Abstract
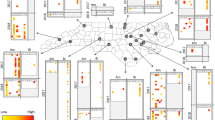
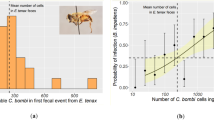
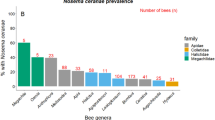
Pollinator reductions can leave communities less diverse and potentially at increased risk of infectious diseases. Species-rich plant and bee communities have high species turnover, making the study of disease dynamics challenging. To address how temporal dynamics shape parasite prevalence in plant and bee communities, we screened >5,000 bees and flowers over an entire growing season for five common bee microparasites (Nosema ceranae, Nosema bombi, Crithidia bombi, Crithidia expoeki and neogregarines). Over 110 bee species and 89 flower species were screened, revealing that 42% of bee species (12.2% individual bees) and 70% of flower species (8.7% individual flowers) had at least one parasite in or on them, respectively. Some common flowers (for example, Lychnis flos-cuculi) harboured multiple parasite species whilst others (for example, Lythrum salicaria) had few. Significant temporal variation of parasite prevalence in bees was linked to bee diversity, bee and flower abundance and community composition. Specifically, we found that bee communities had the highest prevalence late in the season, when social bees (Bombus spp. and Apis mellifera) were dominant and bee diversity was lowest. Conversely, prevalence on flowers was lowest late in the season when floral abundance was highest. Thus turnover in the bee community impacted community-wide prevalence, and turnover in the plant community impacted when parasite transmission was likely to occur at flowers. These results imply that efforts to improve bee health will benefit from the promotion of high floral numbers to reduce transmission risk, maintaining bee diversity to dilute parasites and monitoring the abundance of dominant competent hosts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All raw data, including site surveys and screening results, are found on Dryad in addition to all analysis code used (https://doi.org/10.6086/D1X09V). Sequence data are also deposited in the NCBI database, with accession nos. MT212154, MT212155, MT212156, MT212157, MT212158, MT212159, MT296581, MT296582, MT296583, MT296584, MT296585, MT296586, MT302779, MT302780, MT302781, MT302782, MT302783, MT302784, MT359894–MT359896, MT366919, MT387450 and MT387451.
References
Pongsiri, M. J. et al. Biodiversity loss affects global disease ecology. BioScience 59, 945–954 (2009).
Dirzo, R. et al. Defaunation in the Anthropocene. Science 345, 401–406 (2014).
Barnosky, A. D. et al. Has the Earth’s sixth mass extinction already arrived? Nature 471, 51–57 (2011).
Sala, O. E. et al. Accelerated modern human-induced species losses: entering the sixth mass extinction. Sci. Adv. 1, e1400253 (2000).
Anderson, P. K. et al. Emerging infectious diseases of plants: pathogen pollution, climate change and agrotechnology drivers. Trends Ecol. Evol. 19, 535–544 (2004).
Daszak, P., Cunningham, A. A. & Hyatt, A. D. Emerging infectious diseases of wildlife—threats to biodiversity and human health. Science 287, 443–449 (2000).
Johnson, P. T. J. J., de Roode, J. C. & Fenton, A. Why infectious disease research needs community ecology. Science 349, 1259504 (2015).
Goulson, D., Lye, G. C. & Darvill, B. Decline and conservation of bumble bees. Annu. Rev. Entomol. 53, 191–208 (2008).
Williams, P. H. & Osborne, J. L. Bumblebee vulnerability and conservation world-wide. Apidologie 40, 367–387 (2009).
Goulson, D., Nicholls, E., Botias, C. & Rotheray, E. L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 347, 1255957 (2015).
Gallai, N., Salles, J. M., Settele, J. & Vaissiere, B. E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 68, 810–821 (2009).
Paull, S. H. et al. From superspreaders to disease hotspots: linking transmission across hosts and space. Front. Ecol. Environ. 10, 75–82 (2012).
Wood, C. L. et al. Does biodiversity protect humans against infectious disease? Ecology 95, 817–832 (2014).
Salkeld, D. J., Padgett, K. A. & Jones, J. H. A meta-analysis suggesting that the relationship between biodiversity and risk of zoonotic pathogen transmission is idiosyncratic. Ecol. Lett. 16, 679–686 (2013).
Wood, C. L. & Lafferty, K. D. Biodiversity and disease: a synthesis of ecological perspectives on Lyme disease transmission. Trends Ecol. Evol. 28, 239–247 (2013).
Luis, A. D., Kuenzi, A. J. & Mills, J. N. Species diversity concurrently dilutes and amplifies transmission in a zoonotic host–pathogen system through competing mechanisms. Proc. Natl Acad. Sci. USA 115, 7979–7984 (2018).
Keesing, F., Holt, R. D. & Ostfeld, R. S. Effects of species diversity on disease risk. Ecol. Lett. 9, 485–498 (2006).
Ostfeld, R. S. & Keesing, F. Biodiversity and disease risk: the case of Lyme disease. Conserv. Biol. 14, 722–728 (2000).
Schmidt, K. A. & Ostfeld, R. S. Biodiversity and the dilution effect in disease ecology. Ecology 82, 609–619 (2001).
Woolhouse, M. E. J., Dye, C. & Etard, J. Heterogeneities in the transmission of infectious agents: implications for the design of control programs. Proc. Natl Acad. Sci. USA 94, 338–342 (1997).
Graystock, P., Goulson, D. & Hughes, W. O. H. Parasites in bloom: flowers aid dispersal and transmission of pollinator parasites within and between bee species. Proc. R. Soc. B 282, 20151371 (2015).
Rigaud, T., Perrot-Minnot, M.-J. & Brown, M. J. F. Parasite and host assemblages: embracing the reality will improve our knowledge of parasite transmission and virulence. Proc. R. Soc. B 277, 3693–3702 (2010).
Adler, L. S. et al. Disease where you dine: plant species and floral traits associated with pathogen transmission in bumble bees. Ecology 99, 2535–2545 (2018).
McFrederick, Q. S. et al. Flowers and wild megachilid bees share microbes. Microb. Ecol. 73, 188–200 (2017).
CaraDonna, P. J. et al. Interaction rewiring and the rapid turnover of plant-pollinator networks. Ecol. Lett. 20, 385–394 (2017).
Jones, K. E. et al. Global trends in emerging infectious diseases. Nature 451, 990–993 (2008).
Piot, N. et al. Establishment of wildflower fields in poor quality landscapes enhances micro-parasite prevalence in wild bumble bees. Oecologia 189, 149–158 (2019).
Theodorou, P. et al. Pollination services enhanced with urbanization despite increasing pollinator parasitism. Proc. R. Soc. B 283, 21060561 (2016).
Graystock, P., Goulson, D. & Hughes, W. O. H. The relationship between managed bees and the prevalence of parasites in bumblebees. PeerJ 2, e522 (2014).
Graystock, P., Blane, E. J., McFrederick, Q. S., Goulson, D. & Hughes, W. O. H. Do managed bees drive parasite spread and emergence in wild bees? Int. J. Parasitol. Parasites Wildl. 5, 64–75 (2016).
Alger, S. A., Burnham, P. A., Boncristiani, H. F. & Brody, A. K. RNA virus spillover from managed honeybees (Apis mellifera) to wild bumblebees (Bombus spp.). PLoS ONE 14, e0217822 (2019).
Randolph, S. E. & Dobson, A. D. M. Pangloss revisited: a critique of the dilution effect and the biodiversity–buffers–disease paradigm. Parasitology 139, 847–863 (2012).
LoGiudice, K. et al. Impact of host community on Lyme disease risk. Ecology 89, 2841–2849 (2008).
Keesing, F. et al. Impacts of biodiversity on the emergence and transmission of infectious diseases. Nature 468, 647–652 (2010).
Johnson, P. T. J., Lund, P. J., Hartson, R. B. & Yoshino, T. P. Community diversity reduces Schistosoma mansoni transmission, host pathology and human infection risk. Proc. R. Soc. B 276, 1657–1663 (2009).
Mitchell, C. E., Tilman, D. & Groth, J. V. Effects of grassland plant species diversity, abundance, and composition on foliar fungal disease. Ecology 83, 1713–1726 (2013).
Johnson, P. T. J. & Thieltges, D. W. Diversity, decoys and the dilution effect: how ecological communities affect disease risk. J. Exp. Biol. 213, 961–970 (2010).
Becker, D. J., Streicker, D. G. & Altizer, S. Linking anthropogenic resources to wildlife–pathogen dynamics: a review and meta-analysis. Ecol. Lett. 18, 483–495 (2015).
Nunn, C. L., Thrall, P. H. & Kappeler, P. M. Shared resources and disease dynamics in spatially structured populations. Ecol. Modell. 272, 198–207 (2014).
Durrer, S. & Schmid-Hempel, P. Shared use of flowers leads to horizontal pathogen transmission. Proc. R. Soc. B 258, 299–302 (1994).
Figueroa, L. L. et al. Landscape simplification shapes pathogen prevalence in plant-pollinator networks. Ecol. Lett. https://doi.org/10.1111/ele.13521 (2020).
Truitt, L. L., McArt, S. H., Vaughn, A. H. & Ellner, S. P. Trait-based modeling of multihost pathogen transmission: plant-pollinator networks. Am. Nat. 193, E149–E167 (2019).
Lloyd-Smith, J. O., Schreiber, S. J., Kopp, P. E. & Getz, W. M. Superspreading and the effect of individual variation on disease emergence. Nature 438, 355–359 (2005).
Daszak, P. et al. Interdisciplinary approaches to understanding disease emergence: the past, present, and future drivers of Nipah virus emergence. Proc. Natl Acad. Sci. USA 110, 3681–3688 (2013).
Lafferty, K. D. & Gerber, L. R. Good medicine for conservation biology: the intersection of epidemiology and conservation theory. Conserv. Biol. 16, 593–604 (2002).
Cottam, E. M. et al. Integrating genetic and epidemiological data to determine transmission pathways of foot-and-mouth disease virus. Proc. R. Soc. B 275, 887–895 (2008).
Bhatt, S. et al. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature 526, 207–211 (2015).
Pyšek, P. & Richardson, D. M. Invasive species, environmental change and management, and health. Annu. Rev. Environ. Resour. 35, 25–55 (2010).
Malone, J. D. et al. U.S. airport entry screening in response to pandemic influenza: modeling and analysis. Travel Med. Infect. Dis. 7, 181–191 (2009).
Tatem, A. J., Rogers, D. J. & Hay, S. I. Global transport networks and infectious disease spread. Adv. Parasitol. 62, 293–343 (2006).
Nicolaides, C., Cueto-Felgueroso, L., González, M. C. & Juanes, R. A metric of influential spreading during contagion dynamics through the air transportation network. PLoS ONE 7, e40961 (2012).
Gardner, L. & Sarkar, S. A global airport-based risk model for the spread of dengue infection via the air transport network. PLoS ONE 8, e72129 (2013).
Urbanowicz, C. M., Muñiz, P. A. & McArt, S. H. Honey bees and wild bees differ in their preference for and use of introduced floral resources. Ecol. Evol. https://doi.org/10.1002/ece3.6417 (2020).
Wiegand, K. M. & Eames, A. J. The Flora of the Cayuga Lake Basin, New York https://doi.org/10.5962/bhl.title.59518 (The University, 1926).
Medina, B. F. & Medina, V. Central Appalachian Wildflowers (Falcon Guides, 2002).
House, H. D. The Wild Flowers of New York (Univ. of New York Albany, 1918).
Niering, W. A., Olmstead, N. C., Rayfield, S. & Nehring, C. National Audubon Society Field Guide to North American Wildflowers (Eastern Region) (AbeBooks, 1979).
Ascher, J. S. & Pickering, J. DiscoverLife Bee Species Guide and World Checklist (Hymenoptera: Apoidea: Anthophila) (Discover Life, 2020); http://www.discoverlife.org/mp/20q?guide=Apoidea_species
Gibbs, J. Revision of the metallic Lasioglossum (Dialictus) of eastern North America (Hymenoptera: Halictidae: Halictini). Zootaxa 216, 1–216 (2011).
Grixti, J. C., Wong, L. T., Cameron, S. A. & Favret, C. Decline of bumble bees (Bombus) in the North American Midwest. Biol. Conserv. 142, 75–84 (2009).
Sheffield, C. S., Ratti, C., Packer, L. & Griswold, T. Leafcutter and mason bees of the genus Megachile Latreille (Hymenoptera: Megachilidae) in Canada and Alaska. Can. J. Arthropod Identif. 18, 1–107 (2011).
Schwarz, R. S. & Evans, J. D. Single and mixed-species trypanosome and microsporidia infections elicit distinct, ephemeral cellular and humoral immune responses in honey bees. Dev. Comp. Immunol. 40, 300–310 (2013).
Meeus, I., Brown, M. J. F., de Graaf, D. C. & Smagghe, G. Effects of invasive parasites on bumble bee declines. Conserv. Biol. 25, 662–671 (2011).
Solter, L. F. in Microsporidia: Pathogens of Opportunity 1st edn (eds Weiss, L. M. & Becnel, J. J.) 165–194 (Wiley–Blackwell, 2014).
Otti, O. & Schmid-Hempel, P. Nosema bombi: a pollinator parasite with detrimental fitness effects. J. Invertebr. Pathol. 96, 118–124 (2007).
Graystock, P., Yates, K., Darvill, B., Goulson, D. & Hughes, W. O. H. Emerging dangers: deadly effects of an emergent parasite in a new pollinator host. J. Invertebr. Pathol. 114, 114–119 (2013).
Fürst, M. A., McMahon, D. P., Osborne, J. L., Paxton, R. J. & Brown, M. J. F. Disease associations between honeybees and bumblebees as a threat to wild pollinators. Nature 506, 364–366 (2014).
Otti, O. & Schmid-Hempel, P. A field experiment on the effect of Nosema bombi in colonies of the bumblebee Bombus terrestris. Ecol. Entomol. 33, 577–582 (2008).
Higes, M., Martín-Hernández, R. & Meana, A. Nosema ceranae in Europe: an emergent type C nosemosis. Apidologie 41, 375–392 (2010).
Li, J. et al. Diversity of Nosema associated with bumblebees (Bombus spp.) from China. Int. J. Parasitol. Parasites Wildl. 42, 49–61 (2012).
Sinpoo, C., Disayathanoowat, T., Williams, P. H. & Chantawannakul, P. Prevalence of infection by the microsporidian Nosema spp. in native bumblebees (Bombus spp.) in northern Thailand. PLoS ONE 14, e0213171 (2019).
Müller, U., McMahon, D. P. & Rolff, J. Exposure of the wild bee Osmia bicornis to the honey bee pathogen Nosema ceranae. Agric. Entomol. 21, 363–371 (2019).
Bramke, K., Müller, U., McMahon, D. P. & Rolff, J. Exposure of larvae of the solitary bee Osmia bicornis to the honey bee pathogen Nosema ceranae affects life history. Insects 10, 380 (2019).
Brown, M. J. F., Schmid-Hempel, R. & Schmid-Hempel, P. Strong context-dependent virulence in a host–parasite system: reconciling genetic evidence with theory. J. Anim. Ecol. 72, 994–1002 (2003).
Yourth, C. P., Brown, M. J. F. & Schmid-Hempel, P. Effects of natal and novel Crithidia bombi (Trypanosomatidae) infections on Bombus terrestris hosts. Insectes Soc. 55, 86–90 (2008).
Brown, M. J. F., Loosli, R. & Schmid-Hempel, P. Condition-dependent expression of virulence in a trypanosome infecting bumblebees. Oikos 91, 421–427 (2000).
Gegear, R. J., Otterstatter, M. C. & Thomson, J. D. Bumble-bee foragers infected by a gut parasite have an impaired ability to utilize floral information. Proc. R. Soc. B 273, 1073–1078 (2006).
Imhoof, B. & Schmid-Hempel, P. Patterns of local adaptation of a protozoan parasite to its bumblebee host. Oikos 82, 59–65 (1998).
Dill, L. M. Costs of energy shortfall for bumble bee colonies: predation, social parasitism, and brood development. Can. Entomol. 123, 283–293 (1991).
Strobl, V., Yañez, O., Straub, L., Albrecht, M. & Neumann, P. Trypanosomatid parasites infecting managed honeybees and wild solitary bees. Int. J. Parasitol. 49, 605–613 (2019).
Ravoet, J. et al. Differential diagnosis of the honey bee trypanosomatids Crithidia mellificae and Lotmaria passim. J. Invertebr. Pathol. 130, 21–27 (2015).
Ngor, L. et al. Cross-infectivity of honey and bumble bee-associated parasites across three bee families. Parasitology https://doi.org/10.1017/S0031182020001018 (2020).
Lipa, J. J. & Triggiani, O. Apicystis gen. nov. and Apicystis bombi (Liu, Macfarlane & Pengelly) comb. nov. (Protozoa: Neogregarinida), a cosmopolitan parasite of Bombus and Apis (Hymenoptera: Apidae). Apidologie 27, 29–34 (1996).
Graystock, P., Meeus, I., Smagghe, G., Goulson, D. & Hughes, W. O. H. The effects of single and mixed infections of Apicystis bombi and deformed wing virus in Bombus terrestris. Parasitology 143, 358–365 (2016).
Maharramov, J. et al. Genetic variability of the neogregarine Apicystis bombi, an etiological agent of an emergent bumblebee disease. PLoS ONE 8, e81475 (2013).
Rutrecht, S. T. & Brown, M. J. F. The life-history impact and implications of multiple parasites for bumble bee queens. Int. J. Parasitol. 38, 799–808 (2008).
Plischuk, S., Meeus, I., Smagghe, G. & Lange, C. E. Apicystis bombi (Apicomplexa: Neogregarinorida) parasitizing Apis mellifera and Bombus terrestris (Hymenoptera: Apidae) in Argentina. Environ. Microbiol. Rep. 3, 565–568 (2011).
Tian, T., Piot, N., Meeus, I. & Smagghe, G. Infection with the multi-host micro-parasite Apicystis bombi (Apicomplexa: Neogregarinorida) decreases survival of the solitary bee Osmia bicornis. J. Invertebr. Pathol. 158, 43–45 (2018).
Lacey, L. A. Manual of Techniques in Insect Pathology (Academic Press, 1997).
Fries, I. et al. Standard methods for Nosema research. J. Apic. Res. 52, 1–28 (2013).
Mullins, J. L., Strange, J. P. & Tripodi, A. D. Why are queens broodless? Failed nest initiation not linked to parasites, mating status, or ovary development in two bumble bee species of Pyrobombus (Hymenoptera: Apidae: Bombus). J. Econ. Entomol. 113, 575–581 (2019).
Schmid-Hempel, R. & Tognazzo, M. Molecular divergence defines two distinct lineages of Crithidia bombi (Trypanosomatidae), parasites of bumblebees. J. Eukaryot. Microbiol. 57, 337–345 (2010).
Tripodi, A. D., Szalanski, A. L. & Strange, J. P. Novel multiplex PCR reveals multiple trypanosomatid species infecting North American bumble bees (Hymenoptera: Apidae: Bombus). J. Invertebr. Pathol. 153, 147–155 (2018).
King, G. & Zeng, L. Logistic regression in rare events data. Polit. Anal. 9, 137–163 (2001).
Nelder, J. A. A reformulation of linear models. J. R. Stat. Soc. Ser. A 140, 48–77 (1977).
Venables, W. N. Exegeses on linear models. In SPLUS User’s Conference (2000); https://www.stats.ox.ac.uk/pub/MASS3/Exegeses.pdf
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2018).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Brooks, Mollie et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R. J. 9, 378–400 (2017).
Hartig, F. DHARMa: Residual Diagnostics for Hierarchical (Multi-level/Mixed) Regression Models. R package v.0.2.0 https://CRAN.R-project.org/package=DHARMa (2018).
Signorell, A. DescTools: Tools for Descriptive Statistics https://cran.r-project.org/web/packages/DescTools/index.html (2019).
Wood, S. N., Pya, N. & Säfken, B. Smoothing parameter and model selection for general smooth models. J. Am. Stat. Assoc. 111, 1548–1563 (2016).
Engels, B. XNomial: Exact Goodness-of-Fit Test for Multinomial Data with Fixed Probabilities https://cran.r-project.org/web/packages/XNomial/vignettes/XNomial.html (2015).
Hothorn, T., Bretz, F. & Westfall, P. Simultaneous inference in general parametric models. Biom. J. 50, 346–363 (2008).
Oksanen, J. et al. vegan: Community Ecology Package. R package version 2.4-3 https://www.researchgate.net/publication/323265822_vegan_Community_Ecology_Package_R_package_version_24-3_2017_accessed_2016_Jan_1 (2017).
Acknowledgements
T. Salazar and D. Lewis assisted with fieldwork, J. Teague helped with bee dissections, M. Arduser confirmed bee identifications and J. Strange (USDA-ARS-PIRU) provided support in the development of diagnostic primers. The research group of R. Gill provided comments on the manuscript. Research reported in this publication was supported by the National Institute of General Medical Sciences of the National Institutes of Health (NIH, award no. R01GM122062). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Contributions
P.G., Q.S.M., C.R.M. and S.H.M. conceived the study. P.G., A.A.F., Q.S.M., C.R.M. and S.H.M. contributed to study design. P.G. and A.A.F. collected the field data. P.G., K.P. and Q.S.M. conducted the molecular work. A.D.T. developed molecular primers. P.A.M. identified and pinned the bee samples collected. W.H.N., P.G., C.R.M. and S.H.M. contributed to data analysis and wrote the first draft of the manuscript. All authors contributed substantially to the final draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests. A.D.T. contributed to this article in her personal capacity. The views expressed are her own and do not necessarily represent the views of the Agricultural Research Service or the United States Government.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, Tables 1–11 and Figs. 1–10.
Rights and permissions
About this article
Cite this article
Graystock, P., Ng, W.H., Parks, K. et al. Dominant bee species and floral abundance drive parasite temporal dynamics in plant-pollinator communities. Nat Ecol Evol 4, 1358–1367 (2020). https://doi.org/10.1038/s41559-020-1247-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-020-1247-x
This article is cited by
-
Ecological and social factors influence interspecific pathogens occurrence among bees
Scientific Reports (2024)
-
Dominance and identity of the dominant bee drive bee diversity on flowers
Biodiversity and Conservation (2024)
-
Effects of planted pollinator habitat on pathogen prevalence and interspecific detection between bee species
Scientific Reports (2022)
-
Parasitism of urban bumble bees influenced by pollinator taxonomic richness, local garden management, and surrounding impervious cover
Urban Ecosystems (2022)
-
Detection of Microsporidia in Pollinator Communities of a Mediterranean Biodiversity Hotspot for Wild Bees
Microbial Ecology (2022)