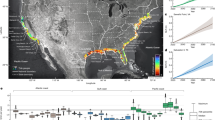
Abstract
The distribution of living organisms on Earth is spatially structured. Early biogeographers identified the existence of multiple zoogeographical regions, characterized by faunas with homogeneous composition that are separated by biogeographical boundaries. Yet, no study has deciphered the factors shaping the distributions of terrestrial biogeographical boundaries at the global scale. Here, using spatial regression analyses, we show that tectonic movements, sharp changes in climatic conditions and orographic barriers determine extant biogeographical boundaries. These factors lead to abrupt zoogeographical transitions when they act in concert, but their prominence varies across the globe. Clear differences exist among boundaries representing profound or shallow dissimilarities between faunas. Boundaries separating zoogeographical regions with limited divergence occur in areas with abrupt climatic transitions. In contrast, plate tectonics determine the separation between deeply divergent biogeographical realms, particularly in the Old World. Our study reveals the multiple drivers that have shaped the biogeographical regions of the world.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fabricius, J. C. Philosophia Entomologica (Impensis Carol. Ernest. Bohnii, 1778).
De Candolle, A. P. Essai Élémentaire de Géographie Botanique (F. Levrault, 1820).
Swainson, W. A Treatise on the Geography and Classification of Animals (Longman, Rees, Brown, Green, & Longman, 1835).
Wallace, A. R. The Geographical Distribution of Animals (Harper, 1876).
Sclater, P. L. On the general geographical distribution of the members of the class Aves. J. Proc. Linn. Soc. Lond. Zool. 2, 130–145 (1858).
Cox, B. The biogeographic regions reconsidered. J. Biogeogr. 28, 511–523 (2001).
Morrone, J. J. Biogeographical regionalisation of the world: a reappraisal. Aust. Syst. Bot. 28, 81–90 (2015).
Crisci, J. V., Cigliano, M. M., Morrone, J. J. & Roig-Junent, S. Historical biogeography of southern South America. Syst. Biol. 40, 152–171 (1991).
Rueda, M., Rodriguez, M. A. & Hawkins, B. A. Towards a biogeographic regionalization of the European biota. J. Biogeogr. 37, 2067–2076 (2010).
Proches, S. & Ramdhani, S. The world’s zoogeographical regions confirmed by cross-taxon analyses. Bioscience 62, 260–270 (2012).
Holt, B. et al. An update of Wallace’s zoogeographic regions of the world. Science 339, 74–78 (2013).
Rueda, M., Rodriguez, M. A. & Hawkins, B. A. Identifying global zoogeographical regions: lessons from Wallace. J. Biogeogr. 40, 2215–2225 (2013).
Vilhena, D. A. & Antonelli, A. A network approach for identifying and delimiting biogeographical regions. Nat. Commun. 6, 6848 (2015).
Kreft, H. & Jetz, W. A framework for delineating biogeographical regions based on species distributions. J. Biogeogr. 37, 2029–2053 (2010).
Edler, D., Guedes, T., Zizka, A., Rosvall, M. & Antonelli, A. Infomap bioregions: interactive mapping of biogeographical regions from species distributions. Syst. Biol. https://doi.org/10.1093/sysbio/syw087 (in the press).
Lomolino, M. V., Riddle, B. R., Whittaker, R. J. & Brown, J. H. Biogeography 4th edn (Sinauer Associates, 2010).
Riddle, B. R. & Hafner, D. J. Integrating pattern with process at biogeographic boundaries: the legacy of Wallace. Ecography 33, 321–325 (2010).
Glor, R. E. & Warren, D. Testing ecological explanations for biogeographic boundaries. Evolution 65, 673–683 (2011).
Kreft, H. & Jetz, W. Comment on ‘An update of Wallace’s zoogeographic regions of the world’. Science 341, 343 (2013).
Sexton, J. P., McIntyre, P. J., Angert, A. L. & Rice, K. J. Evolution and ecology of species range limits. Annu. Rev. Ecol. Evol. Syst. 40, 415–436 (2009).
Buckley, L. B. & Jetz, W. Linking global turnover of species and environments. Proc. Natl Acad. Sci. USA 105, 17836–17841 (2008).
Melo, A. S., Rangel, T. F. L. V. B. & Diniz-Filho, J. A. F. Environmental drivers of beta-diversity patterns in New-World birds and mammals. Ecography 32, 226–236 (2009).
Graham, R. W. et al. Spatial response of mammals to Late Quaternary environmental fluctuations. Science 272, 1601–1606 (1996).
Davis, M. B. & Shaw, R. G. Range shifts and adaptive responses to Quaternary climate change. Science 292, 673–679 (2001).
Taberlet, P., Fumagalli, L., Wust-Saucy, A. G. & Cosson, J. F. Comparative phylogeography and postglacial colonization routes in Europe. Mol. Ecol. 7, 453–464 (1998).
Sandel, B. et al. The influence of Late Quaternary climate-change velocity on species endemism. Science 334, 660–664 (2011).
Mao, K. S. et al. Distribution of living Cupressaceae reflects the breakup of Pangea. Proc. Natl Acad. Sci. USA 109, 7793–7798 (2012).
Chan, W.-P. et al. Seasonal and daily climate variation have opposite effects on species elevational range size. Science 351, 1437–1439 (2016).
Bivand, R., Pebesma, E. J. & Gómez-Rubio, V. Applied Spatial Data Analysis with R (Springer, 2008).
Vermeij, G. J. When biotas meet: understanding biotic interchange. Science 253, 1099–1104 (1991).
Simpson, G. G. S. Splendid Isolation: The Curious History of South American Mammals (Yale Univ. Press, 1980).
Bacon, C. D. et al. Biological evidence supports an early and complex emergence of the Isthmus of Panama. Proc. Natl Acad. Sci. USA 112, 6110–6115 (2015).
Daza, J. M., Castoe, T. A. & Parkinson, C. L. Using regional comparative phylogeographic data from snake lineages to infer historical processes in middle America. Ecography 33, 343–354 (2010).
Montes, C. et al. Middle Miocene closure of the Central American Seaway. Science 348, 226 (2015).
Müller, P. Aspects of Biogeography (Springer, 1974).
Holt, B. G. et al. Response to comment on ‘An update of Wallace’s zoogeographic regions of the world’. Science 341, 343 (2013).
Seton, M. et al. Global continental and ocean basin reconstructions since 200 Ma. Earth-Sci. Rev. 113, 212–270 (2012).
Briggs, J. C. Biogeography and Plate Tectonics (Elsevier, 1987).
He, J., Kreft, H., Gao, E., Wang, Z. & Jiang, H. Patterns and drivers of zoogeographical regions of terrestrial vertebrates in China. J. Biogeogr. https://doi.org/10.1111/jbi.12892 (2016).
Smith, B. T. & Klicka, J. The profound influence of the Late Pliocene Panamanian uplift on the exchange, diversification, and distribution of New World birds. Ecography 33, 333–342 (2010).
Pomara, L. Y., Ruokolainen, K. & Young, K. R. Avian species composition across the Amazon River: the roles of dispersal limitation and environmental heterogeneity. J. Biogeogr. 41, 784–796 (2014).
Hurlbert, A. H. & Jetz, W. Species richness, hotspots, and the scale dependence of range maps in ecology and conservation. Proc. Natl Acad. Sci. USA 104, 13384–13389 (2007).
Ficetola, G. F. et al. An evaluation of the robustness of global amphibian range maps. J. Biogeogr. 41, 211–221 (2014).
Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. High resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005).
Boucher-Lalonde, V., Morin, A. & Currie, D. J. A consistent occupancy–climate relationship across birds and mammals of the Americas. Oikos 123, 1029–1036 (2014).
Harrison, S. P., Bartlein, P. J. & Prentice, I. C. What have we learnt from palaeoclimate simulations? J. Quaternary Sci. 31, 363–385 (2016).
Harrison, S. P. et al. Evaluation of CMIP5 palaeo-simulations to improve climate projections. Nat. Clim. Change 5, 735–743 (2015).
Mauri, A., Davis, B. A. S., Collins, P. M. & Kaplan, J. O. The influence of atmospheric circulation on the mid-Holocene climate of Europe: a data-model comparison. Clim. Past 10, 1925–1938 (2014).
Williams, S., Müller, R., Landgrebe, T. & Whittaker, J. An open-source software environment for visualizing and refining plate tectonic reconstructions using high-resolution geological and geophysical data sets. GSA Today 22, 4–9 (2012).
Boyden, J. A. et al. in Geoinformatics: Cyberinfrastructure for the Solid Earth Sciences (eds Keller G. R. & Baru C. ) 99–113 (Cambridge Univ. Press, 2011).
Beale, C. M., Lennon, J. J., Yearsley, J. M., Brewer, M. J. & Elston, D. A. Regression analysis of spatial data. Ecol. Lett. 13, 246–264 (2010).
Dormann, C. F. et al. Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography 30, 609–628 (2007).
Kissling, W. D. & Carl, G. Spatial autocorrelation and the selection of simultaneous autoregressive models. Global Ecol. Biogeogr. 17, 59–71 (2008).
Alam, M., Roennegard, L. & Shen, X. Fitting conditional and simultaneous autoregressive spatial models in hglm. R Journal 7, 5–18 (2015).
Rousset, F. & Ferdy, J.-B. Testing environmental and genetic effects in the presence of spatial autocorrelation. Ecography 37, 781–790 (2014).
Dormann, C. F. et al. Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36, 27–46 (2013).
Hijmans, R. J. raster: Geographic Data Analysis and Modeling. R package version 2.5-2 (2015); http://CRAN.R-project.org/package=raster
Ronnegard, L., Shen, X. & Alam, M. hglm: a package for fitting hierarchical generalized linear models. R Journal 2, 20–28 (2010).
Fox, J. & Weisberg, S. An R Companion to Applied Regression (Sage, 2011).
Bivand, R. & Lewin-Koh, N. maptools: Tools for Reading and Handling Spatial Objects (www.r-project.org, 2014).
Allouche, O., Tsoar, A. & Kadmon, R. Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J. Appl. Ecol. 43, 1223–1232 (2006).
Rosenthal, R. in The Handbook of Research Synthesis (eds Cooper, H. & Hedges, L. V. ) 231–244 (Russel Sage Foundation, 1994).
Mellin, C., Mengersen, K., Bradshaw, C. J. A. & Caley, M. J. Generalizing the use of geographical weights in biodiversity modelling. Global Ecol. Biogeogr. 23, 1314–1323 (2014).
Nakaya, T., Fotheringham, A. S., Brunsdon, C. & Charlton, M. Geographically weighted Poisson regression for disease association mapping. Stat. Med. 24, 2695–2717 (2005).
da Silva, A. R. & Fotheringham, A. S. The multiple testing issue in geographically weighted regression. Geogr. Anal. 48, 233–247 (2016).
Acknowledgements
We thank S. Ramdhani for providing high-resolution maps of bioregions. The research leading to these results has received funding from the European Research Council under the European Community’s Seven Framework Programme FP7/2007–2013 Grant Agreement no. 281422 (TEEMBIO). All authors belong to the Laboratoire d’Écologie Alpine, which is part of Labex OSUG@2020 (ANR10 LABX56).
Author information
Authors and Affiliations
Contributions
G.F.F., F.M. and W.T. conceived the study. G.F.F. performed all analyses with the help of F.M. G.F.F. wrote the first version of the manuscript, with contributions from F.M. and W.T.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–5, Supplementary Tables 1–5, Supplementary Discussion, Supplementary References. (PDF 1576 kb)
Rights and permissions
About this article
Cite this article
Ficetola, G., Mazel, F. & Thuiller, W. Global determinants of zoogeographical boundaries. Nat Ecol Evol 1, 0089 (2017). https://doi.org/10.1038/s41559-017-0089
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41559-017-0089
This article is cited by
-
Range-wide whole-genome resequencing of the brown bear reveals drivers of intraspecies divergence
Communications Biology (2023)
-
An updated floristic map of the world
Nature Communications (2023)
-
Alfred Russel Wallace’s legacy: an interdisciplinary conception of evolution in space and time
npj Biodiversity (2023)
-
Extinction of threatened vertebrates will lead to idiosyncratic changes in functional diversity across the world
Nature Communications (2021)
-
Influence of historical changes in tropical reef habitat on the diversification of coral reef fishes
Scientific Reports (2021)