Abstract
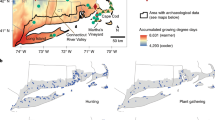
European colonization of South America instigated a continental-scale depopulation of its indigenous peoples. The impact of depopulation on the tropical forests of South America varied across the continent. Furthermore, the role that indigenous peoples played in transforming the biodiverse tropical forests of the Andean–Amazonian corridor before ad 1492 remains unknown. Here, we reconstruct the past 1,000 years of changing human impact on the cloud forest of Ecuador at a key trade route, which connected the Inkan Empire to the peoples of Amazonia. We compare this historical landscape with the pre-human arrival (around 44,000–42,000 years ago) and modern environments. We demonstrate that intensive land-use within the cloud forest before European arrival deforested the landscape to a greater extent than modern (post-ad 1950) cattle farming. Intensive indigenous land-use ended abruptly around ad 1588 following a catastrophic population decline. Forest succession then took around 130 years to establish a structurally intact forest—one comparable to that which occurred before the arrival of the first humans to the continent. We show that nineteenth-century descriptions of the Andean–Amazonian corridor as a pristine wilderness record a shifted ecological baseline—one that less than 250 years earlier had consisted of a heavily managed and cultivated landscape.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dobyns, H. F. An appraisal of techniques with a new hemispheric estimate. Curr. Anthropol. 7, 395–416 (1966).
Mann, C. C. 1493: How the Ecological Collision of Europe and the Americas Gave Rise to the Modern World (Granta, London, 2011).
Crosby, A. W. Ecological Imperialism: The Biological Expansion of Europe, 900–1900 (Cambridge Univ. Press, Cambridge, 2004).
Levis, C. et al. Persistent effects of pre-Columbian plant domestication on Amazonian forest composition. Science 355, 925–931 (2017).
McMichael, C. H., Feeley, K. J., Dick, C. W., Piperno, D. R. & Bush, M. B. Comment on ‘Persistent effects of pre-Columbian plant domestication on Amazonian forest composition’. Science 358, eaan8347 (2017).
Watling, J. et al. Impact of pre-Columbian ‘geoglyph’ builders on Amazonian forests. Proc. Natl Acad. Sci. USA 114, 1868–1873 (2017).
Piperno, D. R., McMichael, C. & Bush, M. B. Further evidence for localized, short-term anthropogenic forest alterations across pre-Columbian Amazonia. Proc. Natl Acad. Sci. USA 114, E4118–E4119 (2017).
McMichael, C. H. et al. Sparse pre-Columbian human habitation in western Amazonia. Science 336, 1429–1431 (2012).
Turner, B. L. & Butzer, K. W. The Columbian encounter and land-use change. Environ. Sci. Policy Sustain. Dev. 34, 16–44 (1992).
Clement, C. R. et al. The domestication of Amazonia before European conquest. Proc. R. Soc. B 282, 1–9 (2015).
Heckenberger, M. J. et al. Amazonia 1492: pristine forest or cultural parkland? Science 301, 1710–1714 (2003).
Myers, N., Mittermeier, R. A., Mittermeier, C. G., da Fonseca, G. A. B. & Kent, J. Biodiversity hotspots for conservation priorities. Nature 403, 853–858 (2000).
Uzendoski, M. The horizontal archipelago: the Quijos/Upper Napo regional system. Ethnohistory 51, 317–357 (2004).
Newson, L. A. Life and Death in Early Colonial Ecuador (Univ. Oklahoma Press, Norman, 1995).
Sarmiento, F. O. The Quijos River Valley: a protected landscape as best management practice for conservation and development in tropandean Ecuador. Georg. Wright Forum 14, 59–66 (1997).
Cuéllar, A. M. The Quijos Chiefdoms: Social Change and Agriculture in the Eastern Andes of Ecuador (Univ. Pittsburgh, Pittsburgh, 2009).
Porras, P. I. Fase Cosanga (Centro de Publicaciones de la Pontificia Universidad Católica del Ecuador, Quito, 1975).
Medina, J. T. The Discovery of the Amazon: According to the Account of Friar Gaspar de Carvajal and Other Documents (The American Geographical Society, New York, 1934).
Convey, R. A. in Handbook of South American Archaeology (eds Silverman, H. & Isbell, W.) 809–830 (Springer, New York, 2008).
de Las Casas, B. A Short Account of the Destruction of the Indies 1552 (Penguin, London, 2004).
Jameson, W. Excursion made from Quito to the River Napo, January to May, 1857. J. R. Geogr. Soc. Lond. 28, 337–349 (1858).
Orton, J. The Andes and the Amazon; or, Across the Continent of South America (Harper & Brothers, New York, 1875).
Hall, M. L. et al. Antisana volcano: a representative andesitic volcano of the eastern cordillera of Ecuador: petrography, chemistry, tephra and glacial stratigraphy. J. South Am. Earth Sci. 73, 50–64 (2017).
Colinvaux, P. A., de Oliveira, P. E. & Patiño, J. E. M. Amazon Pollen Manual and Atlas (Harwood Academic, Amsterdam, 1999).
Faegri, K. & Iversen, J. Textbook of Pollen Analysis (John Wiley & Sons, Chichester, 1989).
Whitlock, C. & Larsen, C. in Tracking Environmental Change Using Lake Sediments Vol. 3 (eds Smol, J. P., Birks, H. J. B. & Last, W. M.) 75–97 (Kluwer Academic, Dordrecht, 2001).
van Geel, B. in Tracking Environmental Change Using Lake Sediments Vol. 3 (eds Smol, J. P., Birks, H. J. B. & Last, W. M.) 99–120 (Kluwer Academic, Dordrecht, 2001).
Loughlin, N. J. D. et al. Landscape-scale drivers of glacial ecosystem change in the montane forests of the eastern Andean flank, Ecuador. Palaeogeogr. Palaeoclimatol. Palaeoecol. 489, 198–208 (2018).
Jørgensen, P. M. & León-Yánez, S. Catalogue of the Vascular Plants of Ecuador (Missouri Botanical Garden Press, Missouri, 1999).
Bush, M. B., Piperno, R. D. & Colinvaux, P. A. A 6,000 year history of Amazonian maize cultivation. Nature 340, 303–305 (1989).
Bush, M. B., Silman, M. R., McMichael, C. & Saatchi, S. Fire, climate change and biodiversity in Amazonia: a late-Holocene perspective. Phil. Trans. R. Soc. B 363, 1795–1802 (2008).
Norden, N., Chazdon, R. L., Chao, A., Jiang, Y.-H. & Vílchez-Alvarado, B. Resilience of tropical rain forests: tree community reassembly in secondary forests. Ecol. Lett. 12, 385–394 (2009).
Pauly, D. Anecdotes and the shifting baseline syndrome of fisheries. Trends Ecol. Evol. 10, 430 (1995).
Bush, M. B. et al. Holocene fire and occupation in Amazonia: records from two lake districts. Phil. Trans. R. Soc. B 362, 209–218 (2007).
Blaauw, M. & Christen, A. J. Flexible paleoclimate age–depth models using autoregressive gamma process. Bayesian Anal. 6, 457–474 (2011).
R Development Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, 2015).
Bennett, K. psimpoll and pscomb Programs for Plotting and Analysis (2008); www.chrono.qub.ac.uk/psimpoll/psimpoll.html
Oksanen, J. et al. Vegan: Community Ecology Package Version 2.3–5 (2016); https://cran.r-project.org/package=vegan
Acknowledgements
This work was supported by the NERC and The Open University through a scholarship to N.J.D.L. (NE/L501888/1), and a NERC fellowship to E.M. (NE/J018562/1). Funding for preliminary radiocarbon dating was awarded by NERC (1881.0415). Permits for fieldwork in Ecuador were provided by the Ministry of Environment, Ecuador (14-2012-IC-FLO-DPAP-MA). X-ray fluorescence analysis was undertaken by the late J. Watson (The Open University). This work was improved by the comments of A. Coe (The Open University), C. McMichael (University of Amsterdam), J. Symonds (University of Amsterdam) and M. Bush (Florida Institute of Technology).
Author information
Authors and Affiliations
Contributions
N.J.D.L., E.M. and W.D.G. determined the research objectives. P.M. located the sample site and secured permissions. Lake sampling was undertaken by N.J.D.L., E.M. and W.D.G. Sediment processing, data collection and analysis was performed by N.J.D.L. The manuscript was written by N.J.D.L., E.M. and W.D.G. with input from P.M. All authors read and approved the manuscript for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures, tables, notes and references
Rights and permissions
About this article
Cite this article
Loughlin, N.J.D., Gosling, W.D., Mothes, P. et al. Ecological consequences of post-Columbian indigenous depopulation in the Andean–Amazonian corridor. Nat Ecol Evol 2, 1233–1236 (2018). https://doi.org/10.1038/s41559-018-0602-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0602-7
This article is cited by
-
Phytolith assemblages reflect variability in human land use and the modern environment
Vegetation History and Archaeobotany (2024)
-
The loss of an indigenous constructed landscape following British invasion of Australia: An insight into the deep human imprint on the Australian landscape
Ambio (2021)
-
Forests protect aquatic communities from detrimental impact by volcanic deposits in the tropical Andes (Ecuador)
Regional Environmental Change (2021)
-
How Joannites’ economy eradicated primeval forest and created anthroecosystems in medieval Central Europe
Scientific Reports (2020)
-
2,100 years of human adaptation to climate change in the High Andes
Nature Ecology & Evolution (2019)