Abstract
Evolutionary ecologists often seek to identify the mechanisms maintaining intraspecific variation. In social animals, whole groups can exhibit between-group differences in their collective traits. We examined whether negative frequency-dependent selection (that is, a rare-type advantage) could help to maintain between-group variation. We engineered neighbourhoods of social spider colonies bearing bold or shy foraging phenotypes and monitored their fecundity in situ. We found that bold colonies enjoyed a rare-type advantage that is lost as the frequency of bold colonies in a neighbourhood increases. The success of shy colonies was not frequency dependent. These dynamics seem to be driven by a foraging advantage of bold colonies that is lost in bold neighbourhoods because prey become scarce, and shy colonies perform better than bold colonies under low-resource conditions. Thus, to understand selection on collective traits, it is insufficient to examine groups in isolation. The phenotypic environment in which groups reside and compete must also be considered.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
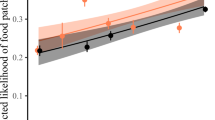
The data used in this study are available from Dryad (https://doi.org/10.5061/dryad.m592p4g). Raw data are depicted in Fig. 3b,c and Supplementary Figs. 1–6.
Change history
18 February 2020
Editorial Note: Readers are alerted that the reliability of some of the data presented in this Article is currently in question and is being investigated by the editors. A further editorial response will follow when the issues are resolved.
References
Bell, A. M., Hankison, S. J. & Laskowski, K. L. The repeatability of behaviour: a meta-analysis. Anim. Behav. 77, 771–783 (2009).
Bolnick, D. I. et al. The ecology of individuals: incidence and implications of individual specialization. Am. Nat. 161, 1–28 (2003).
Bolnick, D. I. et al. Why intraspecific trait variation matters in community ecology. Trends Ecol. Evol. 26, 183–192 (2011).
Jandt, J. M. et al. Behavioral syndromes and social insects: multiple levels of personality. Biol. Rev. 89, 48–67 (2014).
Bengston, S. & Jandt, J. M. The development of collective personality: the ontogenetic drivers of behavioral variation across groups. Front. Ecol. Evol. 2, 81 (2014).
Brown, C. R., Brown, M. B., Roche, E. A., O’Brien, V. A. & Page, C. E. Fluctuating survival selection explains variation in avian group size. Proc. Natl Acad. Sci. USA 113, 5113–5118 (2016).
Ingram, K. K., Pilko, A., Heer, J. & Gordon, D. M. Colony life history and lifetime reproductive success of red harvester ant colonies. J. Anim. Ecol. 82, 540–550 (2013).
Gordon, D. M. The rewards of restraint in the collective regulation of foraging by harvester ant colonies. Nature 498, 91–93 (2013).
Wray, M. K., Mattila, H. R. & Seeley, T. D. Collective personalities in honeybee colonies are linked to colony fitness. Anim. Behav. 81, 559–568 (2011).
Kassen, R. The experimental evolution of specialists, generalists, and the maintenance of diversity. J. Evol. Biol. 15, 173–190 (2002).
Sinervo, B. & Calsbeek, R. The developmental, physiological, neural, and genetical causes and consequences of frequency-dependent selection in the wild. Annu. Rev. Ecol. Evol. Syst. 37, 581–610 (2006).
Maynard Smith, J. Evolution and the Theory of Games (Cambridge Univ. Press, 1982).
Sinervo, B. & Lively, C. M. The rock-paper-scissors game and the evolution of alternative male strategies. Nature 380, 240–243 (1996).
Gigord, L. D. B., Macnair, M. R. & Smithson, A. Negative frequency-dependent selection maintains a dramatic flower color polymorphism in the rewardless orchid Dactylorhiza sambucina (L.) Soò. Proc. Natl Acad. Sci. USA 98, 6253–6255 (2001).
KeiserC. N. & PruittJ. N. Personality composition is more important than group size in determining collective foraging behaviour in the wild. Proc. R. Soc. B 281, 20141424 (2014).
Wright, C. M., Keiser, C. N. & Pruitt, J. N. Colony personality composition alters colony-level plasticity and magnitude of defensive behaviour in a social spider. Anim. Behav. 115, 175–183 (2016).
Pruitt, J. N. et al. Selection for collective aggressiveness favors social susceptibility in social spiders. Curr. Biol. 28, 100–105 (2018).
Smith, D., van Rijn, S., Henschel, J., Bilde, T. & Lubin, Y. Amplified fragment length polymorphism fingerprints support limited gene flow among social spider populations. Biol. J. Linn. Soc. 97, 235–246 (2009).
Modlmeier, A. P. et al. Persistent social interactions beget more pronounced personalities in a desert-dwelling social spider. Biol. Lett. 10, 20140419 (2014).
Hunt, E. R. et al. Social interactions shape individual and collective personality in social spiders. Proc. R. Soc. B 285, 30185649 (2018).
Pruitt, J. N. & Pinter-Wollman, N. The legacy effects of keystone individuals on collective behaviour scale to how long they remain within a group. Proc. R. Soc. B 282, 89–96 (2015).
Johannesen, J., Hennig, A., Dommermuth, B. & Schneider, J. M. Mitochondrial DNA distributions indicate colony propagation by single matri-lineages in the social spider Stegodyphus dumicola (Eresidae). Biol. J. Linn. Soc. 76, 591–600 (2002).
Pyke, G. H., Pulliam, H. R. & Charnov, E. L. Optimal foraging - selective review of theory and tests. Q. Rev. Biol. 52, 137–154 (1977).
Tilman, D. Resource Competition and Community Structure (Princeton Univ. Press, 1982).
Rueffler, C., Van Dooren, T. J. M., Leimar, O. & Abrams, P. A. Disruptive selection and then what? Trends Ecol. Evol. 21, 238–245 (2006).
Lichtenstein, J. L. L. et al. Participation in cooperative prey capture and the benefits gained from it are associated with individual personality. Curr. Zool. 63, 561–567 (2017).
Yip, E. C., Powers, K. S. & Aviles, L. Cooperative capture of large prey solves scaling challenge faced by spider societies. Proc. Natl Acad. Sci. USA 105, 11818–11822 (2008).
Trubl, P., Blackmore, V. & Johnson, J. C. Wasteful killing in urban black widows: gluttony in response to food abundance. Ethology 117, 236–245 (2011).
Riechert, S. E. & Maupin, J. L. Spider effects on prey: tests for superfluous killing in five web-builders. In Proceedings of the 17th European Colloquium of Arachnology (ed. Selden, P. A.) 203–210 (British Arachnological Society, 1997).
Maupin, J. L. & Riechert, S. E. Superfluous killing in spiders: a consequence of adaptation to food-limited environments? Behav. Ecol. 12, 569–576 (2001).
Jolles, J. W., Laskowski K. L., Boogert N. J. & Manica, A. Repeatable group differences in the collective behaviour of stickleback shoals across ecological contexts. Proc. R. Soc. B 285, 29436496 (2018).
Farine, D. R., Aplin, L. M., Garroway, C. J., Mann, R. P. & Sheldon, B. C. Collective decision making and social interaction rules in mixed-species flocks of songbirds. Anim. Behav. 95, 173–182 (2014).
Jolles, J. W., Boogert, N. J., Sridhar, V. H., Couzin, I. D. & Manica, A. Consistent individual differences drive collective behavior and group functioning of schooling fish. Curr. Biol. 27, 2862 (2017).
Shaffer, Z. et al. The foundress’s dilemma: group selection for cooperation among queens of the harvester ant, Pogonomyrmex californicus. Sci. Rep. 6, 29828 (2016).
Haney, B. R. & Fewell, J. H. Ecological drivers and reproductive consequences of non-kin cooperation by ant queens. Oecologia 187, 643–655 (2018).
Pruitt, J. N. & Goodnight, C. J. Site-specific group selection drives locally adapted colony compositions. Nature 28, 1248–1256 (2014).
Keiser, C. N., Jones, D. K., Modlmeier, A. P. & Pruitt, J. N. Exploring the effects of individual traits and within-colony variation on task differentiation and collective behavior in a desert social spider. Behav. Ecol. Sociobiol. 68, 839–850 (2014).
Laskowski, K. L., Montiglio, P. O. & Pruitt, J. N. Individual and group performance suffers from social niche disruption. Am. Nat. 187, 776–785 (2016).
Laskowski K. L. & Pruitt J. N. Evidence of social niche construction: persistent and repeated social interactions generate stronger personalities in a social spider. Proc. R. Soc. B 281, 24671972 (2014).
Wright, C. M., Keiser, C. N. & Pruitt, J. N. Personality and morphology shape task participation, collective foraging and escape behaviour in the social spider Stegodyphus dumicola. Anim. Behav. 105, 47–54 (2015).
Keiser, C. N., Wright, C. M. & Pruitt, J. N. Increased bacterial load can reduce or negate the effects of keystone individuals on group collective behaviour. Anim. Behav. 114, 211–218 (2016).
Keiser C. N., Howell K. A., Pinter-Wollman N. & Pruitt J. N . Personality composition alters the transmission of cuticular bacteria in social groups. Biol. Lett. 12, 27381885 (2016).
Keiser, C. N. et al. Individual differences in boldness influence patterns of social interactions and the transmission of cuticular bacteria among group-mates. Proc. R. Soc. B 283, 27097926 (2016).
Wickler, W. & Seibt, U. Pedogenetic sociogenesis via the sibling-route and some consequences for Stegodyphus spiders. Ethology 95, 1–18 (1993).
Modlmeier, A. P. et al. Persistent social interactions beget more pronounced personalities in a desert-dwelling social spider. Biol. Lett. 10, 2014 (2014).
Grinsted, L., Pruitt, J. N., Settepani, V. & Bilde, T. Individual personalities shape task differentiation in a social spider. Proc. R. Soc. B 280, 23902907 (2013).
Pruitt, J. N., Grinsted, L. & Settepani, V. Linking levels of personality: personalities of the ‘average’ and ‘most extreme’ group members predict colony-level personality. Anim. Behav. 86, 391–399 (2013).
Christenson, T. E. Behavior of colonial and solitary spiders of the theridiid species Anelosimus eximius. Anim. Behav. 32, 725 (1984).
Kullmann, E. J. Evolution of social behavior in spiders (Araneae; Eresidae and Theridiidae). Am. Zool. 12, 419 (1972).
Seibt, U. & Wickler, W. Bionomics and social structure of ‘family spiders’ of the genus Stegodyphus, with special reference to the African species S. Dumicola and S. Mimosarum (Araneidae, Eresidae). Verh. Naturwiss. Ver. Hamb. 30, 255–303 (1988).
Acknowledgements
We thank A. Santoro, E. Eliason, H. Moeller, D. Fisher, A. Little and A. Radford for their help in improving previous versions of this manuscript. Funding was provided by NSF IOS grant numbers 1455895 to J.N.P., 1456010 to N.P.W., NIH GM115509 to N.P.W. and J.N.P., and the Tricounsel Agencies of Canada in association with a Canada 150 Research Chair Professorship to J.N.P.
Author information
Authors and Affiliations
Contributions
All authors contributed to study design, data collection, statistical analysis, and composing of the manuscript. All authors were included in all aspects of the pipeline.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Figures 1–6 and Supplementary Statistical Outputs
Rights and permissions
About this article
Cite this article
Pruitt, J.N., McEwen, B.L., Cassidy, S.T. et al. Experimental evidence of frequency-dependent selection on group behaviour. Nat Ecol Evol 3, 702–707 (2019). https://doi.org/10.1038/s41559-019-0852-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-019-0852-z