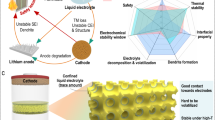
Abstract
Lithium (Li) pulverization and associated large volume expansion during cycling is one of the most critical barriers for the safe operation of Li-metal batteries. Here, we report an approach to minimize the Li pulverization using an electrolyte based on a fluorinated orthoformate solvent. The solid–electrolyte interphase (SEI) formed in this electrolyte clearly exhibits a monolithic feature, which is in sharp contrast with the widely reported mosaic- or multilayer-type SEIs that are not homogeneous and could lead to uneven Li stripping/plating and fast Li and electrolyte depletion over cycling. The highly homogeneous and amorphous SEI not only prevents dendritic Li formation, but also minimizes Li loss and volumetric expansion. Furthermore, this new electrolyte strongly suppresses the phase transformation of the LiNi0.8Co0.1Mn0.1O2 cathode (from layered structure to rock salt) and stabilizes its structure. Tests of high-voltage Li||NMC811 cells show long-term cycling stability and high rate capability, as well as reduced safety concerns.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Whittingham, M. S. Ultimate limits to intercalation reactions for lithium batteries. Chem. Rev. 114, 11414–11443 (2014).
Zhang, J.-G., Xu, W. & Henderson, W. A. Lithium Metal Anode and Rechargeable Li Metal Batteries (Springer International Publishing, 2017).
Liu, J. et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy 4, 180–186 (2019).
Lin, D., Liu, Y. & Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 12, 194–206 (2017).
Wang, J. et al. Superconcentrated electrolytes for a high-voltage lithium-ion battery. Nat. Commun. 7, 12032 (2016).
Erickson, E. M. et al. Review—development of advanced rechargeable batteries: a continuous challenge in the choice of suitable electrolyte solutions. J. Electrochem. Soc. 162, A2424–A2438 (2015).
Cheng, X.-B., Zhang, R., Zhao, C.-Z. & Zhang, Q. Toward safe lithium metal anode in rechargeable batteries: a review. Chem. Rev. 117, 10403–10473 (2017).
Ding, M. S., von Cresce, A. & Xu, K. Conductivity, viscosity, and their correlation of a super-concentrated aqueous electrolyte. J. Phys. Chem. C. 121, 2149–2153 (2017).
Liu, B., Zhang, J.-G. & Xu, W. Advancing lithium metal batteries. Joule 2, 833–845 (2018).
Pang, Q. et al. Tuning the electrolyte network structure to invoke quasi-solid state sulfur conversion and suppress lithium dendrite formation in Li–S batteries. Nat. Energy 3, 783–791 (2018).
Chen, S. et al. High-efficiency lithium metal batteries with fire-retardant electrolytes. Joule 2, 1548–1558 (2018).
Ren, X. et al. Localized high-concentration sulfone electrolytes for high-efficiency lithium-metal batteries. Chem 4, 1877–1892 (2018).
Li, N.-W. et al. A flexible solid electrolyte interphase layer for long-life lithium metal anodes. Angew. Chem. Int. Ed. 57, 1505–1509 (2018).
Fan, X. et al. Non-flammable electrolyte enables Li-metal batteries with aggressive cathode chemistries. Nat. Nanotechnol. 13, 715–722 (2018).
Zeng, Z. et al. Non-flammable electrolytes with high salt-to-solvent ratios for Li-ion and Li-metal batteries. Nat. Energy 3, 674–681 (2018).
Jiao, S. H. et al. Stable cycling of high-voltage lithium metal batteries in ether electrolytes. Nat. Energy 3, 739–746 (2018).
Aurbach, D., Zinigrad, E., Cohen, Y. & Teller, H. A short review of failure mechanisms of lithium metal and lithiated graphite anodes in liquid electrolyte solutions. Solid State Ion. 148, 405–416 (2002).
Peled, E. & Menkin, S. Review—SEI: past, present and future. J. Electrochem. Soc. 164, A1703–A1719 (2017).
Li, Y. et al. Atomic structure of sensitive battery materials and interfaces revealed by cryo–electron microscopy. Science 358, 506–510 (2017).
Xu, K. Electrolytes and interphases in li-ion batteries and beyond. Chem. Rev. 114, 11503–11618 (2014).
Wang, X. et al. New insights on the structure of electrochemically deposited lithium metal and its solid electrolyte interphases via cryogenic TEM. Nano Lett. 17, 7606–7612 (2017).
Noh, H.-J., Youn, S., Yoon, C. S. & Sun, Y.-K. Comparison of the structural and electrochemical properties of layered Li[NixCoyMnz]O2 (x = 1/3, 0.5, 0.6, 0.7, 0.8 and 0.85) cathode material for lithium-ion batteries. J. Power Sources 233, 121–130 (2013).
Yan, G. et al. Fluorinated solvents for high-voltage electrolyte in lithium-ion battery. J. Solid State Electrochem. 21, 1589–1597 (2017).
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4418 (2004).
Cao, X. et al. High voltage LiNi0.5Mn1.5O4/Li4Ti5O12 lithium ion cells at elevated temperatures: carbonate- versus ionic liquid-based electrolytes. ACS Appl. Mater. Interfaces 8, 25971–25978 (2016).
Park, M. S. et al. A highly reversible lithium metal anode. Sci. Rep. 4, 3815 (2014).
Zhang, Z. et al. Fluorinated electrolytes for 5 V lithium-ion battery chemistry. Energy Environ. Sci. 6, 1806–1810 (2013).
Fan, X. et al. Highly fluorinated interphases enable high-voltage Li-metal batteries. Chem 4, 174–185 (2018).
Noguchi, T., Hasegawa, T., Yamauchi, H., Yamazaki, I. & Utsugi, K. Effect of using fluorinated ether and sulfone as electrolyte solvents for lithium ion batteries with lithium-rich layered cathodes and silicon oxide anodes. ECS Trans. 80, 291–303 (2017).
Suo, L. et al. Fluorine-donating electrolytes enable highly reversible 5-V-class Li metal batteries. Proc. Natl Acad. Sci. USA 115, 1156–1161 (2018).
Böttcher, T. et al. in Advanced Fluoride-Based Materials for Energy Conversion (eds Nakajima, T. & Groult, H.) 125–145 (Elsevier, 2015).
Moon, H. et al. Solvent activity in electrolyte solutions controls electrochemical reactions in Li-ion and Li-sulfur batteries. J. Phys. Chem. C 119, 3957–3970 (2015).
Yu, L. et al. A localized high-concentration electrolyte with optimized solvents and lithium difluoro(oxalate)borate additive for stable lithium metal batteries. ACS Energy Lett. 3, 2059–2067 (2018).
Zheng, J. et al. Extremely stable sodium metal batteries enabled by localized high-concentration electrolytes. ACS Energy Lett. 3, 315–321 (2018).
Doi, T., Shimizu, Y., Hashinokuchi, M. & Inaba, M. Dilution of highly concentrated LiBF4/propylene carbonate electrolyte solution with fluoroalkyl ethers for 5-V LiNi0.5Mn1.5O4 positive electrodes. J. Electrochem. Soc. 164, A6412–A6416 (2017).
Chen, S. et al. High-voltage lithium-metal batteries enabled by localized high-concentration electrolytes. Adv. Mater. 30, 1706102 (2018).
Yamada, Y. & Yamada, A. Review—superconcentrated electrolytes for lithium batteries. J. Electrochem. Soc. 162, A2406–A2423 (2015).
Yamada, Y. et al. Unusual stability of acetonitrile-based superconcentrated electrolytes for fast-charging lithium-ion batteries. J. Am. Chem. Soc. 136, 5039–5046 (2014).
Qian, J. et al. High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015).
Ren, X. et al. Enabling high-voltage lithium metal batteries under practical conditions. Joule 3, 1662–1676 (2019).
Von Cresce, A. & Xu, K. Electrolyte additive in support of 5 V Li ion chemistry. J. Electrochem. Soc. 158, A337–A342 (2011).
Tan, S. et al. Tris(hexafluoro-iso-propyl)phosphate as an SEI-forming additive on improving the electrochemical performance of the Li[Li0.2Mn0.56Ni0.16Co0.08]O2 cathode material. J. Electrochem. Soc. 160, A285–A292 (2013).
Von Aspern, N. et al. Phosphorus additives for improving high voltage stability and safety of lithium ion batteries. J. Fluor. Chem. 198, 24–33 (2017).
Ren, X. et al. Localized high-concentration sulfone electrolytes for high-efficiency lithium-metal batteries. Chem 4, 1877–1892 (2018).
Zou, L. et al. Solid–liquid interfacial reaction trigged propagation of phase transition from surface into bulk lattice of Ni-rich layered cathode. Chem. Mater. 30, 7016–7026 (2018).
Wang, C., Meng, Y. S. & Xu, K. Perspective—fluorinating interphases. J. Electrochem. Soc. 166, A5184–A5186 (2019).
Evertz, M. et al. Unraveling transition metal dissolution of Li1.04Ni1/3Co1/3Mn1/3O2 (NCM 111) in lithium ion full cells by using the total reflection X-ray fluorescence technique. J. Power Sources 329, 364–371 (2016).
Cao, X. et al. Ester modified pyrrolidinium based ionic liquids as electrolyte component candidates in rechargeable lithium batteries. Z. Anorg. Allg. Chem. 641, 2536–2542 (2015).
Zhan, C.-G., Nichols, J. A. & Dixon, D. A. Ionization potential, electron affinity, electronegativity, hardness, and electron excitation energy: molecular properties from density functional theory orbital energies. J. Phys. Chem. A 107, 4184–4195 (2003).
Adams, B. D., Zheng, J., Ren, X., Xu, W. & Zhang, J.-G. Accurate determination of coulombic efficiency for lithium metal anodes and lithium metal batteries. Adv. Energy Mater. 8, 1702097 (2018).
Acknowledgements
This work has been supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, Vehicle Technologies Office, of the US Department of Energy (DOE), through the Advanced Battery Materials Research program (Battery500 Consortium) under contract number DE-AC02-05CH11231. The SEM, TEM and XPS were conducted in the William R. Wiley Environmental Molecular Sciences Laboratory—a national scientific user facility sponsored by the DOE’s Office of Biological and Environmental Research and located at the Pacific Northwest National Laboratory. The Pacific Northwest National Laboratory is operated by Battelle for the DOE under the contract DE-AC05-76RL01830. The LiFSI salt was provided by K. Murata of Nippon Shokubai.
Author information
Authors and Affiliations
Contributions
J.-G.Z., W.X. and X.C. proposed the research and designed the experiments. X.C. performed the electrochemical measurements and conducted the SEM observations, with help from X.R. L.Z., B.E.M., B.W.A. and C.W. performed the focused-ion-beam SEM and TEM. M.H.E. performed the XPS measurements. H.W., W.H. and Y.C. carried out the cryo-EM. C.N. and H.L. prepared the NMC811 electrodes. X.C., W.X. and J.-G.Z. prepared the manuscript, with input from all other co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–16
Rights and permissions
About this article
Cite this article
Cao, X., Ren, X., Zou, L. et al. Monolithic solid–electrolyte interphases formed in fluorinated orthoformate-based electrolytes minimize Li depletion and pulverization. Nat Energy 4, 796–805 (2019). https://doi.org/10.1038/s41560-019-0464-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-019-0464-5
This article is cited by
-
Single-phase local-high-concentration solid polymer electrolytes for lithium-metal batteries
Nature Energy (2024)
-
An entanglement association polymer electrolyte for Li-metal batteries
Nature Communications (2024)
-
Controlled large-area lithium deposition to reduce swelling of high-energy lithium metal pouch cells in liquid electrolytes
Nature Energy (2024)
-
Molecular anchoring of free solvents for high-voltage and high-safety lithium metal batteries
Nature Communications (2024)
-
Methylation enables the use of fluorine-free ether electrolytes in high-voltage lithium metal batteries
Nature Chemistry (2024)