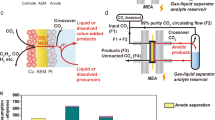
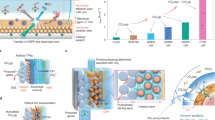
Abstract
Electrocatalytic CO2 reduction is often carried out in a solution electrolyte such as KHCO3(aq), which allows for ion conduction between electrodes. Therefore, liquid products that form are in a mixture with the dissolved salts, requiring energy-intensive downstream separation. Here, we report continuous electrocatalytic conversion of CO2 to pure liquid fuel solutions in cells that utilize solid electrolytes, where electrochemically generated cations (such as H+) and anions (such as HCOO−) are combined to form pure product solutions without mixing with other ions. Using a HCOOH-selective (Faradaic efficiencies > 90%) and easily scaled Bi catalyst at the cathode, we demonstrate production of pure HCOOH solutions with concentrations up to 12 M. We also show 100 h continuous and stable generation of 0.1 M HCOOH with negligible degradation in selectivity and activity. Production of other electrolyte-free C2+ liquid oxygenate solutions, including acetic acid, ethanol and n-propanol, are also demonstrated using a Cu catalyst. Finally, we show that our CO2 reduction cell with solid electrolytes can be modified to suit other, more complex practical applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding authors on reasonable request.
Change history
18 December 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Chu, S., Cui, Y. & Liu, N. The path towards sustainable energy. Nat. Mater. 16, 16 (2017).
Lin, S. et al. Covalent organic frameworks comprising cobalt porphyrins for catalytic CO2 reduction in water. Science 349, 1208–1213 (2015).
Li, F., Chen, L., Knowles, G. P., MacFarlane, D. R. & Zhang, J. Hierarchical mesoporous SnO2 nanosheets on carbon cloth: a robust and flexible electrocatalyst for CO2 reduction with high efficiency and selectivity. Angew. Chem. Int. Ed. 56, 505–509 (2017).
Ripatti, D. S., Veltman, T. R. & Kanan, M. W. Carbon monoxide gas diffusion electrolysis that produces concentrated C2 products with high single-pass conversion. Joule 3, 240–256 (2018).
Kortlever, R., Peters, I., Koper, S. & Koper, M. T. Electrochemical CO2 reduction to formic acid at low overpotential and with high faradaic efficiency on carbon-supported bimetallic Pd-Pt nanoparticles. ACS Catal. 5, 3916–3923 (2015).
Yang, H., Kaczur, J. J., Sajjad, S. D. & Masel, R. I. CO2 conversion to formic acid in a three compartment cell with Sustainion™ membranes. ECS Trans. 77, 1425–1431 (2017).
Klinkova, A. et al. Rational design of efficient palladium catalysts for electroreduction of carbon dioxide to formate. ACS Catal. 6, 8115–8120 (2016).
Lee, W., Kim, Y. E., Youn, M. H., Jeong, S. K. & Park, K. T. Catholyte‐free electrocatalytic CO2 reduction to formate. Angew. Chem. Int. Ed. 57, 6883–6887 (2018).
Ren, D. et al. Selective electrochemical reduction of carbon dioxide to ethylene and ethanol on copper (I) oxide catalysts. ACS Catal. 5, 2814–2821 (2015).
Jiang, K. et al. Metal ion cycling of Cu foil for selective C-C coupling in electrochemical CO2 reduction. Nat. Catal. 1, 111 (2018).
Li, C. W., Ciston, J. & Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 508, 504 (2014).
Ren, D., Wong, N. T., Handoko, A. D., Huang, Y. & Yeo, B. S. Mechanistic insights into the enhanced activity and stability of agglomerated Cu nanocrystals for the electrochemical reduction of carbon dioxide to n-propanol. J. Phys. Chem. Lett. 7, 20–24 (2015).
Kim, D., Kley, C. S., Li, Y. & Yang, P. Copper nanoparticle ensembles for selective electroreduction of CO2 to C2-C3 products. Proc. Natl Acad. Sci. USA 114, 10560–10565 (2017).
Jouny, M., Luc, W. & Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
Bushuyev, O. S. et al. What should we make with CO2 and how can we make it? Joule 2, 825–832 (2018).
He, S. et al. The p‐orbital delocalization of main-group metal boosting CO2 electroreduction. Angew. Chem. Int. Ed. 57, 16114–16119 (2018).
Han, N. et al. Ultrathin bismuth nanosheets from in situ topotactic transformation for selective electrocatalytic CO2 reduction to formate. Nat. Commun. 9, 1320 (2018).
Zheng, X. et al. Sulfur-modulated tin sites enable highly selective electrochemical reduction of CO2 to formate. Joule 1, 794–805 (2017).
Zhang, S., Kang, P. & Meyer, T. J. Nanostructured tin catalysts for selective electrochemical reduction of carbon dioxide to formate. J. Am. Chem. Soc. 136, 1734–1737 (2014).
Zhong, H. et al. Bismuth nanodendrites as a high performance electrocatalyst for selective conversion of CO2 to formate. J. Mater. Chem. A 4, 13746–13753 (2016).
García de Arquer, F. P. et al. 2D metal oxyhalide‐derived catalysts for efficient CO2 electroreduction. Adv. Mater. 30, 1802858 (2018).
Gao, S. et al. Partially oxidized atomic cobalt layers for carbon dioxide electroreduction to liquid fuel. Nature 529, 68 (2016).
Manthiram, A., Yu, X. & Wang, S. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2, 16103 (2017).
Fan, L., Wei, S., Li, S., Li, Q. & Lu, Y. Recent progress of the solid‐state electrolytes for high‐energy metal‐based batteries. Adv. Energy Mater. 8, 1702657 (2018).
Kuehn, C., Leder, F., Jasinski, R. & Gaunt, K. The electrolytic synthesis of hydrogen peroxide in a dual membrane cell. J. Electrochem. Soc. 130, 1117–1119 (1983).
Yamamoto, T., Tryk, D. A., Fujishima, A. & Ohata, H. Production of syngas plus oxygen from CO2 in a gas-diffusion electrode-based electrolytic cell. Electrochim. Acta 47, 3327–3334 (2002).
Dinh, C.-T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Jhong, H. R. M., Brushett, F. R. & Kenis, P. J. The effects of catalyst layer deposition methodology on electrode performance. Adv. Energy Mater. 3, 589–599 (2013).
Verma, S. et al. Insights into the low overpotential electroreduction of CO2 to CO on a supported gold catalyst in an alkaline flow electrolyzer. ACS Energy Lett. 3, 193–198 (2017).
Weekes, D. M., Salvatore, D. A., Reyes, A., Huang, A. & Berlinguette, C. P. Electrolytic CO2 reduction in a flow cell. Acc. Chem. Res. 51, 910–918 (2018).
Zheng, T. et al. Large-scale and highly selective CO2 electrocatalytic reduction on nickel single-atom catalyst. Joule 3, 1–14 (2019).
Jiang, K. et al. Isolated Ni single atoms in graphene nanosheets for high-performance CO2 reduction. Energ. Environ. Sci. 11, 893–903 (2018).
Delacourt, C., Ridgway, P. L., Kerr, J. B. & Newman, J. Design of an electrochemical cell making syngas (CO+H2) from CO2 and H2O reduction at room temperature. J. Electrochem. Soc. 155, B42–B49 (2008).
Natsui, K., Iwakawa, H., Ikemiya, N., Nakata, K. & Einaga, Y. Stable and highly efficient electrochemical production of formic acid from carbon dioxide using diamond electrodes. Angew. Chem. Int. Ed. 130, 2669–2673 (2018).
Min, X. & Kanan, M. W. Pd-catalyzed electrohydrogenation of carbon dioxide to formate: high mass activity at low overpotential and identification of the deactivation pathway. J. Am. Chem. Soc. 137, 4701–4708 (2015).
Lei, F. et al. Metallic tin quantum sheets confined in graphene toward high-efficiency carbon dioxide electroreduction. Nat. Commun. 7, 12697 (2016).
Jiang, B., Zhang, X. G., Jiang, K., Wu, D. Y. & Cai, W. B. Boosting formate production in electrocatalytic CO2 reduction over wide potential window on Pd surfaces. J. Am. Chem. Soc. 140, 2880–2889 (2018).
Hoffman, Z. B., Gray, T. S., Moraveck, K. B., Gunnoe, T. B. & Zangari, G. Electrochemical reduction of carbon dioxide to syngas and formate at dendritic copper-indium electrocatalysts. ACS Catal. 7, 5381–5390 (2017).
Gao, S. et al. Ultrathin Co3O4 layers realizing optimized CO2 electroreduction to formate. Angew. Chem. Int. Ed. 55, 698–702 (2016).
Yang, H. et al. Selective CO2 reduction on 2D mesoporous Bi nanosheets. Adv. Energy Mater. 8, 1801536 (2018).
Zhang, Y. et al. Controllable synthesis of few‐layer bismuth subcarbonate by electrochemical exfoliation for enhanced CO2 reduction performance. Angew. Chem. Int. Ed. 57, 13283–13287 (2018).
Zhou, Y. et al. Monolayered Bi2WO6 nanosheets mimicking heterojunction interface with open surfaces for photocatalysis. Nat. Commun. 6, 8340 (2015).
Henry, N. et al. [Bi2O2]2+ layers in Bi2O2(OH)(NO3): synthesis and structure determination. Z. Naturforsch. 60b, 322–327 (2005).
Zhang, S. et al. Polyethylenimine-enhanced electrocatalytic reduction of CO2 to formate at nitrogen-doped carbon nanomaterials. J. Am. Chem. Soc. 136, 7845–7848 (2014).
Rosen, B. A. et al. Ionic liquid–mediated selective conversion of CO2 to CO at low overpotentials. Science 334, 643–644 (2011).
Jiang, K. et al. Transition-metal single atoms in a graphene shell as active centers for highly efficient artificial photosynthesis. Chem 3, 950–960 (2017).
Lee, Y., Suntivich, J., May, K. J., Perry, E. E. & Shao-Horn, Y. Synthesis and activities of rutile IrO2 and RuO2 nanoparticles for oxygen evolution in acid and alkaline solutions. J. Phys. Chem. Lett. 3, 399–404 (2012).
Jeong, K.-J. et al. Fuel crossover in direct formic acid fuel cells. J. Power Sources 168, 119–125 (2007).
Kuhl, K. P., Cave, E. R., Abram, D. N. & Jaramillo, T. F. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energ. Environ. Sci. 5, 7050–7059 (2012).
Rice, C. et al. Direct formic acid fuel cells. J. Power Sources 111, 83–89 (2002).
Gong, M. et al. An advanced Ni-Fe layered double hydroxide electrocatalyst for water oxidation. J. Am. Chem. Soc. 135, 8452–8455 (2013).
Vermaas, D. A., Sassenburg, M. & Smith, W. A. Photo-assisted water splitting with bipolar membrane induced pH gradients for practical solar fuel devices. J. Mater. Chem. A 3, 19556–19562 (2015).
Masel, R. I., Liu, Z. & Sajjad, S. Anion exchange membrane electrolyzers showing 1 A/cm2 at less than 2 V. ECS Trans. 75, 1143–1146 (2016).
Liu, Z., Yang, H., Kutz, R. & Masel, R. I. CO2 electrolysis to CO and O2 at high selectivity, stability and efficiency using Sustainion membranes. J. Electrochem. Soc. 165, J3371–J3377 (2018).
Liu, Z. et al. The effect of membrane on an alkaline water electrolyzer. Int. J. Hydrogen Energ. 42, 29661–29665 (2017).
Millar, J., Smith, D., Marr, W. & Kressman, T. 33. Solvent-modified polymer networks. Part I. The preparation and characterisation of expanded-network and macroporous styrene–divinylbenzene copolymers and their sulphonates. J. Chem. Soc., 218–225 (1963).
Okuhara, T., Watanabe, H., Nishimura, T., Inumaru, K. & Misono, M. Microstructure of cesium hydrogen salts of 12-tungstophosphoric acid relevant to novel acid catalysis. Chem. Mater. 12, 2230–2238 (2000).
Bard, A. Standard Potentials in Aqueous Solution (Routledge, 2017).
Yoo, J. S., Christensen, R., Vegge, T., Nørskov, J. K. & Studt, F. Theoretical insight into the trends that guide the electrochemical reduction of carbon dioxide to formic acid. ChemSusChem 9, 358–363 (2016).
Acknowledgements
This work was supported by Rice University. This research used the 8-ID (ISS) beamline of the National Synchrotron Light Source II and the Center for Functional Nanomaterials, US Department of Energy Office of Science User Facilities operated for the DOE Office of Science by Brookhaven National Laboratory under contract no. DE-SC0012704. Q.J. and H.N.A. acknowledge the support from King Abdullah University of Science and Technology (KAUST).
Author information
Authors and Affiliations
Contributions
The project was conceptualized by C.X. and H.W., and supervised by H.W.; Bi-catalyst synthesis procedures were developed and performed by C.X.; C.X. and P.Z. conducted the catalytic tests and the related data processing. Materials characterization and analysis was performed by C.X. with the help of Q.J., W.L. and Y.P.; 1H and 13C NMR experiments and analysis were carried out by C.X. and Q.J.; in operando XAS study was performed by C.X. and H.W. with the support of E.S.; ICP-OES was conducted by Q.J.; H.N.A. provided suggestions on the work. C.X. and H.W. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–12, Note 1 and refs 1–2.
Rights and permissions
About this article
Cite this article
Xia, C., Zhu, P., Jiang, Q. et al. Continuous production of pure liquid fuel solutions via electrocatalytic CO2 reduction using solid-electrolyte devices. Nat Energy 4, 776–785 (2019). https://doi.org/10.1038/s41560-019-0451-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-019-0451-x
This article is cited by
-
Coverage enhancement accelerates acidic CO2 electrolysis at ampere-level current with high energy and carbon efficiencies
Nature Communications (2024)
-
Pure-water-fed, electrocatalytic CO2 reduction to ethylene beyond 1,000 h stability at 10 A
Nature Energy (2024)
-
Co-electrolysis of ethylene glycol and carbon dioxide for formate synthesis
Science China Chemistry (2024)
-
Strongly Coupled Ag/Sn–SnO2 Nanosheets Toward CO2 Electroreduction to Pure HCOOH Solutions at Ampere-Level Current
Nano-Micro Letters (2024)
-
Conversion of CO2 to multicarbon products in strong acid by controlling the catalyst microenvironment
Nature Synthesis (2023)