Abstract
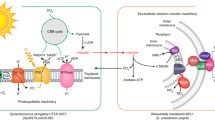
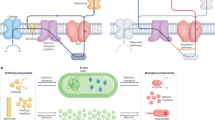
Biophotovoltaic devices (BPVs), which use photosynthetic organisms as active materials to harvest light, have a range of attractive features relative to synthetic and non-biological photovoltaics, including their environmentally friendly nature and ability to self-repair. However, efficiencies of BPVs are currently lower than those of synthetic analogues. Here, we demonstrate BPVs delivering anodic power densities of over 0.5 W m−2, a value five times that for previously described BPVs. We achieved this through the use of cyanobacterial mutants with increased electron export characteristics together with a microscale flow-based design that allowed independent optimization of the charging and power delivery processes, as well as membrane-free operation by exploiting laminar flow to separate the catholyte and anolyte streams. These results suggest that miniaturization of active elements and flow control for decoupled operation and independent optimization of the core processes involved in BPV design are effective strategies for enhancing power output and thus the potential of BPVs as viable systems for sustainable energy generation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Solar Energy Perspectives (International Energy Agency, 2011).
BP Statistical Review of World Energy (British Petroleum, 2015).
Green, M. A. Commercial progress and challenges for photovoltaics. Nat. Energy 1, 15015 (2016).
Crabtee, G. W. & Lewis, N. S. Solar energy conversion. Phys. Today 60, 37–42 (2007).
Lewis, N. S. & Nocera, D. G. Powering the planet: chemical challenges in solar energy utilization. Proc. Natl. Acad. Sci. USA 103, 15729–15735 (2006).
Tao, C. S., Jiang, J. & Tao, M. Natural resource limitations to terawatt-scale solar cells. Sol. Energy Mater. Sol. Cells 95, 3176–3180 (2011).
Peter, L. M. Towards sustainable photovoltaics: the search for new materials. Philos. Trans. A Math. Phys. Eng. Sci. 369, 1840–1856 (2011).
Mazzio, K. A. & Luscombe, C. K. The future of organic photovoltaics. Chem. Soc. Rev. 44, 78–90 (2014).
McCormick, A. J. et al. Biophotovoltaics: oxygenic photosynthetic organisms in the world of bioelectrochemical systems. Energy Environ. Sci. 8, 1092–1109 (2015).
Hasan, K. et al. Photo-electrochemical communication between cyanobacteria (Leptolyngbia sp.) and osmium redox polymer modified electrodes. Phys. Chem. Chem. Phys. 16, 24676–24680 (2014).
Hasan, K. et al. Photoelectrochemical wiring of Paulschulzia pseudovolvox (algae) to osmium polymer modified electrodes for harnessing solar energy. Adv. Energy Mater. 5 (2015).
McCormick, A. J. et al. Photosynthetic biofilms in pure culture harness solar energy in a mediatorless bio-photovoltaic cell (BPV) system. Energy Environ. Sci. 4, 4699–4709 (2011).
Hambourger, M. et al. Biology and technology for photochemical fuel production. Chem. Soc. Rev. 38, 25–35 (2009).
Tanaka, K., Tamamushi, R. & Ogawa, T. Bioelectrochemical fuel-cells operated by the cyanobacterium, Anabaena variabilis. J. Chem. Technol. Biotechnol. 35, 191–197 (1985).
Zou, Y., Pisciotta, J., Billmyre, R. B. & Baskakov, I. V. Photosynthetic microbial fuel cells with positive light response. Biotechnol. Bioeng. 104, 939–946 (2009).
Bradley, R. W., Bombelli, P., Rowden, S. J. & Howe, C. J. Biological photovoltaics: intra- and extra-cellular electron transport by cyanobacteria. Biochem. Soc. Trans. 40, 1302–1307 (2012).
Bombelli, P. et al. Quantitative analysis of the factors limiting solar power transduction by Synechocystis sp. PCC 6803 in biological photovoltaic devices. Energy Environ. Sci. 4, 4690–4698 (2011).
Squires, T. M. & Quake, S. R. Microfluidics: fluid physics at the nanoliter scale. Rev. Mod. Phys. 77, 977 (2005).
Ferrigno, R., Stroock, A. D., Clark, T. D., Mayer, M. & Whitesides, G. M. Membraneless vanadium redox fuel cell using laminar flow. J. Am. Chem. Soc. 124, 12930–12931 (2002).
Wang, H.-Y., Bernarda, A., Huang, C.-Y., Lee, D.-J. & Chang, J.-S. Micro-sized microbial fuel cell: a mini-review. Bioresour. Technol. 102, 235–243 (2011).
Yang, J., Ghobadian, S., Goodrich, P. J., Montazami, R. & Hashemi, N. Miniaturized biological and electrochemical fuel cells: challenges and applications. Phys. Chem. Chem. Phys. 15, 14147–14161 (2013).
Kjeang, E. et al. High-performance microfluidic vanadium redox fuel cell. Electrochim. Acta 52, 4942–4946 (2007).
Ren, H., Torres, C. I., Parameswaran, P., Rittmann, B. E. & Chae, J. Improved current and power density with a micro-scale microbial fuel cell due to a small characteristic length. Biosens. Bioelectron. 61, 587–592 (2014).
Lea-Smith, D. J., Vasudevan, R. & Howe, C. J. Generation of marked and markerless mutants in model cyanobacterial species. J. Vis. Exp., e54001 (2016).
Zhang, P. et al. Operon flv4-flv2 provides cyanobacterial photosystem II with flexibility of electron transfer. Plant. Cell. 24, 1952–1971 (2012).
Brody, J. P. & Yager, P. Diffusion-based extraction in a microfabricated device. Sens. Actuators A Phys. 58, 13–18 (1997).
Kamholz, A. E., Weigl, B. H., Finlayson, B. A. & Yager, P. Quantitative analysis of molecular interaction in a microfluidic channel: the T-sensor. Anal. Chem. 71, 5340–5347 (1999).
Lea-Smith, D. J. et al. Thylakoid terminal oxidases are essential for the cyanobacterium Synechocystis sp. PCC 6803 to survive rapidly changing light intensities. Plant. Physiol. 162, 484–495 (2013).
Zhao, F., Slade, R. C. & Varcoe, J. R. Techniques for the study and development of microbial fuel cells: an electrochemical perspective. Chem. Soc. Rev. 38, 1926–1939 (2009).
Logan, B. E. et al. Microbial fuel cells: methodology and technology. Environ. Sci. Technol. 40, 5181–5192 (2006).
Qian, F., Baum, M., Gu, Q. & Morse, D. E. A 1.5 μl microbial fuel cell for on-chip bioelectricity generation. Lab. Chip 9, 3076–3081 (2009).
Bombelli, P., Müller, T., Herling, T. W., Howe, C. J. & Knowles, T. P. A high power-density, mediator-free, microfluidic biophotovoltaic device for cyanobacterial cells. Adv. Energy Mater. 5, e1401299 (2015).
Kjeang, E., Djilali, N. & Sinton, D. Microfluidic fuel cells: a review. J. Power Sources 186, 353–369 (2009).
Shaegh, S. A. M., Nguyen, N.-T. & Chan, S. H. A review on membraneless laminar flow-based fuel cells. Int. J. Hydrog. Energy 36, 5675–5694 (2011).
Li, Z., Zhang, Y., LeDuc, P. R. & Gregory, K. B. Microbial electricity generation via microfluidic flow control. Biotechnol. Bioeng. 108, 2061–2069 (2011).
Müller, T. et al. Particle-based simulations of steady-state mass transport at high Péclet numbers. arXiv preprint arXiv:1510.05126 (2015).
Lea-Smith, D. J., Bombelli, P., Vasudevan, R. & Howe, C. J. Photosynthetic, respiratory and extracellular electron transport pathways in cyanobacteria. Biochim. Biophys. Acta (BBA) Bioenerg. 1857, 247–255 (2016).
HaoYu, E., Cheng, S., Scott, K. & Logan, B. Microbial fuel cell performance with non-Pt cathode catalysts. J. Power Sources 171, 275–281 (2007).
Kakarla, R. & Min, B. Photoautotrophic microalgae scenedesmus obliquus attached on a cathode as oxygen producers for microbial fuel cell (mfc) operation. Int. J. Hydrog. Energy 39, 10275–10283 (2014).
Schneider, K., Thorne, R. J. & Cameron, P. J. An investigation of anode and cathode materials in photomicrobial fuel cells. Philos. Trans. Math. Phys. Eng. Sci. 374, 20150080 (2016).
Qian, F. & Morse, D. E. Miniaturizing microbial fuel cells. Trends Biotechnol. 29, 62–69 (2011).
Williams, J. G. Construction of specific mutations in photosystem II photosynthetic reaction center by genetic engineering methods in Synechocystis 6803. Methods Enzymol. 167, 766–778 (1988).
Castenholz, R. W. Culturing methods for cyanobacteria. Methods Enzymol. 167, 68–93 (1988).
Lea-Smith, D. J., Vasudevan, R. & Howe, C. J. Generation of marked and markerless mutants in model cyanobacterial species. J. Vis. Exp. 1–11 (2016).
Duffy, D. C., McDonald, J. C., Schueller, O. J. & Whitesides, G. M. Rapid prototyping of microfluidic systems in poly(dimethylsiloxane). Anal. Chem. 70, 4974–4984 (1998).
So, J.-H. & Dickey, M. D. Inherently aligned microfluidic electrodes composed of liquid metal. Lab. Chip 11, 905–911 (2011).
Herling, T. et al. Integration and characterization of solid wall electrodes in microfluidic devices fabricated in a single photolithography step. Appl. Phys. Lett. 102, 184102 (2013).
MacKay, D. J. C. Solar energy in the context of energy use, energy transportation and energy storage. Philos. Trans. Math. Phys. Eng. Sci. 371, 20110431 (2013).
Dabiri, J. O. et al. A new approach to wind energy: opportunities and challenges. AIP Conf. Proc. 1652, 51–57 (2015).
De Castro, C., Mediavilla, M., Miguel, L. J. & Frechoso, F. Global solar electric potential: a review of their technical and sustainable limits. Renew. Sust. Energ. Rev. 28, 824–835 (2013).
Acknowledgements
The research leading to these results has received funding from the Engineering and Physical Sciences Research Council (K.L.S., T.P.J.K.), the Leverhulme Trust (P.B., C.J.H., T.P.J.K.; RPG-2015-393), the European Research Council under the European Union’s Seventh Framework Programme (FP7/2007-2013) through the ERC grant PhysProt (agreement no. 337969), the Biotechnology and Biological Sciences Research Council (T.P.C.; BB/J014540/1), the Environmental Services Association Education Trust (D.J.L.-S.) and the EnAlgae consortium (P.B., C.J.H.). We thank L. Lea for help in constructing Fig. 1.
Author information
Authors and Affiliations
Contributions
K.L.S., P.B., T.M., E.M.A., C.J.H. and T.P.J.K. designed the study K.L.S., P.B., D.J.L.S. and T.P.C. performed the experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–5, Supplementary Table 2, Supplementary References.
Supplementary Table
Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Saar, K.L., Bombelli, P., Lea-Smith, D.J. et al. Enhancing power density of biophotovoltaics by decoupling storage and power delivery. Nat Energy 3, 75–81 (2018). https://doi.org/10.1038/s41560-017-0073-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-017-0073-0
This article is cited by
-
Rewiring photosynthetic electron transport chains for solar energy conversion
Nature Reviews Bioengineering (2023)
-
Arraying of microphotosynthetic power cells for enhanced power output
Microsystems & Nanoengineering (2022)
-
3D-printed hierarchical pillar array electrodes for high-performance semi-artificial photosynthesis
Nature Materials (2022)
-
Evaluating application of photosynthetic microbial fuel cell to exhibit efficient carbon sequestration with concomitant value-added product recovery from wastewater: A review
Environmental Science and Pollution Research (2022)
-
The use of marine microalgae in microbial fuel cells, photosynthetic microbial fuel cells and biophotovoltaic platforms for bioelectricity generation
3 Biotech (2022)