Abstract
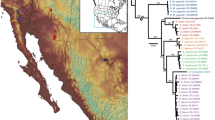
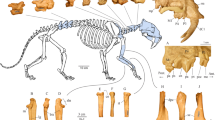
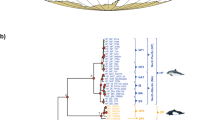
Understanding extinction events requires an unbiased record of the chronology and ecology of victims and survivors. The rhinoceros Elasmotherium sibiricum, known as the ‘Siberian unicorn’, was believed to have gone extinct around 200,000 years ago—well before the late Quaternary megafaunal extinction event. However, no absolute dating, genetic analysis or quantitative ecological assessment of this species has been undertaken. Here, we show, by accelerator mass spectrometry radiocarbon dating of 23 individuals, including cross-validation by compound-specific analysis, that E. sibiricum survived in Eastern Europe and Central Asia until at least 39,000 years ago, corroborating a wave of megafaunal turnover before the Last Glacial Maximum in Eurasia, in addition to the better-known late-glacial event. Stable isotope data indicate a dry steppe niche for E. sibiricum and, together with morphology, a highly specialized diet that probably contributed to its extinction. We further demonstrate, with DNA sequencing data, a very deep phylogenetic split between the subfamilies Elasmotheriinae and Rhinocerotinae that includes all the living rhinoceroses, settling a debate based on fossil evidence and confirming that the two lineages had diverged by the Eocene. As the last surviving member of the Elasmotheriinae, the demise of the ‘Siberian unicorn’ marked the extinction of this subfamily.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The four mitochondrial genome consensus sequences with coverage ≥80% and mean read depth ≥5× are available on GenBank (MH937513-MH937516). All consensus sequences, unmapped sequencing reads and phylogenetic analysis files associated with our ancient DNA work are available on Figshare (https://doi.org/10.25909/5ba34a40ba925). All the radiocarbon data generated at the ORAU and CIO are archived internally at the respective laboratories, and are available upon request. ORAU data are also available on the laboratory’s website, along with a link to the paper.
References
Fortelius, M. New and Old Worlds Database of Fossil Mammals (NOW) (Univ. Helsinki, 2017).
Antoine, P.-O. Phylogénie et évolution des Elasmotheriina (Mammalia, Rhinocerotidae). Mém. Mus. Natl Hist.Nat. 188, 1–359 (2002).
Becker, D., Antoine, P. O. & Maridet, O. A new genus of Rhinocerotidae (Mammalia, Perissodactyla) from the Oligocene of Europe. J. Syst. Palaeontol. 11, 947–972 (2013).
Cerdeño, E. Cladistic analysis of the family Rhinocerotidae (Perissodactyla). Am. Mus. Novit. 3143, 1–25 (1995).
Guérin, C. & Pickford, M. Ougandatherium napakense nov. gen. nov. sp., le plus ancien Rhinocerotidae Iranotheriinae d’Afrique. Ann. Paléontol. 89, 1–35 (2003).
Deng, T. & Zheng, M. Limb bones of Elasmotherium (Rhinocerotidae, Perissodactyla) from Nihewan (Hebei, China). Vert. PalAs. 43, 110–121 (2005).
Schvyreva, A. K. On the importance of the representatives of the genus Elasmotherium (Rhinocerotidae, Mammalia) in the biochronology of the Pleistocene of Eastern Europe. Quat. Int. 379, 128–134 (2015).
Kozamkulova, B. S. Elasmotherium sibiricum und sein Verbreitungsgebiet auf dem Territorium der UdSSR. Quartärpaläontologie 4, 85–91 (1981).
Kosintsev, P. A. in The Quaternary of the Urals: Global Trends and Pan-European Quaternary Records (eds Borodin, A. V., Markova, E. A. & Strukova, T. V.) 67–68 (UrFU, Ekaterinburg, 2014)..
Lister, A. M. & Stuart, A. J. Extinction chronology of the woolly rhinoceros Coelodonta antiquitatis: reply to Kuzmin. Quat. Sci. Rev. 62, 144–146 (2013).
Devièse, T., Comeskey, D., McCullagh, J., Bronk Ramsey, C. & Higham, T. New protocol for compound-specific radiocarbon analysis of archaeological bones. Rapid Commun. Mass Spectrom. 32, 373–379 (2018).
Bourrillon, R. et al. A new Aurignacian engraving from Abri Blanchard, France: implications forunderstanding Aurignacian graphic expression in Western and Central Europe. Quat. Int. 491, 46–64 (2018).
Devièse, T. et al. Direct dating of Neanderthal remains from the site of Vindija Cave and implications for themiddle to upper Paleolithic transition. Proc. Natl Acad. Sci. USA 114, 10606–10611 (2017).
Reynolds, N., Dinnis, R., Bessudnov, A. A., Devièse, T. & Higham, T. The Kostënki 18 child burial and thecultural and funerary landscape of mid upper Palaeolithic European Russia. Antiquity 91, 1435–1450 (2017).
Devièse, T. et al. Increasing accuracy for the radiocarbon dating of sites occupied by the first Americans. Quat. Sci. Rev. 198, 171–180 (2018).
Becerra-Valdivia, L. et al. Reassessing the chronology of the archaeological site of Anzick. Proc. Natl Acad. Sci. USA 115, 7000–7003 (2018).
Reimer, P. J. et al. Intcal13 and Marine13 radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 55, 1869–1887 (2013).
Bronk Ramsey, C. Recent and planned developments of the program OxCal. Radiocarbon 55, 720–730 (2013).
Bronk Ramsey, C. Bayesian analysis of radiocarbon dates. Radiocarbon 51, 337–360 (2009).
Kosintsev, P. A. Ural and Siberia Faunas at Pleistocene and Holocene Times (IPAE UB RAS, Chelyabinsk, 2005).
Vasil'ev, S. A. Faunal exploitation, subsistence practices and Pleistocene extinctions in Palaeolithic Siberia. Deinsea 9, 513–556 (2003).
Price, S. A. & Bininda-Emonds, O. R. P. A comprehensive phylogeny of extant horses, rhinos and tapirs (Perissodactyla) through data combination. Zoosystematics Evol. 85, 277–292 (2009).
Steiner, C. C. & Ryder, O. A. Molecular phylogeny and evolution of the Perissodactyla. Zool. J. Linn. Soc. 163, 1289–1303 (2011).
Meredith, R. W. et al. Impacts of the Cretaceous terrestrial revolution and KPg extinction on mammal diversification. Science 334, 521–524 (2011).
Heissig, K. The American genus Penetrigonias Tanner & Martin, 1976 (Mammalia: Rhinocerotidae) as a stem group elasmothere and ancestor of Menoceras Troxell, 1921. Zitteliana A 52, 79–95 (2012).
Boehme, M. et al. Na Duong (northern Vietnam)—an exceptional window into Eocene ecosystems from Southeast Asia. Zitteliana A 53, 120–167 (2013).
Shpansky, A. V., Ilyina, S. A. & Aliyasova, V. N. The Quaternary mammals from Kozhamzhar locality (Pavlodar Region, Kazakhstan). Am. J. Appl. Sci. 13, 189–199 (2016).
Reimer, P. J. & Svyatko, S. V. Comment on Shpansky et al. 2016, The Quaternary mammals fromKozhamzhar locality (Pavlodar region, Kazakhstan). Am. J. Appl. Sci. 13, 477–478 (2016).
Rasmussen, S. O. et al. A stratigraphic framework for abrupt climatic changes during the Last Glacial period based on three synchronized Greenland ice-core records: refining and extending the INTIMATE event stratigraphy. Quat. Sci. Rev. 106, 14–28 (2014).
Stuart, A. J. Late Quaternary megafaunal extinctions on the continents: a short review. Geol. J. 50, 338–363 (2015).
Stuart, A. J. & Lister, A. M . Patterns of late Quaternary megafaunal extinctions in Europe and northern Asia. Cour. Forsch. Inst. Senckenberg 259, 289–299 (2007).
Cooper, A. Abrupt warming events drove Late Pleistocene Holarctic megafaunal turnover. Science 349, 602–606 (2015).
Hughes, P. D. & Gibbard, P. L. A stratigraphical basis for the Last Glacial Maximum (LGM). Quat. Int. 383, 174–185 (2015).
Monegato, G., Scardia, G., Hajdas, I., Rizzini, F. & Piccin, A. The Alpine LGM in the boreal ice-sheets game. Sci. Rep. 7, 2078 (2017).
Higham, T. et al. The timing and spatiotemporal patterning of Neanderthal disappearance. Nature 512, 306–309 (2014).
Heintzman, P. D. et al. A new genus of horse from Pleistocene North America. eLife 6, e29944 (2017)..
Stuart, A. J. & Lister, A. M. Extinction chronology of the woolly rhinoceros Coelodonta antiquitatis in the context of late Quaternary megafaunal extinctions in northern Eurasia. Quat. Sci. Rev. 51, 1–17 (2012).
Stuart, A. J. & Lister, A. M. New radiocarbon evidence on the extirpation of the spotted hyaena (Crocuta crocuta (Erxl.)) in northern Eurasia. Quat. Sci. Rev. 96, 108–116 (2014).
Dinnis, R., Pate, A. & Reynolds, N. Mid-to-late Marine Isotope Stage 3 mammal faunas of Britain: a new look. Proc. Geol. Assoc. 127, 435–444 (2016).
Pacher, M. & Stuart, A. J. Extinction chronology and palaeobiology of the cave bear (Ursus spelaeus). Boreas 38, 189–206 (2009).
Bocherens, H. et al. The last of its kind? Radiocarbon, ancient DNA and stable isotope evidence from a late cave bear (Ursus spelaeus ROSENMÜLLER, 1794) from Rochedane (France). Quat. Int. 339–340, 179–188 (2014).
Stuart, A. J., Kosintsev, P. A., Higham, T. & Lister, A. M. Pleistocene to Holocene extinction dynamics in giant deer and woolly mammoth. Nature 431, 684–689 (2004).
Guillevic, M. et al. Evidence for a three-phase sequence during Heinrich stadial 4 using a multiproxy approach based on Greenland ice core records. Clim. Past 10, 2115–2133 (2014).
Hubberten, H. W. et al. The periglacial climate and environment in northern Eurasia during the Last Glaciation. Quat. Sci. Rev. 23, 1333–1357 (2004).
Boeskorov, G. G. et al. Woolly rhino discovery in the lower Kolyma River. Quat. Sci. Rev. 30, 2262–2272 (2011).
Rivals, F. & Lister, A. M. Dietary flexibility and niche partitioning of large herbivores through the Pleistocene of Britain. Quat. Sci. Rev. 146, 116–133 (2016).
Saarinen, J., Eronen, J., Fortelius, M., Seppä, H. & Lister, A. M. Patterns of diet and body mass of large ungulates from the Pleistocene of Western Europe, and their relation to vegetation. Palaeontol. Electron. 19, 1–58 (2016).
Pushkina, D., Bocherens, H. & Ziegler, R. Unexpected palaeoecological features of the middle and latePleistocene large herbivores in southwestern Germany revealed by stable isotopic abundances in tooth enamel. Quat. Int. 339-340, 164–178 (2014).
Zeuner, F. E. New reconstructions of the woolly rhinoceros and Merck’s rhinoceros. Proc. Linn. Soc. Lond. 156, 183–195 (1945).
Zhegallo, V. et al. On the fossil rhinoceros Elasmotherium (including the collections of the Russian Academy of Sciences). Cranium 22, 17–40 (2005).
Grichuk, V. P. Dynamics of Terrestrial Landscape Components and Inner Marine Basins of Northern Eurasia During the Last 130,000 Years (ed. Velichko, A. A.) 64–88 (GEOS, Moscow, 2002).
Bocherens, H. Isotopic biogeochemistry and the paleoecology of the mammoth steppe fauna. Deinsea 9, 57–76 (2003).
Jürgensen, J. et al. Diet and habitat of the saiga antelope during the late Quaternary using stable carbon and nitrogen isotope ratios. Quat. Sci. Rev. 160, 150–161 (2017).
Badeck, F.-W., Tcherkez, G., Nogués, S., Piel, C. & Ghashghaie, J. Post-photosynthetic fractionation of stable carbon isotopes between plant organs—a widespread phenomenon. Rapid Commun. Mass Spectrom. 19, 1381–1391 (2005).
Drucker, G. et al. Tracking possible decline of woolly mammoth during the Gravettian in Dordogne (France) and the Ach Valley (Germany) using multi-isotope tracking (13C, 14C, 15N, 34S, 18O). Quat. Int. 360, 304–317 (2015).
Drucker, D. G., Bocherens, H. & Péan, S. Isotopes stables (13C, 15N) du collagène des mammouths de Mezhyrich (Epigravettien, Ukraine): implications paléoécologiques. L'Anthropologie 118, 504–517 (2014).
Johnson, C.N. Determinants of loss of mammal species during the late Quaternary ‘megafauna’ extinctions: life history and ecology, but not body size. Proc. R. Soc. B 269, 2221–2227 (2009)..
Higham, T. et al. The timing and spatiotemporal patterning of Neanderthal disappearance. Nature 512, 306–309 (2014).
Mook, W. G. & Streurman, H. J. Physical and chemical aspects of radiocarbon dating. In Proc. 1st Symposium on C and Archaeology (eds Mook, W. G. & Waterbolk, H. T.) 31–55 (PACT, Groningen, 1983).
Wijma, S., Aerts, A. T., van der Plicht, J. & Zondervan, A. The Groningen AMS facility. Nucl. Instrum. Methods Phys. Res. B 113, 465–469 (1996).
Aerts-Bijma, A. T., Meijer, H. A. J. & van der Plicht, J. AMS sample handling in Groningen. Nucl. Instrum. Methods Phys. Res. B 123, 221–225 (1997).
Van der Plicht, J., Wijma, S., Aerts, A. T., Pertuisot, M. H. & Meijer, H. A. J. Status report: the Groningen AMS facility. Nucl. Instrum. Methods Phys. Res. B 172, 58–65 (2000).
Brock, F., Higham, T., Ditchfield, P. & Ramsey, C. B. Current pretreatment methods for AMS radiocarbon dating at the Oxford Radiocarbon Accelerator Unit (ORAU). Radiocarbon 52, 103–112 (2010).
Metcalf, J. L. et al. Synergistic roles of climate warming and human occupation in Patagonian megafaunal extinctions during the Last Deglaciation. Sci. Adv. 2, e1501682 (2016).
Turney, C. S. M., Jones, R. T., Thomas, Z. A., Palmer, J. G. & Brown, D. Extreme wet conditions coincident with Bronze Age abandonment of upland areas in Britain. Anthropocene 13, 69–79 (2016).
Finkelstein, I. & Piasetzky, E. Radiocarbon dating the Iron Age in the Levant: a Bayesian model for six ceramic phases and six transitions. Antiquity 84, 374–385 (2010).
Lienkaemper, J. J. & Ramsey, C. B. OxCal: versatile tool for developing paleoearthquake chronologies—a primer. Seismol. Res. Lett. 80, 431–434 (2009).
Kohn, M. J. You are what you eat. Science 283, 335–336 (1999).
Szpak, P. Complexities of nitrogen isotope biogeochemistry in plant–soil systems: implications for the study of ancient agricultural and animal management practices. Front. Plant Sci. 5, 288 (2014).
Nadelhoffer, K. et al. 15N natural abundances and N use by tundra plants. Oecologia 107, 386–394 (1996).
Michener, R. & Lajtha, K. Stable Isotopes in Ecology and Environmental Science (Blackwell, Oxford, 2007).
Kuitems, M. et al. Carbon and nitrogen stable isotopes of well-preserved, middle Pleistocene bone collagenfrom Schöningen (Germany) and their palaeoecological implications. J. Hum. Evol. 89, 105–113 (2015).
Brotherton, P. et al. Neolithic mitochondrial haplogroup H genomes and the genetic origins of Europeans. Nat. Commun. 4, 1764 (2013).
Meyer, M. & Kircher, M. Illumina sequencing library preparation for highly multiplexed target capture and sequencing. Cold Spring Harb. Protoc. 5, pdb.prot5448 (2010).
Mitchell, K. J. et al. Ancient mitochondrial DNA reveals convergent evolution of giant short-faced bears (Tremarctinae) in North and South America. Biol. Lett. 12, 20160062 (2016).
Drummond, A. J. & Rambaut, A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol. Biol. 7, 214 (2007).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Rohling, E. J. et al. New constraints on the timing of sea level fluctuations during early to middle MarineIsotope Stage 3. Paleoceanography 23, PA3219 (2008).
Ehlers, J. & Gibbard, P. L. Quaternary Glaciations: Extent and Chronology Parts 1-3 (Developments inQuaternary Science 2, Elsevier, Amsterdam, 2004).
Andersen, K. K. et al. The Greenland ice core chronology 2005, 15–42 ka. Part 1: constructing the time scale. Quat. Sci. Rev. 25, 3246–3257 (2006).
Acknowledgements
We thank P.-O. Antoine for discussion, J. Saarinen for estimating the body mass of Elasmotherium, the Museum of the IPAE UB RAS and L. Petrov for providing bone samples for analysis, P. Campos for help with the stable isotope data, S. Brace for initial work on ancient DNA, the team of the ORAU for AMS dating and J. Hagstrum for an early stimulus to the study. Funding was provided by the Australian Research Council and Natural Environment Research Council, UK (grant number NE/G005982/1). Funding for part of the research was provided by the European Research Council under the European Union’s Seventh Framework Programme (FP7/2007-2013)—ERC grant 324139 ‘PalaeoChron’ award to T.H. This study was partly supported by the programme of the UB RAS (project number 18-4-4-3).
Author information
Authors and Affiliations
Contributions
P.K., T.v.K., A.J.S., A.M.L. and A.C. conceived the project. P.K., A.T. and E.P. provided samples and contextual information. Ancient DNA work and phylogenetic analyses were performed by K.J.M. and coordinated by A.C. Radiocarbon data were obtained and analysed by T.H., T.D. and D.C. at the ORAU, and J.v.d.P. at the CIO, while C.T. and T.H. undertook age modelling. Stable isotope analysis was performed and interpreted by M.K., while C.T. and A.J.S. provided context on climate and extinctions, respectively. All authors contributed to interpretation of the results and writing of the manuscript, which was coordinated by A.M.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Specimen provenances; detailed methods for DNA, stable isotope and radiocarbon dating analysis; Supplementary Figures 1,2 and Supplementary Tables 1–9; OxCal code for PHASE modelling; Supplementary References
Supplementary Table 1
Specimen numbers, localities, and summary of radiocarbon results. Raw radiocarbon dates are quoted ± 1-sigma; calibrated dates as 2-sigma range rounded to the nearest 10
Rights and permissions
About this article
Cite this article
Kosintsev, P., Mitchell, K.J., Devièse, T. et al. Evolution and extinction of the giant rhinoceros Elasmotherium sibiricum sheds light on late Quaternary megafaunal extinctions. Nat Ecol Evol 3, 31–38 (2019). https://doi.org/10.1038/s41559-018-0722-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0722-0
This article is cited by
-
Worldwide Late Pleistocene and Early Holocene population declines in extant megafauna are associated with Homo sapiens expansion rather than climate change
Nature Communications (2023)
-
Mammal extinction facilitated biome shift and human population change during the last glacial termination in East-Central Europe
Scientific Reports (2022)
-
Radiocarbon dating
Nature Reviews Methods Primers (2021)
-
Functional traits of the world’s late Quaternary large-bodied avian and mammalian herbivores
Scientific Data (2021)
-
The origin of Rhinocerotoidea and phylogeny of Ceratomorpha (Mammalia, Perissodactyla)
Communications Biology (2020)