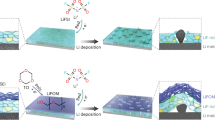
Abstract
Practical lithium metal batteries require full and reversible utilization of thin metallic Li anodes. This introduces a fundamental challenge concerning how to create solid-electrolyte interphases (SEIs) that are able to regulate interfacial transport and protect the reactive metal, without adding appreciably to the cell mass. Here, we report on physicochemical characteristics of Langmuir–Blodgett artificial SEIs (LBASEIs) created using phosphate-functionalized reduced graphene oxides. We find that LBASEIs not only meet the challenges of stabilizing the Li anode, but can be facilely assembled in a simple, scalable process. The LBASEI derives its effectiveness primarily from its ability to form a durable coating on Li that regulates electromigration at the anode/electrolyte interface. In a first step towards practical cells in which the anode and cathode capacities are matched, we report that it is possible to achieve stable operations in both coin and pouch cells composed of a thin Li anode with the LBASEI and a high-loading intercalation cathode.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding authors upon reasonable request.
References
Albertus, P., Babinec, S., Litzelman, S. & Newman, A. Status and challenges in enabling the lithium metal electrode for high-energy and low-cost rechargeable batteries. Nat. Energy 3, 16–21 (2018).
Cheng, X. B., Zhang, R., Zhao, C. Z. & Zhang, Q. Toward safe lithium metal anode in rechargeable batteries: a review. Chem. Rev. 117, 10403–10473 (2017).
Larcher, D. & Tarascon, J.-M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 7, 19–29 (2015).
Armand, M. & Tarascon, J.-M. Building better batteries. Nature 451, 652–657 (2008).
Goodenough, J. B. & Kim, Y. Challenges for rechargeable Li batteries. Chem. Mater. 22, 587–603 (2010).
Wei, S., Choudhury, S., Tu, Z., Zhang, K. & Archer, L. A. Electrochemical interphases for high-energy storage using reactive metal anodes. Acc. Chem. Res. 51, 80–88 (2018).
Liang, X. et al. A facile surface chemistry route to a stabilized lithium metal anode. Nat. Energy 6, 17119 (2017).
Tu, Z. et al. Fast ion transport at solid–solid interfaces in hybrid battery anodes. Nat. Energy 3, 310–316 (2018).
Choudhury, S. et al. Designer interphases for the lithium-oxygen electrochemical cell. Sci. Adv. 3, 1602809 (2017).
Lin, D., Liu, Y. & Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotech. 12, 194–206 (2017).
Tikekar, M. D., Choudhury, S., Tu, Z. & Archer, L. A. Design principles for electrolytes and interfaces for stable lithium-metal batteries. Nat. Energy 1, 1–7 (2016).
Tikekar, M. D., Archer, L. A. & Koch, D. L. Stabilizing electrodeposition in elastic solid electrolytes containing immobilized anions. Sci. Adv. 2, 1600320 (2016).
Tu, Z. et al. Designing artificial solid-electrolyte interphases for single-ion and high-efficiency transport in batteries. Joule 1, 394–406 (2017).
Guo, Y., Li, H. & Zhai, T. Reviving lithium-metal anodes for next-generation high-energy batteries. Adv. Mater. 29, 1–25 (2017).
Kim, M. S. et al. Designing solid-electrolyte interphases for lithium sulfur electrodes using ionic shields. Nano Energy 41, 573–582 (2017).
Lin, D. et al. Layered reduced graphene oxide with nanoscale interlayer gaps as a stable host for lithium metal anodes. Nat. Nanotech. 11, 626–632 (2016).
Liu, S. et al. Crumpled graphene balls stabilized dendrite-free lithium metal anodes. Joule 2, 184–193 (2018).
Liu, L. et al. Free-standing hollow carbon fibers as high-capacity containers for stable lithium metal anodes. Joule 1, 563–575 (2017).
Yan, K. et al. Selective deposition and stable encapsulation of lithium through heterogeneous seeded growth. Nat. Energy 1, 16010 (2016).
Liu, L. et al. Uniform lithium nucleation/growth induced by lightweight nitrogen-doped graphitic carbon foams for high-performance lithium metal anodes. Adv. Mater. 30, 1706216 (2018).
Deng, W., Zhou, X., Fang, Q. & Liu, Z. Microscale lithium metal stored inside cellular graphene scaffold toward advanced metallic lithium anodes. Adv. Energy Mater. 8, 1703152 (2018).
Zhang, R. et al. Lithiophilic sites in doped graphene guide uniform lithium nucleation for dendrite-free lithium metal anodes. Angew. Chem. Int. Ed. 56, 7764–7768 (2017).
Raji, A. O. et al. Lithium batteries with nearly maximum metal storage. ACS Nano 11, 6362–6369 (2017).
Lin, D. et al. Layered reduced graphene oxide with nanoscale interlayer gaps as a stable host for lithium metal anodes. Nat Nano 11, 626–632 (2016).
Liang, Z. et al. Composite lithium metal anode by melt infusion of lithium into a 3D conducting scaffold with lithiophilic coating. Proc. Natl Acad. Sci. USA 113, 2862–2867 (2016).
Liu, Y. et al. Lithium-coated polymeric matrix as a minimum volume-change and dendrite-free lithium metal anode. Nat. Commun. 7, 10992 (2016).
Zhi, J., Zehtab Yazdi, A., Valappil, G., Haime, J. & Chen, P. Artificial solid electrolyte interphase for aqueous lithium energy storage systems. Sci. Adv. 3, 1701010 (2017).
Kim, M. S., Choudhury, S., Moganty, S. S., Wei, S. & Archer, L. A. Fabricating multifunctional nanoparticle membranes by a fast layer-by-layer Langmuir–Blodgett process: application in lithium–sulfur batteries. J. Mater. Chem. A 4, 14709–14719 (2016).
Kim, M. S., Ma, L., Choudhury, S. & Archer, L. A. Multifunctional separator coatings for high-performance lithium–sulfur batteries. Adv. Mater. Interfaces 3, 1600450 (2016).
Kresse, G. & Furthmu, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Adams, B. D., Zheng, J., Ren, X., Xu, W. & Zhang, J. G. Accurate determination of coulombic efficiency for lithium metal anodes and lithium metal batteries. Adv. Energy Mater. 8, 1702097 (2017).
Zheng, J. et al. Electrolyte additive enabled fast charging and stable cycling lithium metal batteries. Nat. Energy 2, 17012 (2017).
Qian, J. et al. Anode-free rechargeable lithium metal batteries. Adv. Funct. Mater. 26, 7094–7102 (2016).
Jiao, S. et al. Behavior of lithium metal anodes under various capacity utilization and high current density in lithium metal batteries. Joule 2, 110–124 (2018).
Suo, L. et al. Fluorine-donating electrolytes enable highly reversible 5-V-class Li metal batteries. Proc. Natl Acad. Sci. USA 115, 1156–1161 (2018).
Fan, X. et al. Non-flammable electrolyte enables Li-metal batteries with aggressive cathode chemistries. Nat. Nanotech. 13, 715–722 (2018).
Li, X. et al. Dendrite-free and performance-enhanced lithium metal batteries through optimizing solvent compositions and adding combinational additives. Adv. Energy Mater. 8, 1703022 (2018).
Kovtyukhova, N. I. et al. Layer-by-layer assembly of ultrathin composite films from micron-sized graphite oxide sheets and polycations. Chem. Mater. 11, 771–778 (1999).
Hummers, W. S. & Offeman, R. E. Preparation of graphitic oxide. J. Am. Chem. Soc. 80, 1339–1339 (1958).
Kresse, G. & Hafner, J. Norm-conserving and ultrasoft pseudopotentials for first-row and transition elements. J. Phys. Condens. Matter. 6, 8245–8257 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Perdew, J. P. & Wang, Y. Accurate and simple analytic representation of the electron-gas correlation energy. Phys. Rev. B 45, 244–249 (1992).
Deepika, Kumar, S., Shukla, A. & Kumar, R. Origin of multiple band gap values in single width nanoribbons. Sci. Rep. 6, 36168 (2016).
Deepika, Kumar, T. J. D., Shukla, A. & Kumar, R. Edge configurational effect on band gaps in graphene nanoribbons. Phys. Rev. B 91, 1–5 (2015).
Pack, J. D. & Monkhorst, H. J. Special points for Brillouin-zone integrations. Phys. Rev. B 16, 1748–1749 (1977).
Klimeš, J., Bowler, D. R. & Michaelides, A. Chemical accuracy for the van der Waals density functional. J. Phys. Condens. Matter. 22, 246401 (2010).
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF-2016M1B3A1A01937324) and the Korea Institute of Science and Technology (KIST) Institutional Program (Project No. 2E28141). L.A.A. also acknowledges support from the US Advanced Research Projects Agency — Energy (ARPA-E) through award #DE-AR0000750. D. thanks Virtual lab www.vfab.org for the Cloud Computing Interface and the KIST supercomputing facility.
Author information
Authors and Affiliations
Contributions
M.S.K., L.A.A. and W.I.C. designed and conceptualized the study. J.-H.R. prepared Langmuir–Blodgett artificial SEIs on specified substrates and provided technical support. D. performed density functional theory calculations for the Li atom binding energies and the charge density analysis on the specified species and atomic structures. Y.R.L. and I.W.N. prepared graphene oxides and helped with XPS experiments. K.-R.L supervised the computational study. W.I.C and L.A.A supervised the overall study. M.S.K performed all the experiments, characterization and analysis and wrote the manuscript. All the authors discussed the manuscript and provided comments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Notes 1–6, Supplementary Figures 1–11, Supplementary References, Supplementary Videos 1–2
Video 1
Demonstration of Langmuir Blodgett Scooping as a scalable, continuous process for fabricating artificial solid electrolyte interphases (ASEI) on a metallic substrate.
Video 2
Illustration of the roll-coating method used to transfer LBASEI from a copper substrate onto a metallic Lithium electrode.
Rights and permissions
About this article
Cite this article
Kim, M.S., Ryu, JH., Deepika et al. Langmuir–Blodgett artificial solid-electrolyte interphases for practical lithium metal batteries. Nat Energy 3, 889–898 (2018). https://doi.org/10.1038/s41560-018-0237-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-018-0237-6
This article is cited by
-
Tuning the solvation structure with salts for stable sodium-metal batteries
Nature Energy (2024)
-
Interfacial self-healing polymer electrolytes for long-cycle solid-state lithium-sulfur batteries
Nature Communications (2024)
-
Lithium-induced graphene layer containing Li3P alloy phase to achieve ultra-stable electrode interface for lithium metal anode
Rare Metals (2024)
-
Investigating microstructure evolution of lithium metal during plating and stripping via operando X-ray tomographic microscopy
Nature Communications (2023)
-
Growing single-crystalline seeds on lithiophobic substrates to enable fast-charging lithium-metal batteries
Nature Energy (2023)