Abstract
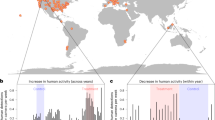
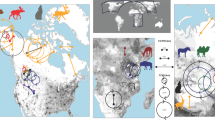
Disturbance and habitat modification by humans can alter animal movement, leading to negative impacts on fitness, survival and population viability. However, the ubiquity and nature of these impacts across diverse taxa has not been quantified. We compiled 208 studies on 167 species from terrestrial and aquatic ecosystems across the globe to assess how human disturbance influences animal movement. We show that disturbance by humans has widespread impacts on the movements of birds, mammals, reptiles, amphibians, fish and arthropods. More than two-thirds of 719 cases represented a change in movement of 20% or more, with increases in movement averaging 70% and decreases −37%. Disturbance from human activities, such as recreation and hunting, had stronger impacts on animal movement than habitat modification, such as logging and agriculture. Our results point to a global restructuring of animal movement and emphasize the need to reduce the negative impacts of humans on animal movement.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data is available at Figshare https://doi.org/10.6084/m9.figshare.12768350.
Code availability
Code is available at Figshare https://doi.org/10.6084/m9.figshare.12768350.
References
Newbold, T. et al. Global effects of land use on local terrestrial biodiversity. Nature 520, 45–50 (2015).
Halpern, B. S. et al. A global map of human impact on marine ecosystems. Science 319, 948–952 (2008).
Queiroz, N. et al. Global spatial risk assessment of sharks under the footprint of fisheries. Nature 572, 461–466 (2019).
Tucker, M. A. et al. Moving in the anthropocene: global reductions in terrestrial mammalian movements. Science 359, 466–469 (2018).
Wang, X. et al. Stochastic simulations reveal few green wave surfing populations among spring migrating herbivorous waterfowl. Nat. Commun. 10, 2187 (2019).
Fahrig, L. Non-optimal animal movement in human-altered landscapes. Funct. Ecol. 21, 1003–1015 (2007).
Cosgrove, A. J., McWhorter, T. J. & Maron, M. Consequences of impediments to animal movements at different scales: a conceptual framework and review. Divers. Distrib. 24, 448–459 (2018).
Mergey, M., Helder, R. & Roeder, J.-J. Effect of forest fragmentation on space-use patterns in the European pine marten (Martes martes). J. Mammal. 92, 328–335 (2011).
Main, M. T., Davis, R. A., Blake, D., Mills, H. & Doherty, T. S. Human impact overrides bioclimatic drivers of red fox home range size globally. Divers. Distrib. https://doi.org/10.1111/ddi.13115 (2020).
Laver, P. N. & Alexander, K. A. Association with humans and seasonality interact to reverse predictions for animal space use. Mov. Ecol. 6, 5 (2018).
Riotte-Lambert, L. & Matthiopoulos, J. Environmental predictability as a cause and consequence of animal movement. Trends Ecol. Evol. 35, 163–174 (2020).
Laurian, C., Ouellet, J.-P., Courtois, R., Breton, L. & St-Onge, S. Effects of intensive harvesting on moose reproduction. J. Appl. Ecol. 37, 515–531 (2000).
Tamburello, N., Côté, I. M. & Dulvy, N. K. Energy and the scaling of animal space use. Am. Nat. 186, 196–211 (2015).
Jetz, W., Carbone, C., Fulford, J. & Brown, J. H. The scaling of animal space use. Science 306, 266–268 (2004).
Perona, A. M., Urios, V. & López-López, P. Holidays? Not for all. Eagles have larger home ranges on holidays as a consequence of human disturbance. Biol. Conserv. 231, 59–66 (2019).
Staggenborg, J., Schaefer, H. M., Stange, C., Naef-Daenzer, B. & Grüebler, M. U. Time and travelling costs during chick-rearing in relation to habitat quality in little owls Athene noctua. Ibis (Lond. 1859) 159, 519–531 (2017).
Suraci, J. P., Clinchy, M., Zanette, L. Y. & Wilmers, C. C. Fear of humans as apex predators has landscape-scale impacts from mountain lions to mice. Ecol. Lett. 22, 1578–1586 (2019).
Blomquist, S. & Hunter, M. L. Jr A multi-scale assessment of habitat selection and movement patterns by northern leopard frog (Lithobates [Rana] pipiens) in a managed forest. Herpetol. Conserv. Biol. 4, 142–160 (2009).
Peaden, J. M., Nowakowski, A. J., Tuberville, T. D., Buhlmann, K. A. & Todd, B. D. Effects of roads and roadside fencing on movements, space use, and carapace temperatures of a threatened tortoise. Biol. Conserv. 214, 13–22 (2017).
Siffczyk, C., Brotons, L., Kangas, K. & Orell, M. Home range size of willow tits: a response to winter habitat loss. Oecologia 136, 635–642 (2003).
Breininger, D. R., Bolt, M. R., Legare, M. L., Drese, J. H. & Stolen, E. D. Factors influencing home-range sizes of eastern indigo snakes in central Florida. J. Herpetol. 45, 484–490 (2011).
Hirt, M. R., Jetz, W., Rall, B. C. & Brose, U. A general scaling law reveals why the largest animals are not the fastest. Nat. Ecol. Evol. 1, 1116–1122 (2017).
Garland, T. & Albuquerque, R. L. Locomotion, energetics, performance, and behavior: a mammalian perspective on lizards, and vice versa. Integr. Comp. Biol. 57, 252–266 (2017).
Wilson, K. S., Pond, B. A., Brown, G. S. & Schaefer, J. A. The biogeography of home range size of woodland caribou Rangifer tarandus caribou. Divers. Distrib. 25, 205–216 (2019).
Wang, Y., Smith, J. A. & Wilmers, C. C. Residential development alters behavior, movement, and energetics in a top carnivore. PLoS ONE 12, e0184687 (2017).
Vangestel, C., Braeckman, B. P., Matheve, H. & Lens, L. Constraints on home range behaviour affect nutritional condition in urban house sparrows (Passer domesticus). Biol. J. Linn. Soc. Lond. 101, 41–50 (2010).
Hinam, H. L. & St. Clair, C. C. High levels of habitat loss and fragmentation limit reproductive success by reducing home range size and provisioning rates of northern saw-whet owls. Biol. Conserv. 141, 524–535 (2008).
Herrera, J. M., de Sá Teixeira, I., Rodríguez-Pérez, J. & Mira, A. Landscape structure shapes carnivore-mediated seed dispersal kernels. Landsc. Ecol. 31, 731–743 (2016).
Carpenter, J. K., O’Donnell, C. F. J., Moltchanova, E. & Kelly, D. Long seed dispersal distances by an inquisitive flightless rail (Gallirallus australis) are reduced by interaction with humans. R. Soc. Open Sci. 6, 190397 (2019).
Januchowski-Hartley, F. A., Graham, N. A. J., Feary, D. A., Morove, T. & Cinner, J. E. Fear of fishers: human predation explains behavioral changes in coral reef fishes. PLoS ONE 6, e22761 (2011).
Whittington, J., Low, P. & Hunt, B. Temporal road closures improve habitat quality for wildlife. Sci. Rep. 9, 3772 (2019).
Soanes, K. et al. Movement re-established but not restored: inferring the effectiveness of road-crossing mitigation for a gliding mammal by monitoring use. Biol. Conserv. 159, 434–441 (2013).
Jacobsen, L. B., Chrenková, M., Sunde, P. & Salek, M. Effects of food provisioning and habitat management on spatial behaviour of little owls during the breeding season. Ornis Fenn. 93, 121–129 (2016).
Zeller, K. A., Lewsion, R., Fletcher, R. J., Tulbure, M. G. & Jennings, M. K. Understanding the importance of dynamic landscape connectivity. Land (Basel) 9, 303 (2020).
Doherty, T. S. & Driscoll, D. A. Coupling movement and landscape ecology for animal conservation in production landscapes. Proc. R. Soc. Lond. B 285, 20172272 (2018).
Rohatgi, A. WebPlotDigitizer, version 4.2 (2019); https://automeris.io/WebPlotDigitizer
Börger, L. et al. Effects of sampling regime on the mean and variance of home range size estimates. J. Anim. Ecol. 75, 1393–1405 (2006).
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 36, 1–48 (2010).
Hedges, L. V., Gurevitch, J. & Curtis, P. S. The meta-analysis of response ratios in experimental ecology. Ecology 80, 1150–1156 (1999).
Neumann, W., Ericsson, G. & Dettki, H. Does off-trail backcountry skiing disturb moose? Eur. J. Wildl. Res. 56, 513–518 (2010).
Wilman, H. et al. EltonTraits 1.0: species-level foraging attributes of the world’s birds and mammals. Ecology 95, 2027 (2014).
Feldman, A., Sabath, N., Pyron, R. A., Mayrose, I. & Meiri, S. Body sizes and diversification rates of lizards, snakes, amphisbaenians and the tuatara. Glob. Ecol. Biogeogr. 25, 187–197 (2016).
Oliveira, B. F., São-Pedro, V. A., Santos-Barrera, G., Penone, C. & Costa, G. C. AmphiBIO, a global database for amphibian ecological traits. Sci. Data 4, 170123 (2017).
Froese, R. & Pauly, D. (eds) Fishbase (2019); www.fishbase.org
Myers, P. et al. The Animal Diversity Web (Univ. Michigan, 2020); https://animaldiversity.org
AmphibiaWeb (Univ. California Berkeley, 2020); https://amphibiaweb.org
Froese, R., Thorson, J. T. & Reyes, R. B. A Bayesian approach for estimating length–weight relationships in fishes. J. Appl. Ichthyol. 30, 78–85 (2014).
Meiri, S. Traits of lizards of the world: variation around a successful evolutionary design. Glob. Ecol. Biogeogr. 27, 1168–1172 (2018).
Bürkner, P. C. Advanced Bayesian multilevel modeling with the R package brms. R J. 10, 395–411 (2018).
Bürkner, P. C. brms: an R package for Bayesian multilevel models using Stan. J. Stat. Softw. 80, 1–28 (2017).
Williams, D. R., Rast, P. & Bürkner, P.-C. Bayesian meta-analysis with weakly informative prior distributions. Preprint at PsyArXiv https://doi.org/10.31234/osf.io/7tbrm (2018).
Gelman, A. & Rubin, D. B. Inference from iterative simulation using multiple sequences. Stat. Sci. 7, 457–511 (1992).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Rosenberg, M. S. The file-drawer problem revisted: a general weighted method for calculating fail-safe numbers in meta-analysis. Evolution 59, 464–468 (2005).
Acknowledgements
We thank the following people for providing additional information about their studies: L. Amo, J. Beasley, K. Borkin, H. Ling Chen, T. Crist, S. Dale, C. Dussault, C. Gómez Posada, T. Gehring, N. Haddad, C. Huveneers, C. Lanctôt, K. Mabry, L. Powell, A. Trochet, C. Vangestel, K. VerCauteren and D. Zeller. We acknowledge the Wurundjeri people of the Kulin nations as the traditional custodians of the land on which this review was conducted. We acknowledge the technical assistance of H. Lydecker from the Sydney Informatics Hub, a Core Research Facility of the University of Sydney, and the use of the University of Sydney’s high performance computing cluster, Artemis. T.S.D. was supported by an Alfred Deakin Postdoctoral Research Fellowship from Deakin University and a Discovery Early Career Researcher Award from the Australian Research Council.
Author information
Authors and Affiliations
Contributions
T.S.D. conceived the study, collated and analysed the data, and led the writing of the manuscript. G.C.H. and D.A.D. helped write the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Ecology & Evolution thanks Laura Prugh and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Database collation summary.
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) diagram detailing the procedure for identifying and including relevant publications.
Extended Data Fig. 2 Funnel plots for the movement distance data (left) and home range data (right).
The white area bordered by dashed lines represents the region of 95% pseudo confidence intervals where 95% of studies are expected to fall in the absence of bias and heterogeneity.
Extended Data Fig. 3 Definition of disturbance types.
Full reference details for study ID numbers can be found in the data file at Figshare.
Extended Data Fig. 4 Modelling results to estimate (a) the mean effects of disturbance on each movement type and (b) the mean positive and negative effects.
Posterior means (‘estimate’) and 95% credible intervals are presented for weighted and unweighted analyses of movement distance and home range size.
Extended Data Fig. 5 Modelling results for the effect of taxonomic group on animal movement responses to disturbance.
Posterior means (‘estimate’) and 95% credible intervals are presented for weighted and unweighted analyses of movement distance and home range size. – indicates that insufficient data was available to fit that particular model.
Extended Data Fig. 6 Modelling results for the effect of trophic level on animal movement responses to disturbance.
Posterior means (‘estimate’) and 95% credible intervals are presented for weighted and unweighted analyses of movement distance and home range size.
Extended Data Fig. 7 Effects of disturbance on animal movement distances and home range size according to trophic level (herbivore, omnivore or carnivore).
Positive effects represent increased movement in response to disturbance, and the opposite for negative effects. Symbols represent posterior means and coloured bands represent 95%, 80% and 50% credible intervals.
Extended Data Fig. 8 Modelling results for the effect of body mass on animal movement responses to disturbance.
Posterior means (‘estimate’) and 95% credible intervals are presented for weighted and unweighted analyses of movement distance and home range size. Sample size for each model is given in parentheses below taxonomic group. – indicates that insufficient data was available to fit that particular model.
Extended Data Fig. 9 Modelling results for the effect of (a) broad disturbance type (human activities or habitat modification) and (b) individual disturbance type on animal movement responses to disturbance.
Posterior means (‘estimate’) and 95% credible intervals are presented for weighted and unweighted analyses of movement distance and home range size. The analysis of individual disturbance types was restricted to combinations of taxonomic group and disturbance that had a minimum of 10 effect sizes from a minimum of three studies (combined). – indicates that insufficient data was available to fit that particular model.
Extended Data Fig. 10 Modelling results for the effect of geographic region on animal movement responses to disturbance.
Posterior means (‘estimate’) and 95% credible intervals are presented for unweighted analyses of movement distance and home range size.
Supplementary information
Rights and permissions
About this article
Cite this article
Doherty, T.S., Hays, G.C. & Driscoll, D.A. Human disturbance causes widespread disruption of animal movement. Nat Ecol Evol 5, 513–519 (2021). https://doi.org/10.1038/s41559-020-01380-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-020-01380-1
This article is cited by
-
Movement ecology of an endangered mesopredator in a mining landscape
Movement Ecology (2024)
-
HPAIV outbreak triggers short-term colony connectivity in a seabird metapopulation
Scientific Reports (2024)
-
Coexistence in multi-use landscape: linking human activities with functional traits of wild mammals in southern India
Landscape Ecology (2024)
-
Predicting movement speed of beetles from body size and temperature
Movement Ecology (2023)
-
Integrating Habitat Availability, Permeability, and Configuration in a Model of Landscape Connectivity: The Contribution of Habitat’s Site-to-Site
Environmental Management (2023)