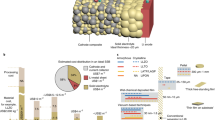
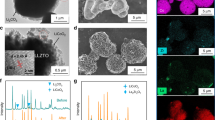
Abstract
A critical parameter for the large-scale integration of solid-state batteries is to establish processing strategies to assemble battery materials at the lowest processing temperature possible while keeping lithium conduction up. Despite extensive research efforts, integrating ceramic film electrolytes while keeping a high lithium concentration and conduction at a low processing temperature remains challenging. Here, we report an alternative ceramic processing strategy through the evolution of multilayers establishing lithium reservoirs directly in lithium–garnet films that allow for lithiated and fast-conducting cubic solid-state battery electrolytes at unusually low processing temperatures. A lithium–garnet film processed via the multilayer processing approach exhibited the fastest ionic conductivity of 2.9 ± 0.05 × 10−5 S cm−1 (at room temperature) and the desired cubic phase, but was stabilized at a processing temperature lowered by 400 °C. This method enables future solid-state battery architectures with more room for cathode volumes by design, and reduces the processing temperature.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Janek, J. & Zeier, W. G. A solid future for battery development. Nat. Energy 1, 16141 (2016).
Thompson, T. et al. Electrochemical window of the Li-ion solid electrolyte Li7La3Zr2O12. ACS Energy Lett. 2, 462–468 (2017).
Thangadurai, V., Narayanan, S. & Pinzaru, D. Garnet-type solid-state fast Li ion conductors for Li batteries: critical review. Chem. Soc. Rev. 43, 4714–4727 (2014).
Murugan, R., Thangadurai, V. & Weppner, W. Fast lithium ion conduction in garnet-type Li7La3Zr2O12. Angew. Chem. Int. Ed. 46, 7778–7781 (2007).
Kamaya, N. et al. A lithium superionic conductor. Nat. Mater. 10, 682–686 (2011).
Kato, Y. et al. High-power all-solid-state batteries using sulfide superionic conductors. Nat. Energy 1, 16030 (2016).
Kanno, R. & Murayama, M. Lithium ionic conductor thio-LISICON: the Li2S GeS2 P2S5 system. J. Electrochem. Soc. 148, A742–A746 (2001).
Park, K. H. et al. Design strategies, practical considerations, and new solution processes of sulfide solid electrolytes for all-solid-state batteries. Adv. Energy Mater. 8, 1800035 (2018).
Yamane, H. et al. Crystal structure of a superionic conductor, Li7P3S11. Solid State Ion. 178, 1163–1167 (2007).
Mizuno, F., Hayashi, A., Tadanaga, K. & Tatsumisago, M. New, highly ion-conductive crystals precipitated from Li2S–P2S5 glasses. Adv. Mater. 17, 918–921 (2005).
Seino, Y., Ota, T., Takada, K., Hayashi, A. & Tatsumisago, M. A sulphide lithium super ion conductor is superior to liquid ion conductors for use in rechargeable batteries. Energy Environ. Sci. 7, 627–631 (2014).
Richards, W. D., Miara, L. J., Wang, Y., Kim, J. C. & Ceder, G. Interface stability in solid-state batteries. Chem. Mater. 28, 266–273 (2016).
Porz, L. et al. Mechanism of lithium metal penetration through inorganic solid electrolytes. Adv. Energy Mater. 7, 1701003 (2017).
Ma, C. et al. Interfacial stability of Li metal–solid electrolyte elucidated via in situ electron microscopy. Nano Lett. 16, 7030–7036 (2016).
Awaka, J. et al. Crystal structure of fast lithium-ion-conducting cubic Li7La3Zr2O12. Chem. Lett. 40, 60–62 (2011).
Wagner, R. et al. Crystal structure of garnet-related Li-ion conductor Li7–3xGaxLa3Zr2O12: fast Li-ion conduction caused by a different cubic modification? Chem. Mater. 28, 1861–1871 (2016).
Wagner, R. et al. Fast Li-ion-conducting garnet-related Li7–3xFexLa3Zr2O12 with uncommon I4̅3d structure. Chem. Mater. 28, 5943–5951 (2016).
Bernuy-Lopez, C. et al. Atmosphere controlled processing of Ga-substituted garnets for high Li-ion conductivity ceramics. Chem. Mater. 26, 3610–3617 (2014).
Wolfenstine, J., Ratchford, J., Rangasamy, E., Sakamoto, J. & Allen, J. L. Synthesis and high Li-ion conductivity of Ga-stabilized cubic Li7La3Zr2O12. Mater. Chem. Phys. 134, 571–575 (2012).
Rettenwander, D. et al. Crystal chemistry of “Li7La3Zr2O12” garnet doped with Al, Ga, and Fe: a short review on local structures as revealed by NMR and Mößbauer spectroscopy studies. Eur. J. Mineral. 28, 619–629 (2016).
Rettenwander, D. et al. Structural and electrochemical consequences of Al and Ga cosubstitution in Li7La3Zr2O12 solid electrolytes. Chem. Mater. 28, 2384–2392 (2016).
Howard, M. A. et al. Synthesis, conductivity and structural aspects of Nd3Zr2Li7−3xAlxO12. J. Mater. Chem. A 1, 14013–14022 (2013).
Dumon, A., Huang, M., Shen, Y. & Nan, C.-W. High Li ion conductivity in strontium doped Li7La3Zr2O12 garnet. Solid State Ion. 243, 36–41 (2013).
Zeier, W. G., Zhou, S., Lopez-Bermudez, B., Page, K. & Melot, B. C. Dependence of the Li-ion conductivity and activation energies on the crystal structure and ionic radii in Li6MLa2Ta2O12. ACS Appl. Mater. Interfaces 6, 10900–10907 (2014).
Li, Y., Han, J.-T., Wang, C.-A., Xie, H. & Goodenough, J. B. Optimizing Li+ conductivity in a garnet framework. J. Mater. Chem. 22, 15357–15361 (2012).
Rettenwander, D. et al. Synthesis, crystal chemistry, and electrochemical properties of Li7–2xLa3Zr2–xMoxO12 (x = 0.1–0.4): stabilization of the cubic garnet polymorph via substitution of Zr4+ by Mo6+. Inorg. Chem. 54, 10440–10449 (2015).
Allen, J. L., Wolfenstine, J., Rangasamy, E. & Sakamoto, J. Effect of substitution (Ta, Al, Ga) on the conductivity of Li7La3Zr2O12. J. Power Sources 206, 315–319 (2012).
Yi, E., Wang, W., Kieffer, J. & Laine, R. M. Flame made nanoparticles permit processing of dense, flexible, Li+ conducting ceramic electrolyte thin films of cubic-Li7La3Zr2O12 (c-LLZO). J. Mater. Chem. A 4, 12947–12954 (2016).
Fu, K. Toward garnet electrolyte-based Li metal batteries: an ultrathin, highly effective, artificial solid-state electrolyte/metallic Li interface. Sci. Adv. 3, e1601659 (2017).
Fu, K. Three-dimensional bilayer garnet solid electrolyte based high energy density lithium metal–sulfur batteries. Energy Environ. Sci. 10, 1568–1575 (2017).
Miara, L. et al. About the compatibility between high voltage spinel cathode materials and solid oxide electrolytes as a function of temperature. ACS Appl. Mater. Interfaces 8, 26842–26850 (2016).
Hirayama, M., Kim, K., Toujigamori, T., Cho, W. & Kanno, R. Epitaxial growth and electrochemical properties of Li4Ti5O12 thin-film lithium battery anodes. Dalton Trans. 40, 2882–2887 (2011).
Lu, Z. G., Cheng, H., Lo, M. F. & Chung, C. Y. Pulsed laser deposition and electrochemical characterization of LiFePO4–Ag composite thin films. Adv. Funct. Mater. 17, 3885–3896 (2007).
Lacivita, V. et al. Resolving the amorphous structure of lithium phosphorus oxynitride (LiPON). J. Am. Chem. Soc. 140, 11029–11038 (2018).
Yu, X., Bates, J. B., Jellison, G. E. & Hart, F. X. A stable thin‐film lithium electrolyte: lithium phosphorus oxynitride. J. Electrochem. Soc. 144, 524–532 (1997).
Zhu, Y., He, X. & Mo, Y. Origin of outstanding stability in the lithium solid electrolyte materials: insights from thermodynamic analyses based on first-principles calculations. ACS Appl. Mater. Interfaces 7, 23685–23693 (2015).
Oudenhoven, J. F. M., Baggetto, L. & Notten, P. H. L. All-solid-state lithium-ion microbatteries: a review of various three-dimensional concepts. Adv. Energy Mater. 1, 10–33 (2011).
Bates, J. B., Dudney, N. J., Neudecker, B., Ueda, A. & Evans, C. D. Thin-film lithium and lithium-ion batteries. Solid State Ion. 135, 33–45 (2000).
Li, J., Ma, C., Chi, M., Liang, C. & Dudney, N. J. Solid electrolyte: the key for high-voltage lithium batteries. Adv. Energy Mater. 5, 1401408 (2015).
Patil, A. et al. Issue and challenges facing rechargeable thin film lithium batteries. Mater. Res. Bull. 43, 1913–1942 (2008).
Han, X. et al. Negating interfacial impedance in garnet-based solid-state Li metal batteries. Nat. Mater. 16, 572–579 (2017).
Schwöbel, A., Hausbrand, R. & Jaegermann, W. Interface reactions between LiPON and lithium studied by in-situ X-ray photoemission. Solid State Ion. 273, 51–54 (2015).
Schichtel, P. et al. On the impedance and phase transition of thin film all-solid-state batteries based on the Li4Ti5O12 system. J. Power Sources 360, 593–604 (2017).
Dai, J., Yang, C., Wang, C., Pastel, G. & Hu, L. Interface engineering for garnet-based solid-state lithium-metal batteries: materials, structures, and characterization. Adv. Mater. 30, 1802068 (2018).
Rawlence, M. et al. Effect of gallium substitution on lithium-ion conductivity and phase evolution in sputtered Li7–3xGaxLa3Zr2O12 thin films. ACS Appl. Mater. Interfaces 10, 13720–13728 (2018).
Kalita, D. J., Lee, S. H., Lee, K. S., Ko, D. H. & Yoon, Y. S. Ionic conductivity properties of amorphous Li–La–Zr–O solid electrolyte for thin film batteries. Solid State Ion. 229, 14–19 (2012).
Nong, J., Xu, H., Yu, Z., Zhu, G. & Yu, A. Properties and preparation of Li–La–Ti–Zr–O thin film electrolyte. Mater. Lett. 154, 167–169 (2015).
Tan, J. & Tiwari, A. Fabrication and characterization of Li7La3Zr2O12 thin films for lithium ion battery. ECS Solid State Lett. 1, Q57–Q60 (2012).
Kim, S., Hirayama, M., Taminato, S. & Kanno, R. Epitaxial growth and lithium ion conductivity of lithium-oxide garnet for an all solid-state battery electrolyte. Dalton Trans. 42, 13112–13117 (2013).
Garbayo, I. et al. Glass-type polyamorphism in Li–garnet thin film solid state battery conductors. Adv. Energy Mater. 8, 1702265 (2018).
Park, J. S. et al. Effects of crystallinity and impurities on the electrical conductivity of Li–La–Zr–O thin films. Thin Solid Films 576, 55–60 (2015).
Katsui, H. & Goto, T. Preparation of cubic and tetragonal Li7La3Zr2O12 film by metal organic chemical vapor deposition. Thin Solid Films 584, 130–134 (2015).
Tadanaga, K. et al. Preparation of lithium ion conductive Al-doped Li7La3Zr2O12 thin films by a sol–gel process. J. Power Sources 273, 844–847 (2015).
Bitzer, M., Van Gestel, T., Uhlenbruck, S. & Buchkremer, H.-P. Sol–gel synthesis of thin solid Li7La3Zr2O12 electrolyte films for Li-ion batteries. Thin Solid Films 615, 128–134 (2016).
Chen, R.-J. et al. Sol–gel derived Li–La–Zr–O thin films as solid electrolytes for lithium-ion batteries. J. Mater. Chem. A 2, 13277–13282 (2014).
Rawlence, M., Garbayo, I., Buecheler, S. & Rupp, J. L. M. On the chemical stability of post-lithiated garnet Al-stabilized Li7La3Zr2O12 solid state electrolyte thin films. Nanoscale 8, 14746–14753 (2016).
El Shinawi, H. & Janek, J. Stabilization of cubic lithium-stuffed garnets of the type “Li7La3Zr2O12” by addition of gallium. J. Power Sources 225, 13–19 (2013).
Buschmann, H. et al. Structure and dynamics of the fast lithium ion conductor “Li7La3Zr2O12”. Phys. Chem. Chem. Phys. 13, 19378–19392 (2011).
Rettenwander, D. et al. Site occupation of Ga and Al in stabilized cubic Li7–3(x+y)GaxAlyLa3Zr2O12 garnets as deduced from 27Al and 71Ga MAS NMR at ultrahigh magnetic fields. Chem. Mater. 27, 3135–3142 (2015).
Rettenwander, D., Geiger, C. A., Tribus, M., Tropper, P. & Amthauer, G. A synthesis and crystal chemical study of the fast ion conductor Li7–3xGaxLa3Zr2O12 with x = 0.08 to 0.84. Inorg. Chem. 53, 6264–6269 (2014).
Rettenwander, D. et al. DFT study of the role of Al3+ in the fast ion-conductor Li7–3xAl3+ xLa3Zr2O12 garnet. Chem. Mater. 26, 2617–2623 (2014).
Wachter-Welzl, A. et al. The origin of conductivity variations in Al-stabilized Li7La3Zr2O12 ceramics. Solid State Ion. 319, 203–208 (2018).
Chandrasekhar, H. R., Bhattacharya, G., Migoni, R. & Bilz, H. Infrared and Raman spectra and lattice dynamics of the superionic conductor Li3N. Phys. Rev. B 17, 884–893 (1978).
Tietz, F., Wegener, T., Gerhards, M. T., Giarola, M. & Mariotto, G. Synthesis and Raman micro-spectroscopy investigation of Li7La3Zr2O12. Solid State Ion. 230, 77–82 (2013).
Larraz, G., Orera, A. & Sanjuán, M. L. Cubic phases of garnet-type Li7La3Zr2O12: the role of hydration. J. Mater. Chem. A 1, 11419–11428 (2013).
Kong, L. et al. A novel chemical route to prepare La2Zr2O7 pyrochlore. J. Am. Ceram. Soc. 96, 935–941 (2013).
Xie, H., Alonso, J. A., Li, Y., Fernández-Díaz, M. T. & Goodenough, J. B. Lithium distribution in aluminum-free cubic Li7La3Zr2O12. Chem. Mater. 23, 3587–3589 (2011).
Cheng, L. et al. Interrelationships among grain size, surface composition, air stability, and interfacial resistance of Al-substituted Li7La3Zr2O12 solid electrolytes. ACS Appl. Mater. Interfaces 7, 17649–17655 (2015).
Kubicek, M. et al. Oxygen vacancies in fast lithium-ion conducting garnets. Chem. Mater. 29, 7189–7196 (2017).
Van den Broek, J., Afyon, S. & Rupp, J. L. M. Interface-engineered all-solid-state Li-ion batteries based on garnet-type fast Li+ conductors. Adv. Energy Mater. 6, 1600736 (2016).
Matsuda, Y. et al. Phase formation of a garnet-type lithium-ion conductor Li7−3xAlxLa3Zr2O12. Solid State Ion. 277, 23–29 (2015).
Struzik, M., Garbayo, I., Pfenninger, R. & Rupp, J. L. M. A simple and fast electrochemical CO2 sensor based on Li7La3Zr2O12 for environmental monitoring. Adv. Mater. 30, 1804098 (2018).
Zhang, Y. et al. Effect of lithium ion concentration on the microstructure evolution and its association with the ionic conductivity of cubic garnet-type nominal Li7Al0.25La3Zr2O12 solid electrolytes. Solid State Ion. 284, 53–60 (2016).
Shin, D. O. et al. Synergistic multi-doping effects on the Li7La3Zr2O12 solid electrolyte for fast lithium ion conduction. Sci. Rep. 5, 18053 (2015).
Djenadic, R. et al. Nebulized spray pyrolysis of Al-doped Li7La3Zr2O12 solid electrolyte for battery applications. Solid State Ion. 263, 49–56 (2014).
Jonson, R. A. & McGinn, P. J. Tape casting and sintering of Li7La3Zr1.75Nb0.25Al0.1O12 with Li3BO3 additions. Solid State Ion. 323, 49–55 (2018).
Yi, E., Wang, W., Kieffer, J. & Laine, R. M. Key parameters governing the densification of cubic-Li7La3Zr2O12 Li+ conductors. J. Power Sources 352, 156–164 (2017).
Bates, J. B. et al. Fabrication and characterization of amorphous lithium electrolyte thin films and rechargeable thin-film batteries. J. Power Sources 43, 103–110 (1993).
Ohta, S., Kobayashi, T. & Asaoka, T. High lithium ionic conductivity in the garnet-type oxide Li7−XLa3(Zr2−X, NbX)O12 (X = 0−2). J. Power Sources 196, 3342–3345 (2011).
Acknowledgements
The authors thank the Competence Center Energy and Mobility and Swiss Electrics for funding the project ‘All solid state Li-ion batteries based on new ceramic Li-ion electrolytes’ (proposal 911). J.L.M.R. thanks Lincoln Laboratory project ACC 697 (2018) and the Thomas Lord Foundation for financial support.
Author information
Authors and Affiliations
Contributions
R.P., M.S., I.G. and E.S. carried out the experiments. R.P., M.S., I.G., E.S. and J.L.M.R. performed the analysis and discussed the data. R.P. and J.L.M.R. wrote the manuscript with help from all of the co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary notes 1 and 2, Supplementary Figs. 1–7, Supplementary discussion, Supplementary references
Rights and permissions
About this article
Cite this article
Pfenninger, R., Struzik, M., Garbayo, I. et al. A low ride on processing temperature for fast lithium conduction in garnet solid-state battery films. Nat Energy 4, 475–483 (2019). https://doi.org/10.1038/s41560-019-0384-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-019-0384-4
This article is cited by
-
Boosting the interfacial superionic conduction of halide solid electrolytes for all-solid-state batteries
Nature Communications (2023)
-
Stress and Manufacturability in Solid-State Lithium-Ion Batteries
International Journal of Precision Engineering and Manufacturing-Green Technology (2023)
-
Printed Solid-State Batteries
Electrochemical Energy Reviews (2023)
-
Photo-enhanced ionic conductivity across grain boundaries in polycrystalline ceramics
Nature Materials (2022)
-
Digestion processes and elemental analysis of oxide and sulfide solid electrolytes
Ionics (2022)