Abstract
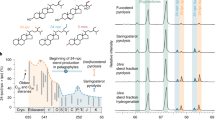
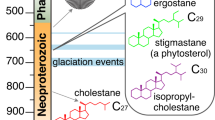
The absence of unambiguous animal body fossils in rocks older than the late Ediacaran has rendered fossil lipids the most promising tracers of early organismic complexity. Yet much debate surrounds the various potential biological sources of putative metazoan steroids found in Precambrian rocks. Here we show that 26-methylated steranes—hydrocarbon structures currently attributed to the earliest animals—can form via geological alteration of common algal sterols, which carries important implications for palaeo-ecological interpretations and inhibits the use of such unconventional ‘sponge’ steranes for reconstructing early animal evolution.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All data are available in the Supplementary Information.
References
Lamb, D. M., Awramik, S. M., Chapman, D. J. & Zhu, S. Evidence for eukaryotic diversification in the ∼1800 million-year-old Changzhougou Formation, North China. Precambrian Res. 173, 93–104 (2009).
Knoll, A. H. & Nowak, M. A. The timetable of evolution. Sci. Adv. 3, e1603076 (2017).
Antcliffe, J. B., Callow, R. H. T. & Brasier, M. D. Giving the early fossil record of sponges a squeeze. Biol. Rev. 89, 972–1004 (2014).
Chang, S., Zhang, L., Clausen, S., Bottjer, D. J. & Feng, Q. The Ediacaran-Cambrian rise of siliceous sponges and development of modern oceanic ecosystems. Precambrian Res. 333, 105438 (2019).
Xiao, S., Hu, J., Yuan, X., Parsley, R. L. & Cao, R. Articulated sponges from the Lower Cambrian Hetang Formation in southern Anhui, South China: their age and implications for the early evolution of sponges. Palaeogeogr. Palaeoclimatol. Palaeoecol. 220, 89–117 (2005).
Bobrovskiy, I. et al. Ancient steroids establish the Ediacaran fossil Dickinsonia as one of the earliest animals. Science 361, 1246–1249 (2018).
Love, G. D. et al. Fossil steroids record the appearance of Demospongiae during the Cryogenian period. Nature 457, 718–721 (2009).
Zumberge, J. A. et al. Demosponge steroid biomarker 26-methylstigmastane provides evidence for Neoproterozoic animals. Nat. Ecol. Evol. 2, 1709–1714 (2018).
Zumberge, J. A., Rocher, D. & Love, G. D. Free and kerogen-bound biomarkers from late Tonian sedimentary rocks record abundant eukaryotes in mid-Neoproterozoic marine communities. Geobiology 18, 326–347 (2020).
Brocks, J. J. et al. The rise of algae in Cryogenian oceans and the emergence of animals. Nature 548, 578–581 (2017).
Brocks, J. J. The transition from a cyanobacterial to algal world and the emergence of animals. Emerg. Top. Life Sci. 2, 181–190 (2018).
Brocks, J. J. et al. Early sponges and toxic protists: possible sources of cryostane, an age diagnostic biomarker antedating Sturtian Snowball Earth. Geobiology 14, 129–149 (2016).
Antcliffe, J. B. The oldest compelling evidence for sponges is still early Cambrian in age - Reply to Love and Summons (2015). Palaeontology 58, 1137–1139 (2015).
Botting, J. P. & Muir, L. A. Early sponge evolution: a review and phylogenetic framework. Palaeoworld 27, 1–29 (2018).
Botting, J. P. & Nettersheim, B. J. Searching for sponge origins. Nat. Ecol. Evol. 2, 1685–1686 (2018).
Nettersheim, B. J. et al. Putative sponge biomarkers in unicellular Rhizaria question an early rise of animals. Nat. Ecol. Evol. 3, 577–581 (2019).
Hallmann, C. et al. Reply to: Sources of C30 steroid biomarkers in Neoproterozoic–Cambrian rocks and oils. Nat. Ecol. Evol. 4, 37–39 (2020).
Love, G. D. et al. Sources of C30 steroid biomarkers in Neoproterozoic–Cambrian rocks and oils. Nat. Ecol. Evol. 4, 34–46 (2020).
Urry, W. H., Stacey, F. W., Huyser, E. S. & Juveland, O. O. The peroxide- and light-induced additions of alcohols to olefins. J. Am. Chem. Soc. 76, 450–455 (1954).
Summons, R. E. & Capon, R. J. Identification and significance of 3β-ethyl steranes in sediments and petroleum. Geochim. Cosmochim. Acta 55, 2391–2395 (1991).
Summons, R. E. & Capon, R. J. Fossil steranes with unprecedented methylation in ring-A. Geochim. Cosmochim. Acta 52, 2733–2736 (1988).
Alexander, R., Berwick, L. & Pierce, K. Single carbon surface reactions of 1-octadecene and 2,3,6-trimethylphenol on activated carbon: implications for methane formation in sediments. Org. Geochem. 42, 540–547 (2011).
Given, P. H. & Hill, L. W. Catalysis of the isomerisation and polymerisation of olefins on carbon blacks. Carbon 6, 525–535 (1968).
Meier, J. A. & Hill, L. W. Carbon black catalyzed olefin isomerization: a heterogeneous site model based on rate dependence on catalyst concentration. J. Catal. 87, 80–87 (1974).
Alexander, R., Dawson, D., Pierce, K. & Murray, A. Carbon catalysed hydrogen exchange in petroleum source rocks. Org. Geochem. 40, 951–955 (2009).
Hoshino, Y. et al. Cryogenian evolution of stigmasterol biosynthesis. Sci. Adv. 3, e1700887 (2017).
Butterfield, N. J., Knoll, A. H. & Swett, K. A bangiophyte red alga from the proterozoic of arctic Canada. Science 250, 104–107 (1990).
Idler, D. R., Saito, A. & Wiseman, P. Sterols in red algae (Rhodophyceae). Steroids 11, 465–473 (1968).
Fattorusso, E. et al. Sterols of some red algae. Phytochemistry 14, 1579–1582 (1975).
Adam, P., Philippe, E. & Albrecht, P. Photochemical sulfurization of sedimentary organic matter: a widespread process occurring at early diagenesis in natural environments? Geochim. Cosmochim. Acta 62, 265–271 (1998).
Rubinstein, B. I., Sieskind, O. & Albrecht, P. Rearranged sterenes in a shale: occurrence and simulated formation. Org. Geochem. 11, 1973–1976 (1975).
Dahl, J. E., Moldowan, J. M., McCaffrey, M. A. & Lipton, P. A. A new class of natural products revealed by 3β-alkyl steranes in petroleum. Nature 355, 472–475 (1992).
McCaffrey, M. A. et al. Paleoenvironmental implications of novel C30 steranes in Precambrian to Cenozoic Age petroleum and bitumen. Geochim. Cosmochim. Acta 58, 529–532 (1994).
Bobrovskiy, I. et al. Algal origin of sponge sterane biomarkers negates the oldest evidence for animals in the rock record. Nat. Ecol. Evol. https://doi.org/10.1038/s41559-020-01334-7 (2020).
Xiao, S. & Laflamme, M. On the eve of animal radiation: phylogeny, ecology and evolution of the Ediacara biota. Trends Ecol. Evol. 24, 31–40 (2008).
van Maldegem, L. M. et al. Bisnorgammacerane traces predatory pressure and the persistent rise of algal ecosystems after Snowball Earth. Nat. Commun. 10, 476 (2019).
Hallmann, C., Kelly, A. E., Gupta, S. N. & Summons, R. E. in Quantifying the Evolution of Early Life: Numerical Approaches to the Evaluation of Fossils and Ancient Ecosystems Vol. 36 (eds Laflamme, M. et al.) 355–401 (Springer, 2011).
van Maldegem, L. M. Molecular and Isotopic Signatures of Life Surrounding the Neoproterozoic Snowball Earth Events. PhD thesis, Univ. Bremen (2017); https://media.suub.uni-bremen.de/handle/elib/1535
Adam, P., Schaeffer, P. & Brocks, J. J. Synthesis of 26-methyl cholestane and identification of cryostanes in mid-Neoproterozoic sediments. Org. Geochem. 115, 246–249 (2018).
Sousa Júnior, G. R. et al. Organic matter in the Neoproterozoic cap carbonate from the Amazonian Craton, Brazil. J. S. Am. Earth Sci. 72, 7–24 (2016).
Dahl, J. E. et al. Extended 3β-alkyl steranes and 3-alkyl triaromatic steroids in crude oils and rock extracts. Geochim. Cosmochim. Acta 59, 3717–3729 (1995).
Weiss, H. et al. The Norwegian Industry Guide to Organic Geochemical Analyses 4th edn (Norsk Hydro, Statoil, Geolab Nor, SINTEF Petroleum Research and the Norwegian Petroleum Directorate, 2000).
Schaeffer, P., Fache-Dany, F., Trendel, J. M. & Albrecht, P. Polar constituents of organic matter rich marls from evaporitic series of the Mulhouse basin. Org. Geochem. 20, 1227–1236 (1993).
Parfrey, L. W., Lahr, D. J. G., Knoll, A. H. & Katz, L. A. Estimating the timing of early eukaryotic diversification with multigene molecular clocks. Proc. Natl Acad. Sci. USA 108, 13624–13629 (2011).
Erwin, D. H. et al. The Cambrian conundrum: early divergence and later ecological success in the early history of animals. Science 334, 1091–1097 (2011).
Brocks, J. J. et al. Biomarker evidence for green and purple sulphur bacteria in a stratified Palaeoproterozoic sea. Nature 437, 866–870 (2005).
Tang, Q., Pang, K., Yuan, X. & Xiao, S. A one-billion-year-old multicellular chlorophyte. Nat. Ecol. Evol. 4, 543–549 (2020).
Acknowledgements
We thank R. Tarozo and P. Pringle for laboratory support, I. Bobrovskiy for discussions and helpful comments on the manuscript, P. Sansjofre for sharing Araras Group samples, the National Park Service (GRCA-00645) for permission to sample the Chuar Group and G. Love for providing a 26-mes reference sample. This work was funded by the Max-Planck-Society and the Deutsche Forschungsgemeinschaft (Research Center/Cluster of Excellence 309: MARUM - Center for Marine Environmental Sciences). We further acknowledge the French National Research Agency (CNRS; P.S. and P.A.) and the Australian Research Council (grant nos. DP1095247 and DP160100607 to J.J.B.).
Author information
Authors and Affiliations
Contributions
L.M.v.M., B.J.N. and C.H. designed the research, analysed all geochemical data and wrote the manuscript with input from A.L., J.J.B., P.S. and P.A., who also assisted with interpretation. L.M.v.M. conducted the geological analyses. B.J.N. and A.L. performed pyrolysis experiments on modern sterols. P.A. and P.S. synthesized standards of 3-alkylsteranes and 26-mec.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Discussion, Figs. 1–5, Tables 1–4 and refs. 1–21.
Rights and permissions
About this article
Cite this article
van Maldegem, L.M., Nettersheim, B.J., Leider, A. et al. Geological alteration of Precambrian steroids mimics early animal signatures. Nat Ecol Evol 5, 169–173 (2021). https://doi.org/10.1038/s41559-020-01336-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-020-01336-5
This article is cited by
-
Lost world of complex life and the late rise of the eukaryotic crown
Nature (2023)
-
Sterol methyltransferases in uncultured bacteria complicate eukaryotic biomarker interpretations
Nature Communications (2023)
-
Current understanding on the Cambrian Explosion: questions and answers
PalZ (2021)
-
Late Cretaceous paleoclimate and paleoenvironment in the Songliao Basin, China
Journal of Paleolimnology (2021)
-
Algal origin of sponge sterane biomarkers negates the oldest evidence for animals in the rock record
Nature Ecology & Evolution (2020)