Abstract
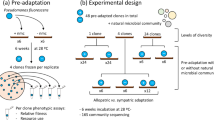
In an era of pervasive anthropogenic ecological disturbances, there is a pressing need to understand the factors that constitute community response and resilience. A detailed understanding of disturbance response needs to go beyond associations and incorporate features of disturbances, species traits, rapid evolution and dispersal. Multispecies microbial communities that experience antibiotic perturbation represent a key system with important medical dimensions. However, previous microbiome studies on this theme have relied on high-throughput sequencing data from uncultured species without the ability to explicitly account for the role of species traits and immigration. Here, we serially passage a 34-species defined bacterial community through different levels of pulse antibiotic disturbance, manipulating the presence or absence of species immigration. To understand the ecological community response measured using amplicon sequencing, we combine initial trait data measured for each species separately and metagenome sequencing data revealing adaptive mutations during the experiment. We found that the ecological community response was highly repeatable within the experimental treatments, which could be attributed in part to key species traits (antibiotic susceptibility and growth rate). Increasing antibiotic levels were also coupled with an increasing probability of species extinction, making species immigration critical for community resilience. Moreover, we detected signals of antibiotic-resistance evolution occurring within species at the same time scale, leaving evolutionary changes in communities despite recovery at the species compositional level. Together, these observations reveal a disturbance response that presents as classic species sorting, but is nevertheless accompanied by rapid within-species evolution.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Raw sequencing data (FASTQ files) have been deposited at the NCBI Sequence Read Archive (SRA) under the accession number PRJNA632457. All preprocessed data needed to reproduce the downstream analyses and figures are available at GitHub (https://github.com/johannescairns/repeatable_dynamics; permanent URL, https://doi.org/10.5281/zenodo.3908935).
Code availability
All code needed to reproduce the downstream analyses and figures are available at GitHub (https://github.com/johannescairns/repeatable_dynamics; permanent URL, https://doi.org/10.5281/zenodo.3908935).
References
Smith, M. D., Knapp, A. K. & Collins, S. L. A framework for assessing ecosystem dynamics in response to chronic resource alterations induced by global change. Ecology 90, 3279–3289 (2009).
Rockstrom, J. et al. A safe operating space for humanity. Nature 461, 472–475 (2009).
Morley, V. J., Woods, R. J. & Read, A. F. Bystander selection for antimicrobial resistance: implications for patient health. Trends Microbiol. 27, 864–877 (2019).
Vrancken, G., Gregory, A. C., Huys, G. R. B., Faust, K. & Raes, J. Synthetic ecology of the human gut microbiota. Nat. Rev. Microbiol. 17, 754–763 (2019).
Lässig, M., Mustonen, V. & Walczak, A. M. Predicting evolution. Nat. Ecol. Evol. 1, 0077 (2017).
Robinson, J. I. et al. Metabolomic networks connect host-microbiome processes to human Clostridioides difficile infections. J. Clin. Invest. 130, 3792–3806 (2019).
Oh, S., Choi, D. & Cha, C. J. Ecological processes underpinning microbial community structure during exposure to subinhibitory level of triclosan. Sci. Rep. 9, 4598 (2019).
Zorner, P., Farmer, S. & Alibek, K. Quantifying crop rhizosphere microbiome ecology: the next frontier in enhancing the commercial utility of agricultural microbes. Ind. Biotechnol. 14, 116–119 (2018).
Lozada-Gobilard, S. et al. Environmental filtering predicts plant-community trait distribution and diversity: kettle holes as models of meta-community systems. Ecol. Evol. 9, 1898–1910 (2019).
Li, J. et al. Shared molecular targets confer resistance over short and long evolutionary timescales. Mol. Biol. Evol. 36, 691–708 (2019).
Vázquez-García, I. et al. Clonal heterogeneity influences the fate of new adaptive mutations. Cell Rep. 21, 732–744 (2017).
Illingworth, C. J. R., Parts, L., Schiffels, S., Liti, G. & Mustonen, V. Quantifying selection acting on a complex trait using allele frequency time series data. Mol. Biol. Evol. 29, 1187–1197 (2012).
Leibler, S. & Kussell, E. Individual histories and selection in heterogeneous populations. Proc. Natl Acad. Sci. USA 107, 13183–13188 (2010).
Des Roches, S. et al. The ecological importance of intraspecific variation. Nat. Ecol. Evol. 2, 57–64 (2018).
Wistrand-Yuen, E. et al. Evolution of high-level resistance during low-level antibiotic exposure. Nat. Commun. 9, 1599 (2018).
Schoener, T. W. The newest synthesis: understanding the interplay of evolutionary and ecological dynamics. Science 331, 426–429 (2011).
Scheuerl, T. et al. Bacterial adaptation is constrained in complex communities. Nat. Commun. 11, 754 (2020).
Roodgar, M. et al. Longitudinal linked read sequencing reveals ecological and evolutionary responses of a human gut microbiome during antibiotic treatment. Preprint at bioRxiv https://doi.org/10.1101/2019.12.21.886093 (2019).
Dethlefsen, L. & Relman, D. A. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl Acad. Sci. USA 108, 4554–4561 (2011).
Palleja, A. et al. Recovery of gut microbiota of healthy adults following antibiotic exposure. Nat. Microbiol. 3, 1255–1265 (2018).
Jernberg, C., Lofmark, S., Edlund, C. & Jansson, J. K. Long-term ecological impacts of antibiotic administration on the human intestinal microbiota. ISME J. 1, 56–66 (2007).
Yassour, M. et al. Natural history of the infant gut microbiome and impact of antibiotic treatment on bacterial strain diversity and stability. Sci. Transl. Med. 8, 343ra81 (2016).
Mack, I. et al. Antimicrobial resistance following azithromycin mass drug administration: potential surveillance strategies to assess public health impact. Clin. Infect. Dis. 70, 1501–1508 (2019).
Tamburini, S., Shen, N., Wu, H. C. & Clemente, J. C. The microbiome in early life: implications for health outcomes. Nat. Med. 22, 713–722 (2016).
Langdon, A., Crook, N. & Dantas, G. The effects of antibiotics on the microbiome throughout development and alternative approaches for therapeutic modulation. Genome Med. 8, 39 (2016).
Brown, J. H. & Kodricbrown, A. Turnover rates in insular biogeography: effect of immigration on extinction. Ecology 58, 445–449 (1977).
Patel, R. & DuPont, H. L. New approaches for bacteriotherapy: prebiotics, new-generation probiotics, and synbiotics. Clin. Infect. Dis. 60, 108–121 (2015).
Lawley, T. D. et al. Targeted restoration of the intestinal microbiota with a simple, defined bacteriotherapy resolves relapsing Clostridium difficile disease in mice. PLoS Pathog. 8, e1002995 (2012).
Ingrisch, J. & Bahn, M. Towards a comparable quantification of resilience. Trends Ecol. Evol. 33, 251–259 (2018).
Morton, J. T. et al. Establishing microbial composition measurement standards with reference frames. Nat. Commun. 10, 2719 (2019).
Trindade, S., Sousa, A. & Gordo, I. Antibiotic resistance and stress in the light of Fisher’s model. Evolution 66, 3815–3824 (2012).
Jasinska, W. et al. Chromosomal barcoding of E. coli populations reveals lineage diversity dynamics at high resolution. Nat. Ecol. Evol. 4, 437–452 (2020).
Springer, B. et al. Mechanisms of streptomycin resistance: selection of mutations in the 16S rRNA gene conferring resistance. Antimicrob. Agents Chemother. 45, 2877–2884 (2001).
Nishimura, K., Hosaka, T., Tokuyama, S., Okamoto, S. & Ochi, K. Mutations in rsmG, encoding a 16S rRNA methyltransferase, result in low-level streptomycin resistance and antibiotic overproduction in Streptomyces coelicolor A3(2). J. Bacteriol. 189, 3876–3883 (2007).
Macfarlane, E. L. A., Kwasnicka, A. & Hancock, R. E. W. Role of Pseudomonas aeruginosa PhoP-PhoQ in resistance to antimicrobial cationic peptides and aminoglycosides. Microbiology 146, 2543–2554 (2000).
Girgis, H. S., Hottes, A. K. & Tavazoie, S. Genetic architecture of intrinsic antibiotic susceptibility. PLoS ONE 4, e5629 (2009).
Liu, Y., Chen, X., Pan, L. & Mao, Z. Differential protein expression of a streptomycin-resistant Streptomyces albulus mutant in high yield production of ε-poly-l-lysine: a proteomics study. RSC Adv. 9, 24092–24104 (2019).
Beardmore, R. E. et al. Drug-mediated metabolic tipping between antibiotic resistant states in a mixed-species community. Nat. Ecol. Evol. 2, 1312–1320 (2018).
Perron, G. G., Gonzalez, A. & Buckling, A. The rate of environmental change drives adaptation to an antibiotic sink. J. Evol. Biol. 21, 1724–1731 (2008).
Retel, C. et al. The feedback between selection and demography shapes genomic diversity during coevolution. Sci. Adv. 5, eaax0530 (2019).
Day, T., Huijben, V. & Read, A. F. Is selection relevant in the evolutionary emergence of drug resistance? Trends Microbiol. 23, 126–133 (2015).
Melnyk, A. H., Wong, A. & Kassen, R. The fitness costs of antibiotic resistance mutations. Evol. Appl. 8, 273–283 (2015).
Wein, T. et al. Carrying capacity and colonization dynamics of Curvibacter in the Hydra host habitat. Front. Microbiol. 9, 443 (2018).
Zhou, J. et al. Stochastic assembly leads to alternative communities with distinct functions in a bioreactor microbial community. mBio 4, e00584-12 (2013).
Lozupone, C. A., Stombaugh, J. I., Gordon, J. I., Jansson, J. K. & Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 489, 220–230 (2012).
Shade, A. et al. Fundamentals of microbial community resistance and resilience. Front. Microbiol. 3, 417 (2012).
Waples, R. S., Beechie, T. & Pess, G. R. Evolutionary history, habitat disturbance regimes, and anthropogenic changes: what do these mean for resilience of Pacific salmon populations? Ecol. Soc. 14, 3 (2009).
Johnstone, J. F. et al. Changing disturbance regimes, ecological memory, and forest resilience. Front. Ecol. Environ. 14, 369–378 (2016).
Cairns, J. et al. Construction and characterization of synthetic bacterial community for experimental ecology and evolution. Front. Genet. 9, 312 (2018).
Datta, N., Hedges, R. W., Shaw, E. J., Sykes, R. B. & Richmond, M. H. Properties of an R factor from Pseudomonas aeruginosa. J. Bacteriol. 108, 1244–1249 (1971).
Cairns, J. et al. Ecology determines how low antibiotic concentration impacts community composition and horizontal transfer of resistance genes. Commun. Biol. 1, 35 (2018).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina paired-end read merger. Bioinformatics 30, 614–620 (2014).
Martin, M. & Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).
Ewels, P., Magnusson, M., Lundin, S. & Kaller, M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics 32, 3047–3048 (2016).
Edgar, R. C. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 10, 996–998 (2013).
Rognes, T., Flouri, T., Nichols, B., Quince, C. & Mahe, F. VSEARCH: a versatile open source tool for metagenomics. PeerJ 4, e2584 (2016).
Edgar, R. C. Accuracy of microbial community diversity estimated by closed- and open-reference OTUs. PeerJ 5, e3889 (2017).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Garrison, E. & Gabor, M. Haplotype-based variant detection from short-read sequencing. Preprint at arXiv https://arxiv.org/abs/1207.3907 (2012).
Cingolani, P. et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w(1118); iso-2; iso-3. Fly 6, 80–92 (2012).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2019); http://www.R-project.org/
Krijthe, J. Rtsne: T-distributed stochastic neighbor embedding using a Barnes-Hut implementation. R package version 0.15 https://github.com/jkrijthe/Rtsne (2015).
Liaw, A. & Wiener, M. Classification and regression by randomForest. R News 2, 18–22 (2002).
Kuhn, M. et al. caret: Classification and regression training. R package version 6.0-84 https://CRAN.R-project.org/package=caret (2019).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & R Core Team nlme: Linear and nonlinear mixed effects models. R package version 3.1-142 https://CRAN.R-project.org/package=nlme (2019).
Venables, W. N. & Ripley, B. D. Modern Applied Statistics With S 4th edn (Springer, 2002)
Oksanen, J. et al. vegan: Community ecology package. R package version 2.5-6 https://CRAN.R-project.org/package=vegan (2019).
Zapala, M. A. & Schork, N. J. Multivariate regression analysis of distance matrices for testing associations between gene expression patterns and related variables. Proc. Natl Acad. Sci. USA 103, 19430–19435 (2006).
Excoffier, L., Smouse, P. E. & Quattro, J. M. Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131, 479–491 (1992).
Anderson, M. J. Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62, 245–253 (2006).
Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 26, 32–46 (2001).
Acknowledgements
We thank A. Ronkainen and P. Typpö for technical assistance. This research was funded by the Academy of Finland (grant nos. 106993, to T.H., and 313270, to V.M.), Jenny and Antti Wihuri Foundation (grant no. 190040 to J.C.) and the Heisenberg Stipend from the German Research Foundation (DFG; grant no. 4135/9, to L.B.).
Author information
Authors and Affiliations
Contributions
Design of serial passage experiment: T.H., J.C. and L.B. Performing serial passage experiment: R.J. Design of data analysis: J.C., V.M. and L.B. Performing data analysis: J.C. Interpreting results: J.C., T.H., L.B. and V.M. J.C. wrote the first manuscript draft, with contributions from all of the authors. All of the authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1
A t-SNE map showing de novo community clustering before, during and after recovery from antibiotic pulse at different antibiotic levels with or without immigration (N = 190). The different antibiotic levels are indicated by color coding, and the time points relative to the antibiotic pulse are indicated by different shapes. Low, intermediate and high antibiotic levels correspond to 4, 16 and 128 μg ml–1 streptomycin, respectively. All data points originate from the same t-SNE analysis and have been separated into two panels (with same arbitrary axis units) only for the sake of visual clarity of immigration effect (at high antibiotic level, post-recovery communities indicated by diamonds more often resume pre-disturbance composition in upper left-hand region).
Extended Data Fig. 2
The extinction probability of species as a function of antibiotic level and the presence/absence of immigration (binomial glm estimate ± 95 % confidence intervals). Extinction is defined as the absence of a species after the antibiotic pulse (day 32 onwards) that was present prior to the pulse (day 16), and has been computed only for the species fulfilling these criteria in at least one experimental community (in total, 146 cases of extinction were observed). Low, intermediate and high antibiotic levels correspond to 4, 16 and 128 μg ml–1 streptomycin, respectively.
Extended Data Fig. 3
Bacterial biomass estimated by optical density (OD) at 600 nm at different levels of antibiotic pulse (expressed in μg ml–1) in the pre-disturbance (day 16), post-disturbance (day 32) and post-recovery (day 48) phases (mean ± standard deviation).
Extended Data Fig. 4
Global comparative view of community composition as shown by Kullback-Leibler (KL) divergence across all samples (N = 190). The color scale from blue to red indicates the degree to which community composition differs between two communities. KL divergence has been computed from species compositional data. The heat map has been color-annotated for the different immigration and antibiotic treatments and experimental phases. Low, intermediate and high antibiotic levels correspond to 4, 16 and 128 μg ml–1 streptomycin, respectively.
Extended Data Fig. 5
Competitive fitness landscapes across all samples during the antibiotic pulse and recovery phases (N = 126). The color scale from blue to red indicates the degree to which the competitive fitness landscapes are correlated between two communities. Correlations have been computed from competitive fitness data for each species in the communities. The heat map has been color-annotated for the different immigration and antibiotic treatments and experimental phases. Low, intermediate and high antibiotic levels correspond to 4, 16 and 128 μg ml–1 streptomycin, respectively. The black boxes show the data presented at species-level detail in Fig. 4 in the main text.
Extended Data Fig. 6
Correlation of competitive fitness landscapes within replicates in each experimental treatment (mean + 95 % confidence interval). Correlations have been computed from competitive fitness data for each species in the communities. The figure aggregates the pairwise correlation values shown for each replicate pair within treatments in Figure S4. Low, intermediate and high antibiotic levels correspond to 4, 16 and 128 μg ml–1 streptomycin, respectively.
Extended Data Fig. 7
Percentage of variance in the competitive fitness of species explained by the experimental treatments (antibiotic level and presence / absence of species immigration) and species traits (antibiotic MIC and intrinsic growth rate). The variance partitioning is based on ANOVA on competitive fitness performed separately for the antibiotic pulse and recovery phases (detailed results are presented in Supplementary Tables 1 and 2).
Extended Data Fig. 8
Illumina read recruitment (median per species) in whole genome alignments for deep sequencing data (two upper panels) or raw 16S rRNA amplicon data (bottom panel). The HAMBI codes of the species are indicated in the horizontal axis, with two exceptions: K12 and RP4 denote the chromosome and plasmid sequence, respectively, from E. coli JE2571. Read recruitment in whole genome alignments is indicated as number of reads (100 bp) in 1,000 bp blocks, and needs to be divided by 10 ((100 bp × read count)/(1,000 bp block)) to obtain an estimate of genome coverage. For instance, a median read count of 1,000 corresponds to roughly 100× genome coverage. In the uppermost panel, deep sequencing data was mapped separately to the genome of each individual species, and in the middle panel, the data was mapped to a multi-FASTA file containing all the genomes, producing comparable results. 16S rRNA amplicon read counts have been normalized to 15,000 reads per sample.
Extended Data Fig. 9
Deep sequencing read recruitment across the genomes of abundant species. Genomic position is indicated as relative position (0–1) across the whole chromosome for closed genomes or largest contig for draft genomes. Read recruitment in whole genome alignments is indicated as number of reads (100 bp) in 1,000 bp blocks, and needs to be divided by 10 ((100 bp × read count)/(1,000 bp block)) to obtain an estimate of genome coverage.
Supplementary information
Rights and permissions
About this article
Cite this article
Cairns, J., Jokela, R., Becks, L. et al. Repeatable ecological dynamics govern the response of experimental communities to antibiotic pulse perturbation. Nat Ecol Evol 4, 1385–1394 (2020). https://doi.org/10.1038/s41559-020-1272-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-020-1272-9
This article is cited by
-
Steering and controlling evolution — from bioengineering to fighting pathogens
Nature Reviews Genetics (2023)
-
Interspecies interaction reduces selection for antibiotic resistance in Escherichia coli
Communications Biology (2023)
-
Non-antibiotic pharmaceuticals promote conjugative plasmid transfer at a community-wide level
Microbiome (2022)
-
Biofilm antimicrobial susceptibility through an experimental evolutionary lens
npj Biofilms and Microbiomes (2022)
-
Rbec: a tool for analysis of amplicon sequencing data from synthetic microbial communities
ISME Communications (2021)