Abstract
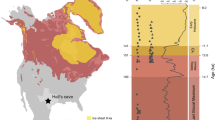
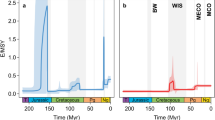
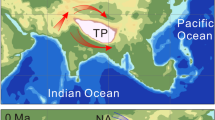
The Carboniferous–Permian transition (CPT) was Earth’s last pre-Quaternary icehouse–greenhouse transition, recording major shifts in late Palaeozoic climate regimes and increased continental seasonality over approximately 40 Myr. Its parallels to Quaternary climate change have inspired recent investigations into the impacts of purported rainforest collapse on palaeotropical vertebrate diversity, but little is known about how the protracted spatial dynamics of this transition impacted the emergence of modern tetrapod lineages. Here, we apply ecological ordinance analyses on a dataset of 286 CPT fossil vertebrate localities binned across four physiographic regions forming a palaeoequatorial transect. Our results clarify the spatiotemporal expansion of land-living vertebrates, demonstrating that the reduction of tropical wetlands accommodated emerging dryland-adapted amniote faunas from a western Pangaean epicentre. We call this west–east lag the ‘Vaughn–Olson model’: CPT climatic transitions were regionally diachronous with delayed proliferation of amniote-dominated dryland assemblages in the east. By combining our ecological analyses with a phylogenetic approach, we demonstrate that this pattern also applies to some co-occurring total-group amphibians, suggesting that there was pervasive selection for such dryland adaptations across the crown tetrapod tree, in contrast with stem tetrapods and ‘fishes’.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Detailed materials and methods, supplementary display items and source data are available in the Supplementary Information. Additional information, including ‘R’ scripts, may be obtained from the corresponding authors upon reasonable request.
References
Tabor, N. J. & Poulson, C. J. Palaeoclimate across the Late Pennsylvanian–Early Permian tropical palaeolatitudes: a review of climate indicators, their distribution, and relation to palaeophysiographic climate indicators. Palaeogeogr. Palaeoclimatol. Palaeoecol. 268, 293–310 (2008).
Montañez, I. P. et al. CO2-forced climate and vegetation instability during the late Paleozoic deglaciation. Science 315, 87–91 (2007).
DiMichele, W. A., Cecil, C. B., Montañez, I. P. & Falcon-Lang, H. J. Cyclic changes in Pennsylvanian paleoclimate and effects on floristic dynamics in tropical Pangaea. Int. J. Coal Geol. 83, 329–344 (2010).
Sahney, S., Benton, M. J. & Falcon-Lang, H. Rainforest collapse triggered Carboniferous tetrapod diversification in Euramerica. Geology 38, 1079–1082 (2010).
Anderson, J. S., Reisz, R. R., Scott, D., Fröbisch, N. B. & Sumida, S. S. A stem batrachian from the Early Permian of Texas and the origin of frogs and salamanders. Nature 453, 515–518 (2008).
Bever, G. S., Lyson, T. R., Field, D. J. & Bhullar, B.-A. S. Evolutionary origin of the turtle skull. Nature 525, 239–242 (2015).
Pardo, J. D., Small, B. J. & Huttenlocker, A. K. Stem caecilian from the Triassic of Colorado sheds light on the origins of Lissamphibia. Proc. Natl Acad. Sci. USA 114, E5389–E5395 (2017).
Pardo, J. D., Szostakiwskyj, M., Ahlberg, P. E. & Anderson, J. S. Hidden morphological diversity among early tetrapods. Nature 546, 642–645 (2017).
Anderson, P. S., Friedman, M. & Ruta, M. Late to the table: diversification of tetrapod mandibular biomechanics lagged behind the evolution of terrestriality. Integr. Comp. Biol. 53, 197–208 (2013).
Vaughn, P. P. Comparison of the Early Permian vertebrate faunas of the Four Corners region and north-central Texas. Contrib. Sci. (Los Angel. Calif.) 105, 1–13 (1966).
Vaughn, P. P. Lower Permian vertebrates of the Four Corners and the midcontinent as indices of climatic differences. In Proc. North American Paleontological Convention, 388–408 (Allen Press, Lawrence, KS, USA, 1969).
Olson, E. C. & Vaughn, P. P. The changes of terrestrial vertebrates and climates during the Permian of North America. Forma Functio 3, 113–138 (1970).
Sahney, S. & Benton, M. J. Recovery from the most profound mass extinction of all time. Proc. R. Soc. B 275, 759–765 (2008).
Brocklehurst, N., Day, M. O., Rubidge, B. S. & Fröbisch, J. Olson’s extinction and the latitudinal biodiversity gradient of tetrapods in the Permian. Proc. R. Soc. B 284, 20170231 (2017).
Dunne, E. M. et al. Diversity changes during the rise of tetrapods and the impact of the ‘Carboniferous rainforest collapse’. Proc. R. Soc. B 285, 20172730 (2018).
Benton, M. J. No gap in the Middle Permian record of terrestrial vertebrates. Geology 40, 339–342 (2012).
Barton, P. S. et al. The spatial scaling of beta diversity. Glob. Ecol. Biogeogr. 22, 639–647 (2012).
Molnar, J. L., Diogo, R., Hutchinson, J. R. & Pierce, S. E. Evolution of hindlimb muscle anatomy across the tetrapod water‐to‐land transition, including comparisons with forelimb anatomy. Anat. Rec. https://doi.org/10.1002/ar.23997 (2018).
Müller, J. & Tsuji, L. A. Impedance-matching hearing in Paleozoic reptiles: evidence of advanced sensory perception at an early stage of amniote evolution. PLoS ONE 2, e889 (2007).
McCahon, T. J. & Miller, K. B. Environmental significance of lungfish burrows (Gnathorhiza) within Lower Permian (Wolfcampian) paleosols of the US midcontinent. Palaeogeogr. Palaeoclimatol. Palaeoecol. 435, 1–12 (2015).
Eberth, D. A., Berman, D. S., Sumida, S. S. & Hopf, H. Lower Permian terrestrial paleoenvironments and vertebrate paleoecology of the Tambach Basin (Thuringia, Central Germany): the upland holy grail. Palaios 15, 293–313 (2000).
MacDougall, M. J., Tabor, N. J., Woodhead, J., Daoust, A. R. & Reisz, R. R. The unique preservational environment of the Early Permian (Cisuralian) fossiliferous cave deposits of the Richards Spur locality, Oklahoma. Palaeogeogr. Palaeoclimatol. Palaeoecol. 475, 1–11 (2017).
Chao, A. & Jost, L. Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93, 2533–2547 (2012).
Sørensen, T. A method of establishing groups of equal amplitude in plant sociology based on similarity of species and its application to analyses of the vegetation on Danish commons. Biol. Skr. 5, 1–34 (1948).
Bapst, D. W. paleotree: an R package for paleontological and phylogenetic analyses of evolution. Methods Ecol. Evol. 3, 803–807 (2012).
Revell, L. J. phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
Fredericksen, N. O. The rise of the mesophytic flora. Geosci. Man 4, 17–28 (1972).
Knoll, A. H. in Extinctions (ed. Nitecki, M.) 21–65 (Univ. Chicago Press, Chicago, 1984).
Rowley, D. B. et al. Carboniferous paleogeographic, phytogeographic, and paleoclimatic reconstructions. Int. J. Coal Geol. 5, 7–42 (1985).
Cleal, C. J. & Thomas, B. A. Tectonics, tropical forest destruction and global warming in the late Palaeozoic. Acta Palaeobotanica 2, 17–19 (1999).
Cleal, C. J. & Thomas, B. A. Palaeozoic tropical rainforests and their effect on global climates: is the past the key to the present? Geobiology 3, 13–31 (2005).
DiMichele, W. A., Pfefferkorn, H. W. & Gastaldo, R. A. Response of Late Carboniferous and Early Permian plant communities to climate change. Annu. Rev. Earth Planet. Sci. 29, 461–487 (2001).
Tabor, N. J. et al. Paleosol archives of environmental and climatic history in paleotropical western Pangea during the latest Pennsylvanian through Early Permian. In Resolving the Late Paleozoic Ice Age in Time and Space 291–303 (Special Paper 441, Geological Society of America, 2008).
Milner, A. R. & Panchen, A. L. in Implications of Continental Drift to the Earth Sciences Vol. 1 (eds Tarling, D. H. & Runcorn, S. K.) 353–368 (Academic Press, London, 1973).
Milner, A. R. in Paleozoic Vertebrate Biostratigraphy and Biogeography (ed. Long, J. A.) 324–353 (Belhaven Press, London, 1993).
Cisneros, J. C. et al. New Permian fauna from tropical Gondwana. Nat. Commun. 6, 8676 (2015).
Badgley, C. et al. Ecological changes in Miocene mammalian record show impact of prolonged climatic forcing. Proc. Natl Acad. Sci. USA 105, 12145–12149 (2008).
Fraser, D., Gorelick, R. & Rybczynski, N. Macroevolution and climate change influence phylogenetic community assembly of North American hoofed mammals. Biol. J. Linn. Soc. 114, 485–494 (2015).
Brennan, I. G. & Oliver, P. M. Mass turnover and recovery dynamics of a diverse Australian continental radiation. Evolution 71, 1352–1365 (2017).
Schoch, R. R. & Fröbisch, N. B. Metamorphosis and neoteny: alternative pathways in an extinct amphibian clade. Evolution 60, 1467–1475 (2006).
Reisz, R. R., Schoch, R. R. & Anderson, J. S. The armoured dissorophid cacops from the Early Permian of Oklahoma and the exploitation of the terrestrial realm by amphibians. Naturwissenschaften 96, 789–796 (2009).
Moen, D. S., Irschick, D. J. & Wiens, J. J. Evolutionary conservatism and convergence both lead to striking similarity in ecology, morphology and performance across continents in frogs. Proc. R. Soc. B 280, 20132156 (2013).
Mueller, R. L., Macey, J. R., Jaekel, M., Wake, D. B. & Boore, J. L. Morphological homoplasy, life history evolution, and historical biogeography of plethodontid salamanders inferred from complete mitochondrial genomes. Proc. Natl Acad. Sci. USA 101, 13820–13825 (2004).
Bemis, W. E. Convergent evolution of jaw-opening muscles in lepidosirenid lungfishes and tetrapods. Can. J. Zool. 65, 2814–2817 (1987).
Diogo, R. Comparative anatomy, homologies, and evolution of mandibular, hyoid, and hypobranchial muscles of bony fish and tetrapods: a new insight. Anim. Biol. 58, 123–172 (2008).
Anderson, J. S., Pardo, J. D., Maddin, H. C., Szostakiwskyj, M. & Tinius, A. Is there an exemplar taxon for modelling the evolution of early tetrapod hearing? Proc. R. Soc. B 283, 20160027 (2016).
Oksanen J. et al. vegan: Community Ecology Package R package version 2.4-5 (2017); https://CRAN.R-project.org/package=vegan
Maddison, W. P. & Maddison D. R. Mesquite: a Modular System for Evolutionary Analysis Version 3.40 (2018); http://mesquiteproject.org
Coates, M. I., Gess, R. W., Finarelli, J. A., Criswell, K. E. & Tietjen, K. A symmoriiform chondrichthyan braincase and the origin of chimaeroid fishes. Nature 541, 208–211 (2017).
Giles, S., Xu, G. H., Near, T. J. & Friedman, M. Early members of ‘living fossil’ lineage imply later origin of modern ray-finned fishes. Nature 549, 265–268 (2017).
Blomberg, S. P., Garland, T. Jr & Ives, A. R. Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57, 717–745 (2003).
Acknowledgements
We thank K. Angielczyk, C. Sidor and S. Sumida for support and helpful discussions during the inception of this project. D. Berman and A. Henrici (Carnegie Museum) and R. Cifelli and J. Person (Sam Noble Oklahoma Museum of Natural History) permitted access to useful collections records. We also thank the Paleobiology Database community and its major data contributors: J. Alroy, A. K. Behrensmeyer, R. Butler and J. Müller. J. Calede, K. Hollis, J. Marcot, K. Middleton and D. Vilhena assisted with preliminary analyses. We also thank M. Barton, J. Benca, M.-E. Benson, A. Bormet, J. Calede, K. Hollis, R. Hook, R. Irmis, E. Leckey, S. Lucas, J. Marcot, W. May, B. Peecook, J. Peters, L. Powers, K. Sears, D. Smith, N. Tabor and C. Wellstead for additional discussions and suggestions for improving this study. Finally, we thank R. Butler and E. Dunne for thoughtful reviews. A.K.H. has been supported by the University of Southern California and US Bureau of Land Management’s National Conservation Lands grants programme (number L17AC00064).
Author information
Authors and Affiliations
Contributions
J.D.P. and A.K.H. collected and analysed the data. A.R.M. assisted with the European records. J.D.P. rendered the graphical illustrations. J.D.P. and A.K.H. composed the figures. All authors contributed to writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplemental text describing data collection and analysis, and relevant references. Supplementary Figure 1: Comparison of diversity estimates across the CPT. a, Stage-specific rarefaction curves. b, Estimated Chao diversity across the CPT. c, Scatterplot of time of first occurrence of families versus mean PCO1 value. d, Scatterplot of time of last occurrence of families versus mean PCO1 value, showing weak negative trend. Supplementary Figure 2: Tree topology used in the comparative phylogenetic analysis. Node numbers represent node identifiers listed in the data sheet ‘Node Minimum Constraints’. Supplementary Figure 3: Time-calibrated phylogeny assembled for comparative phylogenetic analysis. Node minimum divergence constraints are listed in the online Supplementary Dataset 1 (numbers represent node identifiers from Supplementary Figure 2 and listed in the data sheet ‘Node Minimum Constraints’)
Dataset 1
Source data for ecological ordinance analyses, including presence–absence matrix
Dataset 2
Composite tree used for phylogenetic analyses in NEXUS format
Rights and permissions
About this article
Cite this article
Pardo, J.D., Small, B.J., Milner, A.R. et al. Carboniferous–Permian climate change constrained early land vertebrate radiations. Nat Ecol Evol 3, 200–206 (2019). https://doi.org/10.1038/s41559-018-0776-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0776-z
This article is cited by
-
Predatory synapsid ecomorphology signals growing dynamism of late Palaeozoic terrestrial ecosystems
Communications Biology (2024)
-
The rise of predation in Jurassic lampreys
Nature Communications (2023)
-
Mechanistic neutral models show that sampling biases drive the apparent explosion of early tetrapod diversity
Nature Ecology & Evolution (2023)
-
An early Pangaean vicariance model for synapsid evolution
Scientific Reports (2020)
-
The phylogeny of early amniotes and the affinities of Parareptilia and Varanopidae
Nature Ecology & Evolution (2019)