Abstract
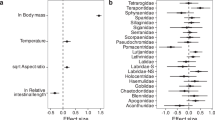
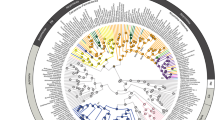
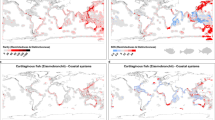
Marine reptiles flourished in the Mesozoic oceans, filling ecological roles today dominated by crocodylians, large fish, sharks and cetaceans. Many groups of these reptiles coexisted for over 50 million years (Myr), through major environmental changes. However, little is known about how the structure of their ecosystems or their ecologies changed over millions of years. We use the most common marine reptile fossils—teeth—to establish a quantitative system that assigns species to dietary guilds and then track the evolution of these guilds over the roughly 18-million-year history of a single seaway, the Jurassic Sub-Boreal Seaway of the United Kingdom. Groups did not significantly overlap in guild space, indicating that dietary niche partitioning enabled many species to live together. Although a highly diverse fauna was present throughout the history of the seaway, fish and squid eaters with piercing teeth declined over time while hard-object and large-prey specialists diversified, in concert with rising sea levels. High niche partitioning and spatial variation in dietary ecology related to sea depth also characterize modern marine tetrapod faunas, indicating a conserved ecological structure of the world’s oceans that has persisted for over 150 Myr.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pyenson, N. D., Kelley, N. P. & Parham, J. F. Marine tetrapod macroevolution: physical and biological drivers on 250 Ma of invasions and evolution in ocean ecosystems. Palaeogeogr. Palaeoclimatol. Palaeoecol. 400, 1–8 (2014).
Kelley, N. P. & Pyenson, N. D. Evolutionary innovation and ecology in marine tetrapods from the Triassic to the Anthropocene. Science 348, aaa3716 (2015).
Benson, R. B. J., Butler, R. J., Lindgren, J. & Smith, A. S. Mesozoic marine tetrapod diversity: mass extinctions and temporal heterogeneity in geological megabiases affecting vertebrates. Proc. R. Soc. B 277, 829–834 (2010).
Ciampaglio, C. N., Wray, G. A. & Corliss, B. H. A toothy tale of evolution: convergence in tooth morphology among marine Mesozoic–Cenozoic sharks, reptiles, and mammals. Sediment. Rec. 3, 4–8 (2005).
Andrews, C. W. A Descriptive Catalogue of the Marine Reptiles of the Oxford Clay: Part I (British Museum (Natural History), London, 1909).
Andrews, C. W. A Descriptive Catalogue of the Marine Reptiles of the Oxford Clay: Part II (British Museum (Natural History), London, 1913).
Massare, J. A. Tooth morphology and prey preference of Mesozoic marine reptiles. J. Vertebr. Paleontol. 7, 121–137 (1987).
Massare, J. A. Swimming capabilities of Mesozoic marine reptiles: implications for method of predation. Paleobiology 14, 187–205 (1988).
Buchy, M.-C. Morphologie dentaire et régime alimentaire des reptiles marins du Mésozoïque: revue critique et réévaluation. Oryctos 9, 49–82 (2010).
Chiarenza, A. A. et al. The youngest record of metriorhynchid crocodylomorphs, with implications for the extinction of Thalattosuchia. Cretaceous Res. 56, 608–616 (2015).
Stubbs, T. L. & Benton, M, J. Ecomorphological diversifications of Mesozoic marine reptiles: the roles of ecological opportunity and extinction. Paleobiology 42, 547–573 (2016).
Fischer, V., Bardet, N., Benson, R. J. B., Arkhangelsky, M. S. & Friedman, M. Extinction of fish-shaped marine reptiles associated with reduced evolutionary rates and global environmental volatility. Nat. Commun. 7, 10825 (2016).
Kelley, N. P. & Motani, R. Trophic convergence drives morphological convergence in marine tetrapods. Biol. Lett. 11, 20140709 (2015).
Young, M. T. et al. The cranial osteology and feeding ecology of the metriorhynchid crocodylomorph genera Dakosaurus and Plesiosuchus from the Late Jurassic of Europe. PLoS ONE 7, e44985 (2012).
Young, M. T., Brusatte, S. L., Andrade, M. B., Beatty, L. & Desojo, J. B. Tooth-on-tooth interlocking occlusion suggests macrophagy in the Mesozoic marine crocodylomorph Dakosaurus. Anat. Rec. 295, 1147–1158 (2012).
Benson, R. B. J. & Druckenmiller, P. S. Faunal turnover of marine tetrapods during the Jurassic–Cretaceous transition. Biol. Rev. 89, 1–23 (2014).
Young, M. T. Filling the ‘Corallian Gap’: re-description of a metriorhynchid crocodylomorph from the Oxfordian (Late Jurassic) of Headington, England. Hist. Biol. 26, 80–90 (2014).
Fischer, V. et al. Peculiar macrophagous adaptations in a new Cretaceous pliosaurid. R. Soc. Open Sci. 2, 150552 (2015).
Fischer, V. et al. Plasticity and convergence in the evolution of short-necked plesiosaurs. Curr. Biol. 27, 1667–1676 (2017).
Cox, B. M. in British Upper Jurassic Stratigraphy (eds Wright, J. K. & Cox, B. M.) Ch. 1, 3–10 (Geological Conservation Review Series 21, Joint Nature Conservation Committee, Peterborough, 2001).
Cope, J. C. in The Geology of England and Wales 2nd edn (eds Brenchley, P. J. & Rawson, P. F.) 325–364 (Geological Society, London, 2006).
Foffa, D., Young, M. T. & Brusatte, S. L. Filling the Corallian gap: new information on Late Jurassic marine reptile faunas from England. Acta Palaeontol. Pol. 63, 287–313 (2018).
Cecca, F., Garin, B. M., Marchand, D., Lathuiliere, B. & Bartolini, A. Paleoclimatic control of biogeographic and sedimentary events in Tethyan and peri-Tethyan areas during the Oxfordian (Late Jurassic). Palaeogeogr. Palaeoclimatol. Palaeoecol. 222, 10–32 (2005).
Armstrong, H. A. et al. Hadley circulation and precipitation changes controlling black shale deposition in the Late Jurassic Boreal Seaway. Paleoceanography 31, 1041–1053 (2016).
Cox, B. M., Hudson, J. D. & Martill, D. M. Lithostratigraphic nomenclature of the Oxford Clay (Jurassic). Proc. Geol. Assoc. 103, 343–345 (1992).
Mettam, C., Johnson, A. L. A., Nunn, E. V. & Schӧne, B. R. Stable isotope (δ18O and δ13C) sclerochronology of Callovian (Middle Jurassic) bivalves (Gryphaea (Bilobissa) dilobotes) and belemnites (Cylindroteuthis puzosiana) from the Peterborough Member of the Oxford Clay Formation (Cambridgeshire, England): evidence of palaeoclimate, water depth and belemnite behaviour. Palaeogeogr. Palaeoclimatol. Palaeoecol. 399, 187–201 (2014).
Dromart, G. et al. Perturbation of the carbon cycle at the Middle–Late Jurassic transition: geological and geochemical evidence. Am. J. Sci. 303, 667–707 (2003).
Dromart, G. et al. Ice age at the Middle–Late Jurassic transition? Earth Planet. Sci. Lett. 213, 205–220 (2003).
Gallois, R. W. The Kimmeridge Clay: the most intensively studied formation in Britain. J. Open Univ. Geol. Soc. 25, 33–38 (2004).
Haq, B. U., Hardenbol, J. & Vail, P. R. in Sea Level Changes—An Integrated Approach (eds Wilgus, C. K. et al.) 71–108 (Special Publication 42, SEPM, Tulsa, 1988).
Pauly, D., Trites, A. W., Capuli, E. & Christensen, V. Diet composition and trophic levels of marine mammals. ICES J. Mar. Sci. 55, 467–481 (1998).
Esteban, R. et al. Identifying key habitat and seasonal patterns of a critically endangered population of killer whales. J. Mar. Biol. Assoc. UK 94, 1317–1325 (2013).
Forney, K. A. Environmental models of cetacean abundance: reducing uncertainty in population trends. Conserv. Biol. 14, 1271–1286 (2000).
Yen, P. P. W., Sydeman, W. J. & Hyrenbach, K. D. Marine birds and cetacean associations with bathymetric habitats and shallow-water topographies: implications for trophic transfer and conservation. J. Mar. Syst. 50, 79–99 (2004).
Balance, L. T., Pitman, R. L. & Fiedler, P. C. Oceanographic influences on seabirds and cetaceans in the eastern tropical Pacific: a review. Prog. Oceanogr. 69, 360–390 (2006).
MacLoed, C. D., Weit, C. R., Pierpoint, C. & Harland, E. J. The habitat preferences of marine mammals west of Scotland (UK). J. Mar. Biol. Assoc. UK 87, 157–164 (2007).
Spitz, J. et al. Prey preferences among the community of deep-diving odontocetes from the Bay of Biscay, Northeast Atlantic. Deep Sea Res. Pt I 58, 273–282 (2011).
Weir, C. R., MacLeod, C. D. & Pierce, G. J. Habitat preferences and evidence for niche partitioning amongst cetaceans in the waters between Gabon and Angola, eastern tropical Atlantic. J. Mar. Biol. Assoc. UK 92, 1735–1749 (2012).
Roberts, J. J. et al. Habitat-based cetacean density models for the U.S. Atlantic and Gulf of Mexico. Sci. Rep. 6, 22615 (2016).
Fossete, S. et al. Resource partitioning facilitates coexistence in sympatric cetaceans in the California Current. Ecol. Evol. 7, 9085–9097 (2017).
Larson, D. W., Brown, C. M. & Evans, D. C. Dental disparity and ecological stability in bird-like dinosaurs prior to the end-Cretaceous mass extinction. Curr. Biol. 26, 1325–1333 (2016).
Smith, J. B., Vann, D. R. & Dodson, P. Dental morphology and variation in theropod dinosaurs: implications for the taxonomic identification of isolated teeth. Anat. Rec. Pt A 285, 699–736 (2005).
Noè, L. F. A Taxonomic and Functional Study of the Callovian (Middle Jurassic) Pliosauroidea (Reptilia, Sauropterygia). PhD thesis, Univ. Derby (2001).
Sassoon, J., Foffa, D. & Marek, R. Dental ontogeny and replacement in Pliosauridae. R. Soc. Open Sci. 2, 150384 (2015).
Foffa, D., Young, M. T., Brusatte, S. L., Graham, M. R. & Steel, L. A new metriorhynchid crocodylomorph from the Oxford Clay Formation (Middle Jurassic) of England, with implications for the origin and diversification of Geosaurini. J. System. Palaeontol. 16, 1123–1143 (2018).
Young, M. T., Hastings, A. K., Allain, R. & Smith, T. J. Revision of the enigmatic crocodyliform Elosuchus felixi de Lapparent de Broin, 2002 from the Lower–Upper Cretaceous boundary of Niger: potential evidence for an early origin of the clade Dyrosauridae. Zool. J. Linn. Soc. 179, 377–403 (2017).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Anderson, P. S. L., Friedman, M., Brazeau, M. D. & Rayfield, E. J. Initial radiation of jaws demonstrated stability despite faunal and environmental change. Nature 476, 206–209 (2011).
Gower, J. C. A general coefficient of similarity and some of its properties. Biometrics 27, 857–871 (1971).
Hammer, Ø, Harper, D. A. T. & Ryan, P. D. PAST: paleontological statistics software package for education and data analysis. Palaeontol. Electron 4, 1–9 (2001).
Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 26, 32–46 (2001).
R Development Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2012).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B. 57, 289–300 (1995).
Wills, M. A., Briggs, D. E. G. & Fortey, R. A. Disparity as an evolutionary index: a comparison of Cambrian and recent arthropods. Paleobiology 20, 93–130 (1994).
Guillerme, T. dispRity: a modular R package for measuring disparity. Methods Ecol. Evol. 9, 1755–1763 (2018).
Brusatte, L. S., Lloyd, G. T., Wang, S. C. & Norell, M. A. Gradual assembly of avian body plan culminated in rapid rates of evolution across the dinosaur–bird transition. Curr. Biol. 20, 2386–2392 (2014).
Foote, M. Contributions of individual taxa to overall morphological disparity. Paleobiology 19, 403–419 (1993).
Navarro, N. MDA: a MATLAB-based program for morphospace-disparity analysis. Comput. Geosci. 29, 655–664 (2003).
Acknowledgements
We thank D. Hutchinson and I. Gladstone (BRSMG), M. Riley (CAMSM), P. Tomlinson (DORCM), N. Clark (GLAHM), S. Etches (MJML), L. Steel (NHMUK), E. Howlett and H. Ketchum (OUMNH), and E. Jarvis, S. King and S. Ogilvy (YORYM) for access and guidance during D.F.’s visits to the museum collections. D.F.’s museum visits were funded by the Small Grant Scheme ‘2015 Wood Award’ (PASW201402), Systematics Research Fund and Richard Owen Research Fund by the Palaeontographical Society. M.T.Y. and S.L.B. are supported by a Leverhulme Trust Research Project grant (RPG-2017-167), and S.L.B. is supported by a Marie Curie Career Integration Grant (630652). We thank P. dePolo for comments on the manuscript, and M. Puttick and T. Guillerme for discussion and technical support.
Author information
Authors and Affiliations
Contributions
D.F. led the project, conceived the study and wrote the initial draft manuscript. M.T.Y. and S.L.B. helped develop the project, edited drafts and provided guidance on the statistical analyses. S.L.B. wrote the final manuscript, which was revised by all authors. D.F. designed and performed the analyses with technical support from T.L.S. and K.G.D.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary text, tables, figures and references
Supplementary Datasets 1-4
Specimen-character matrix; non-parametric MANOVA tests results; results of LDA group classifications; sensitivity analyses of guild groupings
Rights and permissions
About this article
Cite this article
Foffa, D., Young, M.T., Stubbs, T.L. et al. The long-term ecology and evolution of marine reptiles in a Jurassic seaway. Nat Ecol Evol 2, 1548–1555 (2018). https://doi.org/10.1038/s41559-018-0656-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0656-6
This article is cited by
-
Predatory synapsid ecomorphology signals growing dynamism of late Palaeozoic terrestrial ecosystems
Communications Biology (2024)
-
The rise of macropredatory pliosaurids near the Early-Middle Jurassic transition
Scientific Reports (2023)
-
Pterosaurs ate soft-bodied cephalopods (Coleoidea)
Scientific Reports (2020)
-
The macroevolutionary landscape of short-necked plesiosaurians
Scientific Reports (2020)
-
Early high rates and disparity in the evolution of ichthyosaurs
Communications Biology (2020)