Abstract
Recent years have seen an explosion of interest in the overlap between kin selection and sexual selection, particularly concerning how kin selection can put the brakes on harmful sexual conflict. However, there remains a significant disconnect between theory and empirical research. Whilst empirical work has focused on kin-discriminating behaviour, theoretical models have assumed indiscriminating behaviour. Additionally, theoretical work makes particular demographic assumptions that constrain the relationship between genetic relatedness and the scale of competition, and it is not clear that these assumptions reflect the natural setting in which sexual conflict has been empirically studied. Here, we plug this gap between current theoretical and empirical understanding by developing a mathematical model of sexual conflict that incorporates kin discrimination and different patterns of dispersal. We find that kin discrimination and group dispersal inhibit harmful male behaviours at an individual level, but kin discrimination intensifies sexual conflict at the population level.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Code availability
Code used for the simulations is available at https://github.com/GSFaria-wasp/Sexual-conflict.git.
References
Cronin, H. The Ant and the Peacock: Altruism and Sexual Selection from Darwin to Today (Cambridge Univ. Press, 1993).
Boomsma, J. J. Kin selection versus sexual selection: why the ends do not meet. Curr. Biol. 17, R673–R683 (2007).
Pizzari, T. & Gardner, A. The sociobiology of sex: inclusive fitness consequences of inter-sexual interactions. Phil. Trans. R. Soc. Lond. B 367, 2314–2323 (2012).
Rankin, D. J. Kin selection and the evolution of sexual conflict. J. Evol. Biol. 24, 71–81 (2011).
Wild, G., Pizzari, T. & West, S. A. Sexual conflict in viscous populations: the effect of the timing of dispersal. Theor. Popul. Biol. 80, 298–316 (2011).
Carazo, P., Tan, C. K. W., Allen, F., Wigby, S. & Pizzari, T. Within group male relatedness reduces harm to females in Drosophila. Nature 505, 672–675 (2014).
Chippindale, A. K., Berggren, M., Alpern, J. H. M. & Montgomerie, R. Does kin selection moderate sexual conflict in Drosophila? Proc. R. Soc. Lond. B 282, 120151417 (2015).
Pizzari, T., Biernaskie, J. M. & Carazo, P. Inclusive fitness and sexual conflict: how population structure can modulate the battle of the sexes. BioEssays 37, 155–166 (2015).
Faria, G. S., Varela, S. A. M. & Gardner, A. Sex-biased dispersal, kin selection and the evolution of sexual conflict. J. Evol. Biol. 28, 1901–1910 (2015).
Faria, G. S., Varela, S. A. M. & Gardner, A. Sexual selection modulates genetic conflicts and patterns of genomic imprinting. Evolution 71, 526–540 (2017).
Hollis, B. T., Kawecki, T. J. & Keller, L. No evidence that within-group male relatedness reduces harm to females in Drosophila. Ecol. Evol. 5, 979–983 (2015).
Martin, E. S. & Long, T. A. F. Are flies kind to kin? The role of intra- and inter-sexual relatedness in mediating reproductive conflict. Proc. R. Soc. Lond. B 282, 20151991 (2015).
Tan, C. K. W. et al. The contrasting role of male relatedness in different mechanisms of sexual selection in red junglefowl. Evolution 71, 403–420 (2017).
Le Page, S. et al. Male relatedness and familiarity are required to modulate male-induced harm to females in Drosophila. Proc. R. Soc. Lond. B 284, 20170441 (2017).
Łukasiewicz, A., Szubert-Kruszyńska, A. & Radwan, J. Kin selection promotes female productivity and cooperation between the sexes. Sci. Adv. 3, e1602262 (2017).
Lymbery, S. J. & Simmons, L. W. Males harm females less when competing with familiar relatives. Proc. R. Soc. Lond. B 284, 20171984 (2017).
Pitnick, S. & García-González, F. Harm to females increases with male body size in Drosophila melanogaster. Proc. R. Soc. Lond. B 269, 1821–1828 (2002).
Lloyd, W. F. Two Lectures on the Checks to Population (Oxford Univ. Press, 1833).
Hardin, G. The tragedy of the commons. Science 162, 1243–1248 (1968).
Holland, B. & Rice, W. R. Experimental removal of sexual selection reverses intersexual antagonistic coevolution and removes a reproductive load. Proc. Natl Acad. Sci. USA 96, 5083–5088 (1999).
Le Galliard, J. F., Fitze, P. S., Ferrière, R. & Clobert, J. Sex ratio bias, male aggression, and population collapse in lizards. Proc. Natl Acad. Sci. USA 102, 18231–18236 (2005).
Arnqvist, G. & Tuda, M. Sexual conflict and the gender load: correlated evolution between population fitness and sexual dimorphism in seed beetles. Proc. R. Soc. Lond. B 277, 1345–1352 (2010).
Berger, D. et al. Intralocus sexual conflict and the tragedy of the commons in seed beetles. Am. Nat. 188, R98–R112 (2016).
Lymbery, S. J., Tomkins, J. L. & Simmons, L. W. Male responses to sperm competition when rivals vary in number and familiarity. Proc. R. Soc. Lond. B 286, 20182589 (2019).
Hamilton, W. D. The evolution of altruistic behaviour. Am. Nat. 97, 354–356 (1963).
Hamilton, W. D. The genetical evolution of social behaviour. I & II. J. Theor. Biol. 7, 1–52 (1964).
Gardner, A. & West, S. A. Demography, altruism, and the benefits of budding. J. Evol. Biol. 17, 1707–1716 (2006).
Cooper, G. A., Levin, S. R., Wild, G. & West, S. A. Modeling relatedness and demography in social evolution. Evol. Lett. 2, 260–271 (2018).
Frank, S. A. Foundations of Social Evolution (Princeton Univ. Press, 1998).
Gardner, A. Sex-biased dispersal of adults mediates the evolution of altruism among juveniles. J. Theor. Biol. 262, 339–345 (2010).
Taylor, P. D. Altruism in viscous populations – an inclusive fitness approach. Evol. Ecol. 6, 352–356 (1992).
Taylor, P. D. Inclusive fitness in a heterogeneous environment. Proc. R Soc. Lond. B 249, 299–302 (1992).
Queller, D. C. Does population viscosity promote kin selection? Trends Ecol. Evol. 7, 322–324 (1992).
West, S. A., Pen, I. & Griffin, A. S. Cooperation and competition between relatives. Science 296, 72–75 (2002).
Eldakar, O. T., Dlugos, M. J., Pepper, J. W. & Wilson, D. S. Population structure mediates sexual conflict in water striders. Science 326, 816 (2009).
Eldakar, O. T., Wilson, D. S., Dlugos, M. J. & Pepper, J. W. The role of multilevel selection in the evolution of sexual conflict in the water strider Aquarius remiges. Evolution 64, 3183–3189 (2010).
Haldane, J. B. S. The Causes of Evolution (Princeton Univ. Press, 1932).
Goodnight, K. F. The effect of stochastic variation on kin selection in a budding-viscous population. Am. Nat. 140, 1028–1040 (1992).
Lehmann, L., Perrin, N. & Rousset, F. Population demography and the evolution of helping behaviors. Evolution 60, 1137–1151 (2006).
Gardner, A., Arce, A. & Alpedrinha, J. Budding dispersal and the sex ration. J. Evol. Biol. 22, 1036–1045 (2009).
Wright, S. Evolution in Mendelian populations. Genetics 16, 97–159 (1931).
Penn, D. J. & Frommen, J. G. in Animal Behaviour: Evolution and Mechanisms (ed. Kapperler, P.) 55–85 (Springer, 2010).
Bradbury, J. W. & Vehrencamp, S. L. Principles of Animal Communication (Sinauer Associates, 2011).
Holmes, W. G. & Sherman, P. W. Kin recognition in animals: the prevalence of nepotism among animals raises basic questions about how and why they distinguish relatives from unrelated individuals. Am. Sci. 1, 46–55 (1983).
Faria, G. S. & Gardner, A. Does kin discrimination promote cooperation? Biol. Lett. 16, 20190742 (2020).
Griffin, A. S. & West, S. A. Kin discrimination and the benefit of helping in cooperatively breeding vertebrates. Science 302, 634–636 (2003).
Berg, E. C., Lind, M. I., Monahan, S., Bricout, S. & Maklakov, A. A. Kin but less than kind: within-group male relatedness does not increase female fitness in seed beetles. Proc. R. Soc. B 286, 20191664 (2019).
Acknowledgements
G.S.F. acknowledges IAST funding from the French National Research Agency under the Investments for the Future (Investissements d’Avenir) programme (grant no. ANR-17-EURE-0010), and was supported by Portuguese National Funds through a Fundação para a Ciência e a Tecnologia PhD Scholarship (no. SFRH/BD/109726/2015). A.G. was supported by a Natural Environment Research Council Independent Research Fellowship (no. NE/K009524/1) and a European Research Council Consolidator grant (no. 771387). P.C. was supported by a Ramón y Cajal Research Fellowship (no. RYC-2013-12998) and by a Plan Nacional I+D+i Excelencia Research grant (no. CGL2017-89052-P). We thank D. Shuker, J. Kanwal, M. Ritchie, P. Rautiala and T. Hitchcock for helpful comments and discussion.
Author information
Authors and Affiliations
Contributions
G.S.F., A.G. and P.C. conceived the study. G.S.F. led the theoretical analysis with input from A.G. G.S.F., A.G. and P.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
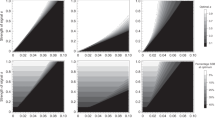
Extended Data Fig. 1 Optimal level of harm as a function of male dispersal (dm).
In the presence of kin discrimination and absence of budding dispersal (A), the optimal level of harm that males express decreases as male dispersal (dm) increases for discriminating males and increases as male dispersal (dm) increases for indiscriminating males. In the absence of kin discrimination and presence of budding dispersal (B), the optimal level of harm that males express increases as male dispersal (dm) increases. In the presence of kin discrimination and budding dispersal (C), the optimal level of harm for discriminating males decreases if males are interacting only with unfamiliar males and increases if males are interacting with familiar males as male dispersal (dm) increases. For indiscriminating males, the optimal level of harm that males express increases as male dispersal (dm) increases. Regardless of absence (A) or presence of budding dispersal (B), males interacting with unfamiliar males express higher level of harm, males interacting with one familiar male and one unfamiliar male express intermediate level of harm, and males interacting with two familiar males express lower level of harm. For all panels, the following parameters were used: marginal benefit of harm β = 0.5; female dispersal rate df = 1; number of females nf = 1; and number of males nm = 3. Additionally, in (B-C) budding dispersal rate dB = 1.
Extended Data Fig. 2 Optimal level of harm in the absence (A) and in the presence (B) of budding dispersal as a function of male dispersal (dm) for discriminating males.
In absence of budding dispersal (A), the optimal level of harm that males express decreases as male dispersal (dm) increases. In the presence of budding dispersal (B), as male dispersal (dm) increases, the optimal level of harm that males express decreases if males are interacting only with unfamiliar males and increases if males are interacting with familiar males. Regardless of absence (A) or presence of budding dispersal (B), males interacting unfamiliar males express higher level of harm, males interacting with one familiar male and one unfamiliar male express intermediate level of harm, and males interacting with two familiar males express lower level of harm. In both panels (A-B), the following parameters were used: marginal benefit of harm β = 0.5; female dispersal rate df = 1; number of females nf = 3; and number of males nm = 3. Additionally, in (B) budding dispersal rate dB = 1. Dots represent the simulations results, with the following additional parameters used: mutation rate of 0.01; population of 4000 patches; number of generations 5 ×104. Each dot is the average of the last 1 ×104 generations.
Extended Data Fig. 3 Level of harm as a function of relatedness between males.
In the absence of kin discrimination, the level of harm that males express changes convexly with relatedness. The following parameters were used: marginal benefit of harm β = 0.5; female dispersal rate df = 1; male dispersal rate dm = 0.5; and relatedness between females and males rfm = 0.
Extended Data Fig. 4 Comparison of different assumptions and how they differ from the main model.
When the level of harm that males express affect all the females in the patch (k = 0), the model is exactly the same as our main model. When harm that the females are subjected to comes half from the male that they mate with and half from the other males (k = 0.5), the model differs from our main model, with lower levels of harm. When harm that the females are subjected comes exclusively from the male that they mate with (k = 1), the model differs from our main model, with lower levels of harm. The following parameters were used: marginal benefit of harm β = 0.5; female dispersal rate df = 1; number of females nf = 3; number of males nm = 3; male dispersal rate dm = 0.5; and relatedness between females and males rfm = 0.
Supplementary information
Supplementary Information
Supplementary information about the mathematical model.
Rights and permissions
About this article
Cite this article
Faria, G.S., Gardner, A. & Carazo, P. Kin discrimination and demography modulate patterns of sexual conflict. Nat Ecol Evol 4, 1141–1148 (2020). https://doi.org/10.1038/s41559-020-1214-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-020-1214-6
This article is cited by
-
The scale of competition impacts parasite virulence evolution
Evolutionary Ecology (2023)