data interpretation. D.L., S.C.F. and Y.S. collected the samples and provided a robust stratigraphic framework. J.S., A.P. and Y.G.Z. designed the biogeochemical model. J.S. wrote code and performed model simulations. J.S. and A.P. wrote the manuscript, with inputs from G.A.H., Y.G.Z., S.C.F. and S.D.W.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures and Tables
Rights and permissions
About this article
Cite this article
Shen, J., Pearson, A., Henkes, G.A. et al. Improved efficiency of the biological pump as a trigger for the Late Ordovician glaciation. Nature Geosci 11, 510–514 (2018). https://doi.org/10.1038/s41561-018-0141-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-018-0141-5
This article is cited by
-
A nutrient control on expanded anoxia and global cooling during the Late Ordovician mass extinction
Communications Earth & Environment (2022)
-
Different controls on the Hg spikes linked the two pulses of the Late Ordovician mass extinction in South China
Scientific Reports (2022)
-
Ordovician–Silurian true polar wander as a mechanism for severe glaciation and mass extinction
Nature Communications (2022)
-
Early and late phases of the Permian–Triassic mass extinction marked by different atmospheric CO2 regimes
Nature Geoscience (2022)
-
Late Ordovician climate change and extinctions driven by elevated volcanic nutrient supply
Nature Geoscience (2021)