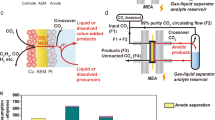
Abstract
Carbon dioxide/monoxide (CO2/CO) electrolysis provides a means to convert emissions into multicarbon products. However, impractical energy and carbon efficiencies limit current systems. Here we show that these inefficiencies originate from uncontrolled gas/ion distributions in the local reaction environment. Understanding of the flows of cations and anions motivated us to seek a route to block cation migration to the catalyst surface—a strategy we instantiate using a covalent organic framework (COF) in bulk heterojunction with a catalyst. The π-conjugated hydrophobic COFs constrain cation (potassium) diffusion via cation–π interactions, while promoting anion (hydroxide) and gaseous feedstock adsorption on the catalyst surface. As a result, a COF-mediated catalyst enables electrosynthesis of multicarbon products from CO for 200 h at a single-pass carbon efficiency of 95%, an energy efficiency of 40% and a current density of 240 mA cm−2.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All of the data supporting the findings of this study are available within the published article and its Supplementary Information files. Source data are provided with this paper.
References
Li, C. W., Ciston, J. & Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 508, 504–507 (2014).
Huang, J. E. et al. CO2 electrolysis to multicarbon products in strong acid. Science 372, 1074–1078 (2021).
Nitopi, S. et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 119, 7610–7672 (2019).
Jouny, M., Hutchings, G. S. & Jiao, F. Carbon monoxide electroreduction as an emerging platform for carbon utilization. Nat. Catal. 2, 1062–1070 (2019).
De Luna, P. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, eaav3506 (2019).
Li, J. et al. Enhanced multi-carbon alcohol electroproduction from CO via modulated hydrogen adsorption. Nat. Commun. 11, 3685 (2020).
Wang, X. et al. Efficient electrically powered CO2-to-ethanol via suppression of deoxygenation. Nat. Energy 5, 478–486 (2020).
Ozden, A. et al. High-rate and efficient ethylene electrosynthesis using a catalyst/promoter/transport layer. ACS Energy Lett. 5, 2811–2818 (2020).
Shin, H., Hansen, K. U. & Jiao, F. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain. 4, 911–919 (2021).
Chen, C., Li, Y. & Yang, P. Address the “alkalinity problem” in CO2 electrolysis with catalyst design and translation. Joule 5, 737–742 (2021).
Rabinowitz, J. A. & Kanan, M. W. The future of low-temperature carbon dioxide electrolysis depends on solving one basic problem. Nat. Commun. 11, 5231 (2020).
Ren, S. et al. Molecular electrocatalysts can mediate fast, selective CO2 reduction in a flow cell. Science 365, 367–369 (2019).
Kim, D. et al. Selective CO2 electrocatalysis at the pseudocapacitive nanoparticle/ordered-ligand interlayer. Nat. Energy 5, 1032–1042 (2020).
Li, J. et al. Efficient electrocatalytic CO2 reduction on a three-phase interface. Nat. Catal. 1, 592–600 (2018).
Ripatti, D. S., Veltman, T. R. & Kanan, M. W. Carbon monoxide gas diffusion electrolysis that produces concentrated C2 products with high single-pass conversion. Joule 3, 240–256 (2019).
De Arquer, P. F. et al. CO2 electrolysis to multicarbon products at activities greater than 1 A cm−2. Science 367, 661–666 (2020).
Dinh, C. T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Jouny, M., Luc, W. & Jiao, F. High-rate electroreduction of carbon monoxide to multi-carbon products. Nat. Catal. 1, 748–755 (2018).
Ma, W. et al. Electrocatalytic reduction of CO2 to ethylene and ethanol through hydrogen-assisted C–C coupling over fluorine-modified copper. Nat. Catal. 3, 478–487 (2020).
Gabardo, C. M. et al. Continuous carbon dioxide electroreduction to concentrated multi-carbon products using a membrane electrode assembly. Joule 3, 2777–2791 (2019).
O’Brien, C. P. et al. Single pass CO2 conversion exceeding 85% in the electrosynthesis of multicarbon products via local CO2 regeneration. ACS Energy Lett. 6, 2952–2959 (2021).
Ozden, A. et al. Cascade CO2 electroreduction enables efficient carbonate-free production of ethylene. Joule 5, 706–719 (2021).
Ozden, A. et al. Carbon-efficient carbon dioxide electrolysers. Nat. Sustain. 5, 563–573 (2022).
Sisler, J. et al. Ethylene electrosynthesis: a comparative techno-economic analysis of alkaline vs membrane electrode assembly vs CO2–CO–C2H4 tandems. ACS Energy Lett. 6, 997–1002 (2021).
Xu, Y. et al. Self-cleaning CO2 reduction systems: unsteady electrochemical forcing enables stability. ACS Energy Lett. 6, 809–815 (2021).
Endrődi, B. et al. Operando cathode activation with alkali metal cations for high current density operation of water-fed zero-gap carbon dioxide electrolysers. Nat. Energy 6, 439–448 (2021).
Bohra, D., Chaudhry, J. H., Burdyny, T., Pidko, E. A. & Smith, W. A. Modeling the electrical double layer to understand the reaction environment in a CO2 electrocatalytic system. Energy Environ. Sci. 12, 3380–3389 (2019).
Lin, S. et al. Covalent organic frameworks comprising cobalt porphyrins for catalytic CO2 reduction in water. Science 349, 1208–1213 (2015).
Kandambeth, S., Kale, V. S., Shekhah, O., Alshareef, H. N. & Eddaoudi, M. 2D covalent-organic framework electrodes for supercapacitors and rechargeable metal-ion batteries. Adv. Energy Mater. 12, 2100177 (2022).
Chen, X., Zhang, H., Ci, C., Sun, W. & Wang, Y. Few-layered boronic ester based covalent organic frameworks/carbon nanotube composites for high-performance K-organic batteries. ACS Nano 13, 3600–3607 (2019).
Kandambeth, S. et al. Covalent organic frameworks as negative electrodes for high-performance asymmetric supercapacitors. Adv. Energy Mater. 10, 2001673 (2020).
Ma, J. C. & Dougherty, D. A. The cation–π interaction. Chem. Rev. 97, 1303–1324 (1997).
Liu, M. et al. Enhanced electrocatalytic CO2 reduction via field-induced reagent concentration. Nature 537, 382–386 (2016).
Zhan, C. et al. Revealing the CO coverage-driven C–C coupling mechanism for electrochemical CO2 reduction on Cu2O nanocubes via operando Raman spectroscopy. ACS Catal. 11, 7694–7701 (2021).
Hoang, T. T. H. et al. Nanoporous copper–silver alloys by additive-controlled electrodeposition for the selective electroreduction of CO2 to ethylene and ethanol. J. Am. Chem. Soc. 140, 5791–5797 (2018).
Niaura, G. Surface-enhanced Raman spectroscopic observation of two kinds of adsorbed OH− ions at copper electrode. Electrochim. Acta 45, 3507–3519 (2000).
Zhao, Y. et al. Speciation of Cu surfaces during the electrochemical CO reduction reaction. J. Am. Chem. Soc. 142, 9735–9743 (2020).
Verdaguer-Casadevall, A. et al. Probing the active surface sites for CO reduction on oxide-derived copper electrocatalysts. J. Am. Chem. Soc. 137, 9808–9811 (2015).
Wakerley, D. et al. Bio-inspired hydrophobicity promotes CO2 reduction on a Cu surface. Nat. Mater. 18, 1222–1227 (2019).
Xing, Z., Hu, L., Ripatti, D. S., Hu, X. & Feng, X. Enhancing carbon dioxide gas-diffusion electrolysis by creating a hydrophobic catalyst microenvironment. Nat. Commun. 12, 136 (2021).
Kim, C. et al. Tailored catalyst microenvironments for CO2 electroreduction to multicarbon products on copper using bilayer ionomer coatings. Nat. Energy 6, 1026–1034 (2021).
Monteiro, M. C. O. et al. Absence of CO2 electroreduction on copper, gold and silver electrodes without metal cations in solution. Nat. Catal. 4, 654–662 (2021).
Bondue, C. J., Graf, M., Goyal, A. & Koper, M. T. M. Suppression of hydrogen evolution in acidic electrolytes by electrochemical CO2 reduction. J. Am. Chem. Soc. 143, 279–285 (2021).
Rubcumintara, T. & Han, K. N. Metal ionic diffusivity: measurement and application. Miner. Process. Extr. Metall. Rev. 7, 23–47 (2007).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Blöchl, P. E., Jepsen, O. & Andersen, O. K. Improved tetrahedron method for Brillouin-zone integrations. Phys. Rev. B 49, 16223–16233 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Acknowledgements
This work was financially supported by the Ontario Research Fund – Research Excellence programme, the Natural Sciences and Engineering Research Council (NSERC) of Canada and Natural Resources Canada’s Clean Growth Program. This research used synchrotron resources of the Advanced Photon Source (an Office of Science User Facility operated for the US Department of Energy (DOE) Office of Science by Argonne National Laboratory) and was supported by the US DOE under contract number DE-AC02-06CH11357, as well as the Canadian Light Source and its funding partners. Support from the Canada Research Chairs Program is gratefully acknowledged, as is support from an NSERC E.W.R. Steacie Fellowship to D.S. J.L. thanks the National Natural Science Foundation of China (grant number BE3250011), the National Key Research and Development Program of China (grant number 2022YFA1505100), and Shanghai Jiao Tong University (grant number WH220432516) for support. F.P.G.d.A. acknowledges funding from CEX2019-000910-S (MCIN/AEI/10.13039/501100011033), Fundación Cellex, Fundació Mir-Puig, Generalitat de Catalunya through CERCA and the La Caixa Foundation (100010434; EU Horizon 2020 Marie Skłodowska-Curie grant agreement 847648).
Author information
Authors and Affiliations
Contributions
D.S., E.H.S., M.E. and J.L. supervised the project. A.O., J.L. and S.K. conceived of the idea. A.O. and J.L. designed and carried out all of the electrochemical experiments. A.O. synthesized the catalysts and fabricated the electrodes and slim flow-cell electrolyser. A.O. performed the SEM, TEM and XPS measurements. S.L. performed the COMSOL simulations. S.K., V.S.K. and P.M.B. synthesized and characterized the Hex–Aza–COF nanosheets. Y.Z.F. performed the XAS measurements. Y.-K.W. performed the XRD measurements. X.-Y.L. performed the DFT calculations with the assistance of P.O. T.A., F.P.G.d.A. and A.H.I. contributed to data analysis. A.O. and J.L. cowrote the manuscript. D.S., E.H.S. and O.S. contributed to manuscript editing. All authors discussed the results and assisted during manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Ezra Clark and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs., 1–57, notes 1–7, Tables 1–50 and references 1–15.
Supplementary Data
Source data for Supplementary Figs. 2a–f, 3a–f, 18a,b, 19a–f, 20, 24a–c, 26a,b, 27a,b, 29b,c, 32, 33, 35, 36a–f, 37, 38a–c, 39, 40a–c, 41, 42a–c, 43a–d, 51–54, 56 and 57.
Source data
Source Data Fig. 1
Source data for Fig. 1b.
Source Data Fig. 3
Source data for Fig. 3f–h.
Source Data Fig. 4
Source data for Fig. 4a.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ozden, A., Li, J., Kandambeth, S. et al. Energy- and carbon-efficient CO2/CO electrolysis to multicarbon products via asymmetric ion migration–adsorption. Nat Energy 8, 179–190 (2023). https://doi.org/10.1038/s41560-022-01188-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-022-01188-2
This article is cited by
-
Acidic media enables oxygen-tolerant electrosynthesis of multicarbon products from simulated flue gas
Nature Communications (2024)
-
Self-supporting BiCu/carbon hybrid nanofiber membrane promotes efficient CO2 electroreduction to formate
Science China Materials (2024)
-
Maximizing single-pass conversion does not result in practical readiness for CO2 reduction electrolyzers
Nature Communications (2023)
-
Accelerating CO2 electrochemical conversion towards industrial implementation
Nature Communications (2023)
-
Enhancing carbon utilization
Nature Energy (2023)