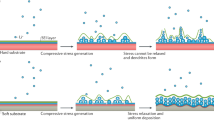
Abstract
Batteries based on multivalent metals have the potential to meet the future needs of large-scale energy storage, due to the relatively high abundance of elements such as magnesium, calcium, aluminium and zinc in the Earth’s crust. However, the complexity of multivalent metal-ion chemistries has led to rampant confusions, technical challenges, and eventually doubts and uncertainties about the future of these technologies. In this Review, we clarify the key strengths as well as common misconceptions of multivalent metal-based batteries. We then examine the growth behaviour of metal anodes, which is crucial for their safety promises but hitherto unestablished. We further discuss scrutiny of anode efficiency and cathode storage mechanism pertaining to complications arising from electrolyte solutions. Finally, we critically review existing cathode materials and discuss design strategies to enable genuine multivalent metal-ion-based energy storage materials with competitive performance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
15 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41560-020-0676-8
References
Weil, M., Ziemann, S. & Peters, J. in Behaviour of Lithium-Ion Batteries in Electric Vehicles: Battery Health, Performance, Safety, and Cost (eds Pistoia, G. & Liaw, B.) 59–74 (Springer International Publishing, 2018).
Salama, M. et al. Metal–sulfur batteries: overview and research methods. ACS Energy Lett. 4, 436–446 (2019).
Liu, M. et al. Spinel compounds as multivalent battery cathodes: a systematic evaluation based on ab initio calculations. Energy Environ. Sci. 8, 964–974 (2015).
Chung, S.-H. & Manthiram, A. Current status and future prospects of metal–sulfur batteries. Adv. Mater. 31, 1901125 (2019).
Rong, Z. et al. Materials design rules for multivalent ion mobility in intercalation structures. Chem. Mater. 27, 6016–6021 (2015). This work proposed the relationship between ion coordination environment and energy barrier for solid-state multivalent metal-ion diffusion.
Matsui, M. Study on electrochemically deposited Mg metal. J. Power Sources 196, 7048–7055 (2011). This work investigated the origin of the non-dendritic magnesium deposition in comparison with the dendritic lithium deposition.
Crowe, A. J., DiMeglio, J. L., Stringham, K. K. & Bartlett, B. M. Kinetics of magnesium deposition and stripping from non-aqueous electrolytes. J. Phys. Chem. C 121, 20613–20620 (2017).
Tutusaus, O. et al. An efficient halogen-free electrolyte for use in rechargeable magnesium batteries. Angew. Chem. Int. Ed. 54, 7900–7904 (2015). This work reported a non-corrosive, non-nucleophilic electrolyte solutions that enable reversible magnesium plating and stripping.
Ta, K., See, K. A. & Gewirth, A. A. Elucidating Zn and Mg electrodeposition mechanisms in nonaqueous electrolytes for next-generation metal batteries. J. Phys. Chem. C 122, 13790–13796 (2018).
Jäckle, M., Helmbrecht, K., Smits, M., Stottmeister, D. & Groß, A. Self-diffusion barriers: possible descriptors for dendrite growth in batteries? Energy Environ. Sci. 11, 3400–3407 (2018).
Jäckle, M. & Groß, A. Microscopic properties of lithium, sodium, and magnesium battery anode materials related to possible dendrite growth. J. Chem. Phys. 141, 174710 (2014).
Ling, C., Banerjee, D. & Matsui, M. Study of the electrochemical deposition of Mg in the atomic level: Why it prefers the non-dendritic morphology. Electrochim. Acta 76, 270–274 (2012).
Gregory, T. D., Hoffman, R. J. & Winterton, R. C. Nonaqueous electrochemistry of magnesium: applications to energy storage. J. Electrochem. Soc. 137, 775–780 (1990).
Davidson, R. et al. Mapping mechanisms and growth regimes of magnesium electrodeposition at high current densities. Mater. Horiz. 7, 843–854 (2020).
Ding, M. S., Diemant, T., Behm, R. J., Passerini, S. & Giffin, G. A. Dendrite growth in Mg metal cells containing Mg(TFSI)2/glyme electrolytes. J. Electrochem. Soc. 165, A1983–A1990 (2018).
Ponrouch, A., Frontera, C., Barde, F. & Palacin, M. R. Towards a calcium-based rechargeable battery. Nat. Mater. 15, 169–172 (2016).
Wang, D. et al. Plating and stripping calcium in an organic electrolyte. Nat. Mater. 17, 16 (2017). This work reported calcium plating and stripping at room temperature in an organic electrolyte solution with low polarization and relatively high efficiency.
Shyamsunder, A., Blanc, L. E., Assoud, A. & Nazar, L. F. Reversible calcium plating and stripping at room temperature using a borate salt. ACS Energy Lett. 4, 2271–2276 (2019).
Li, Z., Fuhr, O., Fichtner, M. & Zhao-Karger, Z. Towards stable and efficient electrolytes for room-temperature rechargeable calcium batteries. Energy Environ. Sci. 12, 3496–3501 (2019).
Pan, H. et al. Reversible aqueous zinc/manganese oxide energy storage from conversion reactions. Nat. Energy 1, 16039 (2016).
Zhao, Q. et al. High-capacity aqueous zinc batteries using sustainable quinone electrodes. Sci. Adv. 4, eaao1761 (2018).
Wang, F. et al. Highly reversible zinc metal anode for aqueous batteries. Nat. Mater. 17, 543–549 (2018).
Han, S.-D. et al. Origin of electrochemical, structural, and transport properties in nonaqueous zinc electrolytes. ACS Appl. Mater. Inter. 8, 3021–3031 (2016).
Qian, J. et al. High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015).
Pradhan, D. & Reddy, R. G. Dendrite-free aluminum electrodeposition from AlCl3-1-ethyl-3-methyl-imidazolium chloride ionic liquid electrolytes. Metall. Mater. Trans. B 43, 519–531 (2012).
Woods, J., Bhattarai, N., Chapagain, P., Yang, Y. & Neupane, S. In situ transmission electron microscopy observations of rechargeable lithium ion batteries. Nano Energy 56, 619–640 (2019).
Chen, H. et al. Oxide film efficiently suppresses dendrite growth in aluminum-ion battery. ACS Appl. Mater. Inter. 9, 22628–22634 (2017).
Peled, E. & Menkin, S. Review—SEI: Past, present and future. J. Electrochem. Soc. 164, A1703–A1719 (2017).
Aurbach, D., Skaletsky, R. & Gofer, Y. The electrochemical behavior of calcium electrodes in a few organic electrolytes. J. Electrochem. Soc. 138, 3536–3545 (1991).
Yu, J., McMahon, B. W., Boatz, J. A. & Anderson, S. L. Aluminum nanoparticle production by acetonitrile-assisted milling: effects of liquid- vs vapor-phase milling and of milling method on particle size and surface chemistry. J. Phys. Chem. C 120, 19613–19629 (2016).
Lu, Z., Schechter, A., Moshkovich, M. & Aurbach, D. On the electrochemical behavior of magnesium electrodes in polar aprotic electrolyte solutions. J. Electroanal. Chem. 466, 203–217 (1999).
Singh, N. et al. Achieving high cycling rates via in situ generation of active nanocomposite metal anodes. ACS Appl. Energy Mater. 1, 4651–4661 (2018).
Son, S.-B. et al. An artificial interphase enables reversible magnesium chemistry in carbonate electrolytes. Nat. Chem. 10, 532–539 (2018).
Gao, T. et al. Existence of solid electrolyte interphase in Mg batteries: Mg/S chemistry as an example. ACS Appl. Mater. Inter. 10, 14767–14776 (2018).
Chen, T., Ceder, G., Sai Gautam, G. & Canepa, P. Evaluation of Mg compounds as coating materials in Mg batteries. Front. Chem. 7, 24 (2019).
Tamura, S., Yamane, M., Hoshino, Y. & Imanaka, N. Highly conducting divalent Mg2+ cation solid electrolytes with well-ordered three-dimensional network structure. J. Solid State Chem. 235, 7–11 (2016).
Kisu, K. et al. Magnesium borohydride ammonia borane as a magnesium ionic conductor. ACS Appl. Energy Mater. 3, 3174–3179 (2020).
Canepa, P. et al. High magnesium mobility in ternary spinel chalcogenides. Nat. Commun. 8, 1759 (2017).
Pour, N., Gofer, Y., Major, D. T. & Aurbach, D. Structural analysis of electrolyte solutions for rechargeable Mg batteries by stereoscopic means and DFT calculations. J. Am. Chem. Soc. 133, 6270–6278 (2011).
Pour, N. et al. Multinuclear magnetic resonance spectroscopy and density function theory calculations for the identification of the equilibrium species in THF solutions of organometallic complexes suitable as electrolyte solutions for rechargeable Mg batteries. Organometallics 32, 3165–3173 (2013).
Kim, H. S. et al. Structure and compatibility of a magnesium electrolyte with a sulphur cathode. Nat. Commun. 2, 427 (2011). This work reported non-nucleophilic electrolyte solutions that enable reversible magnesium plating and stripping.
Doe, R. E. et al. Novel, electrolyte solutions comprising fully inorganic salts with high anodic stability for rechargeable magnesium batteries. Chem. Commun. 50, 243–245 (2014).
Carter, T. J. et al. Boron clusters as highly stable magnesium-battery electrolytes. Angew. Chem. Int. Ed. 53, 3173–3177 (2014).
Zhao-Karger, Z. et al. Toward highly reversible magnesium–sulfur batteries with efficient and practical Mg[B(hfip)4]2 electrolyte. ACS Energy Lett. 3, 2005–2013 (2018).
Lipson, A. L. et al. Practical stability limits of magnesium electrolytes. J. Electrochem. Soc. 163, A2253–A2257 (2016).
Liu, T. B. et al. A facile approach using MgCl2 to formulate high performance Mg2+ electrolytes for rechargeable Mg batteries. J. Mater. Chem. A 2, 3430–3438 (2014).
Du, A. et al. An efficient organic magnesium borate-based electrolyte with non-nucleophilic characteristics for magnesium–sulfur battery. Energy Environ. Sci. 10, 2616–2625 (2017).
Luo, J., Bi, Y., Zhang, L., Zhang, X. & Liu, T. L. A stable, non-corrosive perfluorinated pinacolatoborate Mg electrolyte for rechargeable Mg. batteries. Angew. Chem. Int. Ed. 58, 6967–6971 (2019).
Cheng, Y. W. et al. Highly active electrolytes for rechargeable Mg batteries based on a [Mg2(μ-Cl)2]2+ cation complex in dimethoxyethane. Phys. Chem. Chem. Phys. 17, 13307–13314 (2015).
Shterenberg, I. et al. Evaluation of (CF3SO2)2N− (TFSI) based electrolyte solutions for Mg batteries. J. Electrochem. Soc. 162, A7118–A7128 (2015).
Zhang, Y., Liu, S., Ji, Y., Ma, J. & Yu, H. Emerging nonaqueous aluminum-ion batteries: challenges, status, and perspectives. Adv. Mater. 30, 1706310 (2018).
Attias, R., Salama, M., Hirsch, B., Gofer, Y. & Aurbach, D. Solvent effects on the reversible intercalation of magnesium-ions into V2O5 electrodes. ChemElectroChem 5, 3514–3524 (2018).
Salama, M. et al. Unique behavior of dimethoxyethane (DME)/Mg(N(SO2CF3)2)2 solutions. J. Phys. Chem. C 120, 19586–19594 (2016).
Seguin, T. J., Hahn, N. T., Zavadil, K. R. & Persson, K. A. Elucidating non-aqueous solvent stability and associated decomposition mechanisms for Mg energy storage applications from first-principles. Front. Chem. 7, 175 (2019).
Senoh, H. et al. Sulfone-based electrolyte solutions for rechargeable magnesium batteries using 2,5-dimethoxy-1,4-benzoquinone positive electrode. J. Electrochem. Soc. 161, A1315–A1320 (2014).
Yagi, S., Tanaka, A., Ichikawa, Y., Ichitsubo, T. & Matsubara, E. Electrochemical stability of magnesium battery current collectors in a grignard reagent-based electrolyte. J. Electrochem. Soc. 160, C83–C88 (2013).
Hahn, N. T. et al. Enhanced stability of the carba-closo-dodecaborate anion for high-voltage battery electrolytes through rational design. J. Am. Chem. Soc. 140, 11076–11084 (2018).
Rajput, N. N., Qu, X., Sa, N., Burrell, A. K. & Persson, K. A. The coupling between stability and ion pair formation in magnesium electrolytes from first-principles quantum mechanics and classical molecular dynamics. J. Am. Chem. Soc. 137, 3411–3420 (2015).
Attias, R., Salama, M., Hirsch, B., Goffer, Y. & Aurbach, D. Anode-electrolyte interfaces in secondary magnesium batteries. Joule 3, 27–52 (2019).
Adams, B. D., Zheng, J., Ren, X., Xu, W. & Zhang, J.-G. Accurate determination of coulombic efficiency for lithium metal anodes and lithium metal batteries. Adv. Energy Mater. 8, 1702097 (2018).
Sun, X., Bonnick, P. & Nazar, L. F. Layered TiS2 positive electrode for Mg batteries. ACS Energy Lett. 1, 297–301 (2016).
Yoo, H. D. et al. Intercalation of magnesium into a layered vanadium oxide with high capacity. ACS Energy Lett. 4, 1528–1534 (2019).
Canepa, P. et al. Odyssey of multivalent cathode materials: open questions and future challenges. Chem. Rev. 117, 4287–4341 (2017).
Sa, N. et al. Is alpha-V2O5 a cathode material for Mg insertion batteries? J. Power Sources 323, 44–50 (2016).
Verrelli, R. et al. On the strange case of divalent ions intercalation in V2O5. J. Power Sources 407, 162–172 (2018). This work demonstrated the insuitability of a-V2O5 for magnesium and calcium storage despite the long-time belief of this material as a host for divalent metal ions, highlighting the importance of caution in deducing cathode storage chemistries for multivalent batteries.
Zhao, Q. et al. Solid electrolyte interphases for high-energy aqueous aluminum electrochemical cells. Sci. Adv. 4, eaau8131 (2018).
Dong, H. et al. Directing Mg-storage chemistry in organic polymers toward high-energy Mg batteries. Joule 3, 782–793 (2019). This work distinguished between complex-ion storage and pure metal-ion storage in magnesium battery cathodes and demonstrated the importance of the latter for practical cells performance.
Lin, M.-C. et al. An ultrafast rechargeable aluminium-ion battery. Nature 520, 324–328 (2015).
Kim, D. J. et al. Rechargeable aluminium organic batteries. Nat. Energy 4, 51–59 (2018).
Li, Z. et al. Fast kinetics of multivalent intercalation chemistry enabled by solvated magnesium-ions into self-established metallic layered materials. Nat. Commun. 9, 5115 (2018).
Haber, S. & Leskes, M. What can we learn from solid state NMR on the electrode–electrolyte interface? 30, 1706496 (2018).
Kundu, D. et al. Aqueous vs. nonaqueous Zn-ion batteries: consequences of the desolvation penalty at the interface. Energy Environ. Sci. 11, 881–892 (2018).
Wu, C. et al. Electrochemically activated spinel manganese oxide for rechargeable aqueous aluminum battery. Nat. Commun. 10, 7 (2019).
Wang, R. Y., Wessells, C. D., Huggins, R. A. & Cui, Y. Highly reversible open framework nanoscale electrodes for divalent Ion batteries. Nano Lett. 13, 5748–5752 (2013).
Kraychyk, K. V., Wang, S., Piveteau, L. & Koyalenko, M. V. Efficient aluminum chloride natural graphite battery. Chem. Mater. 29, 4484–4492 (2017).
Aurbach, D. et al. Prototype systems for rechargeable magnesium batteries. Nature 407, 724–727 (2000). This work demonstrated reversible and reasonably fast magnesium-ion battery systems with the discovery of suitable complex ethereal electrolyte solutions and Chevrel phase cathodes such as Mo6S8.
Sun, X. Q. et al. A high capacity thiospinel cathode for Mg batteries. Energy Environ. Sci. 9, 2273–2277 (2016). This work reported the first magnesium storage cathode material since the discovery of Mo6S8 that shows convincing performance.
Pan, C., Zhang, R., Nuzzo, R. G. & Gewirth, A. A. ZnNixMnxCo2–2xO4 spinel as a high-voltage and high-capacity cathode material for nonaqueous Zn-ion batteries. Adv. Energy Mater. 8, 1800589 (2018).
Pan, C., Nuzzo, R. G. & Gewirth, A. A. ZnAlxCo2–xO4 spinels as cathode materials for non-aqueous Zn batteries with an open circuit voltage of ≤2 V. Chem. Mater. 29, 9351–9359 (2017).
Yaghoobnejad Asl, H. & Manthiram, A. Mass transfer of divalent ions in an oxide host: comparison of Mg2+ and Zn2+ diffusion in hexagonal KxW3O9 bronze. Chem. Mater. 31, 2296–2307 (2019).
Geng, L. et al. Crystal structure transformation in Chevrel phase Mo6S8 induced by aluminum intercalation. Chem. Mater. 30, 8420–8425 (2018).
Wan, L. W. F., Perdue, B. R., Apblett, C. A. & Prendergast, D. Mg desolvation and intercalation mechanism at the Mo6S8 chevrel phase surface. Chem. Mater. 27, 5932–5940 (2015).
Levi, M. D. et al. The effect of the anionic framework of Mo6X8 Chevrel phase (X = S, Se) on the thermodynamics and the kinetics of the electrochemical insertion of Mg2+ ions. Solid State Ionics 176, 1695–1699 (2005).
West, A. R. in Basic solid state chemistry Ch. 4 (John Wiley & Sons, 1988).
Adelstein, N. & Wood, B. C. Role of dynamically frustrated bond disorder in a Li+ superionic solid electrolyte. Chem. Mater. 28, 7218–7231 (2016).
Mao, M. et al. Tuning anionic chemistry to improve kinetics of Mg intercalation. Chem. Mater. 31, 3183–3191 (2019).
Brown, I. D. What factors determine cation coordination numbers? Acta Crystallogr. B44, 545–553 (1988).
Hannah, D. C., Sai Gautam, G., Canepa, P., Rong, Z. & Ceder, G. Magnesium ion mobility in post-spinels accessible at ambient pressure. Chem. Commun. 53, 5171–5174 (2017).
Jung, S. C. & Han, Y.-K. Fast magnesium ion transport in the Bi/Mg3Bi2 two-phase electrode. J. Phys. Chem. C 122, 17643–17649 (2018).
Rong, Z. et al. Fast Mg2+ diffusion in Mo3(PO4)3O for Mg batteries. Chem. Commun. 53, 7998–8001 (2017).
Zhao-Karger, Z. et al. Performance improvement of magnesium sulfur batteries with modified non-nucleophilic electrolytes. Adv. Energy Mater. 5, 1401155 (2015).
Zhang, Z. et al. Novel design concepts of efficient Mg-ion electrolytes toward high-performance magnesium-selenium and magnesium-sulfur batteries. Adv. Energy Mater. 7, 1602055 (2017).
Tian, H. et al. High power rechargeable magnesium/iodine battery chemistry. Nat. Commun. 8, 14083 (2017).
Mao, M. et al. High-energy-density rechargeable Mg battery enabled by a displacement reaction. Nano Lett. 19, 6665–6672 (2019).
Gao, T. et al. Reversible S0/MgSx redox chemistry in a MgTFSI2/MgCl2/DME electrolyte for rechargeable Mg/S batteries. Angew. Chem. Int. Ed. 56, 13526–13530 (2017).
Salama, M. et al. On the feasibility of practical Mg–S batteries: practical limitations associated with metallic magnesium anodes. ACS Appl. Mater. Inter. 10, 36910–36917 (2018).
Gao, T. et al. Thermodynamics and kinetics of sulfur cathode during discharge in MgTFSI2–DME electrolyte. Adv. Mater. 30, 1704313 (2018).
Li, X. et al. Reducing Mg anode overpotential via ion conductive surface layer formation by iodine additive. Adv. Energy Mater. 8, 1701728 (2018).
Liu, J. et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy 4, 180–186 (2019).
Andrews, J. L. et al. Reversible Mg-ion insertion in a metastable one-dimensional polymorph of V2O5. Chem 4, 564–585 (2018).
Arthur, T. S., Singh, N. & Matsui, M. Electrodeposited Bi, Sb and Bi1-xSbx alloys as anodes for Mg-ion batteries. Electrochem. Commun. 16, 103–106 (2012).
Yao, Z., Hegde, V. I., Aspuru-Guzik, A. & Wolverton, C. Discovery of calcium-metal alloy anodes for reversible Ca-ion batteries. Adv. Energy Mater. 9, 1802994 (2019).
Gershinsky, G., Yoo, H. D., Gofer, Y. & Aurbach, D. Electrochemical and spectroscopic analysis of Mg2+ intercalation into thin film electrodes of layered oxides: V2O5 and MoO3. Langmuir 29, 10964–10972 (2013).
Licht, S. Zinc sulfur battery. US patent 6207324 (2001).
Acknowledgements
This work was supported by the U.S. Department of Energy’s Office of Energy Efficiency and Renewable Energy (EERE), as part of the Battery 500 Consortium under Contract DE-EE0008234. We acknowledge Mr. Karun Kumar Rao for the assistance in preparing Fig. 4a–h.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Y.Y. has an equity interest in Polymax Energy Inc. Y.Y.’s relationship with Polymax Energy Inc. has been reviewed and approved by the University of Houston in accordance with its conflict of interest policies.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liang, Y., Dong, H., Aurbach, D. et al. Current status and future directions of multivalent metal-ion batteries. Nat Energy 5, 646–656 (2020). https://doi.org/10.1038/s41560-020-0655-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-020-0655-0
This article is cited by
-
Synthesis via cation replacement reactions
Nature Energy (2024)
-
Rapid-charging aluminium-sulfur batteries operated at 85 °C with a quaternary molten salt electrolyte
Nature Communications (2024)
-
Ca-dimers, solvent layering, and dominant electrochemically active species in Ca(BH4)2 in THF
Nature Communications (2024)
-
A rechargeable Ca/Cl2 battery
Nature Communications (2024)
-
Improving rechargeable magnesium batteries through dual cation co-intercalation strategy
Nature Communications (2024)