Abstract
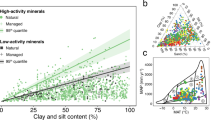
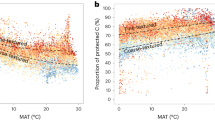
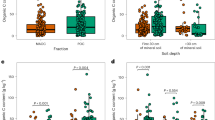
Soil organic matter can release carbon dioxide to the atmosphere as the climate warms. Organic matter sorbed to reactive (iron- and aluminium-bearing) soil minerals is an important mechanism for long-term carbon storage. However, the global distribution of mineral-stored carbon across climate zones and consequently its overall contribution to the global soil carbon pool is poorly known. We measured carbon held by reactive minerals across a broad range of climates. Carbon retained by reactive minerals was found to contribute between 3 and 72% of organic carbon found in mineral soil, depending on mean annual precipitation and potential evapotranspiration. Globally, we estimate ~600 Gt of soil carbon is retained by reactive minerals, with most occurring in wet forested biomes. For many biomes, the fraction of organic carbon retained by reactive minerals is responsive to slight shifts in effective moisture, suggesting high sensitivity to future changes in climate.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Open raster, vector and tabular data are posted on the Harvard Dataverse under a CC0 Public Domain Dedication licence that allows full and unrestricted global use of the data generated during this research while giving proper citation to the original author. These posted data allow for full replication, at the minimum mapping unit, of the results generated during this analysis. The data that support the findings of this study are available at https://doi.org/10.7910/DVN/NGFY6A36. Correspondence and requests for materials should be made to M.G.K.
References
Gu, B., Schmitt, J., Chen, Z., Liang, L. & McCarthy, J. F. Adsorption and desorption of natural organic matter on iron oide: mechanisms and models. Environ. Sci. Technol. 28, 38–46 (1994).
Kramer, M. G., Sanderman, J., Chadwick, O. A., Chorover, J. & Vitousek, P. M. Long-term carbon storage through retention of dissolved aromatic acids by reactive particles in soil. Glob. Change Biol. 18, 2594–2605 (2012).
Kaiser, K., Guggenberger, G. & Zech, W. Sorption of DOM and DOM fractions to forest soils. Geoderma 74, 281–303 (1996).
Kaiser, K. & Guggenberger, G. The role of DOM sorption to mineral surfaces in the preservation of organic matter in soils. Org. Geochem. 31, 711–725 (2000).
Guggenberger, G. & Kaiser, K. Dissolved organic matter in soil: challenging the paradigm of sorptive preservation. Geoderma 113, 293–310 (2003).
Jardine, P., McCarthy, J. & Weber, N. Mechanisms of dissolved organic carbon adsorption on soil. Soil Sci. Soc. Am. J. 53, 1378–1385 (1989).
Kalbitz, K. & Kaiser, K. Contribution of dissolved organic matter to carbon storage in forest mineral soils. J. Plant Nutr. Soil Sci. 171, 52–60 (2008).
Kalbitz, K., Schwesig, D., Rethemeyer, J. & Matzner, E. Stabilization of dissolved organic matter by sorption to the mineral soil. Soil Biol. Biochem. 37, 1319–1331 (2005).
Kalbitz, K., Solinger, S., Park, J.-H., Michalzik, B. & Matzner, E. Controls on the dynamics of dissolved organic matter in soils: a review. Soil Sci. 165, 277–304 (2000).
Jenny, H. Factors of Soil Formation: A System of Quantitative Pedology (Courier Corporation, New York, 1994).
Chadwick, O. A. & Chorover, J. The chemistry of pedogenic thresholds. Geoderma 100, 321–353 (2001).
Dixon, J. L., Chadwick, O. A. & Vitousek, P. M. Climate‐driven thresholds for chemical weathering in postglacial soils of New Zealand. J. Geophys. Res. Earth Surf. 121, 1619–1634 (2016).
Vitousek, P., Dixon, J. L. & Chadwick, O. A. Parent material and pedogenic thresholds: observations and a simple model. Biogeochemistry 130, 147–157 (2016).
Dahlgren, R., Boettinger, J., Huntington, G. & Amundson, R. Soil development along an elevational transect in the western Sierra Nevada, California. Geoderma 78, 207–236 (1997).
Peay, K. G. et al. Convergence and contrast in the community structure of Bacteria, Fungi and Archaea along a tropical elevation-climate gradient. FEMS Microbiol. Ecol. 93, 5 (2017).
Von Sperber, C., Stallforth, R., Du Preez, C. & Amelung, W. Changes in soil phosphorus pools during prolonged arable cropping in semiarid grasslands. Eur. J. Soil Sci. 68, 462–471 (2017).
Kramer, M. G. & Chadwick, O. A. Controls on carbon storage and weathering in volcanic soils across a high‐elevation climate gradient on Mauna Kea, Hawaii. Ecology 97, 2384–2395 (2016).
Slessarev, E. et al. Water balance creates a threshold in soil pH at the global scale. Nature 540, 567–569 (2016).
Rasmussen, C. et al. Beyond clay: towards an improved set of variables for predicting soil organic matter content. Biogeochemistry 137, 297–306 (2018).
Muhs, D. R. Intrinsic thresholds in soil systems. Phys. Geogr. 5, 99–110 (1984).
Sowers, T., Adhikari, D., Wang, J., Yang, Y. & Sparks, D. L. Spatial associations and chemical composition of organic carbon sequestered in Fe, Ca, and organic carbon ternary systems. Environ. Sci. Technol. 52, 6936–6944 (2018).
Sowers, T. D., Stuckey, J. W. & Sparks, D. L. The synergistic effect of calcium on organic carbon sequestration to ferrihydrite. Geochem. Trans. 19, 4 (2018).
Rowley, M. C., Grand, S. & Verrecchia, É. P. Calcium-mediated stabilisation of soil organic carbon. Biogeochemistry 137, 27–49 (2018).
Zomer, R. J. et al. Trees and Water: Smallholder Agroforestry on Irrigated Lands in Northern India Research Report No. 122 (IWMI, 2007).
Zomer, R. J., Trabucco, A., Bossio, D. A. & Verchot, L. V. Climate change mitigation: a spatial analysis of global land suitability for clean development mechanism afforestation and reforestation. Agric. Ecosyst. Environ. 126, 67–80 (2008).
Hengl, T. et al. SoilGrids250m: global gridded soil information based on machine learning. PLoS ONE 12, e0169748 (2017).
Batjes, N. Overview of Procedures and Standards in Use at ISRIC WDC—Soils Report No. 2016/02 (ISRIC, 2016).
Jackson, R. B. et al. The ecology of soil carbon: pools, vulnerabilities, and biotic and abiotic controls. Annu. Rev. Ecol. Evol. System. 48, 419–445 (2017).
Holdridge, L. R. Determination of world plant formations from simple climatic data. Science 105, 367–368 (1947).
Holdridge, L. R. Life Zone Ecology (Tropcial Science Center, San Jose, 1967).
Post, W. M., Emanuel, W. R., Zinke, P. J. & Stangenberger, A. G. Soil carbon pools and world life zones. Nature 298, 156–159 (1982).
IPCC Climate Change 2013: The Physical Science Basis (eds. Stocker, T. F. et al.) (Cambridge Univ. Press, 2013).
Kögel‐Knabner, I. et al. Organo‐mineral associations in temperate soils: integrating biology, mineralogy, and organic matter chemistry. J. Plant Nutr. Soil Sci. 171, 61–82 (2008).
Trumbore, S. E., Chadwick, O. A. & Amundson, R. Rapid exchange between soil carbon and atmospheric carbon dioxide driven by temperature change. Science 272, 393–396 (1996).
Buettner, S. W., Kramer, M. G., Chadwick, O. A. & Thompson, A. Mobilization of colloidal carbon during iron reduction in basaltic soils. Geoderma 221, 139–145 (2014).
Harvard Dataverse (Harvard Univ., 2018); https://doi.org/10.7910/DVN/NGFY6A
McKeague, J. An evaluation of 0.1 M pyrophosphate and pyrophosphate-dithionite in comparison with oxalate as extractants of the accumulation products in podzols and some other soils. Can. J. Soil Sci. 47, 95–99 (1967).
Mehra, O. & Jackson, M. Iron oxide removal from soils and clays by a dithionite-citrate system buffered with sodium bicarbonate. Clays Clay Miner. 7, 317–327 (1958).
Franzmeier, D., Hajek, B. & Simonson, C. Use of amorphous material to identify spodic horizons. Soil Sci. Soc. Am. J. 29, 737–743 (1965).
Lalonde, K., Mucci, A., Ouellet, A. & Gélinas, Y. Preservation of organic matter in sediments promoted by iron. Nature 483, 198–200 (2012).
Walter, I. A. et al. ASCE’s standardized reference evapotranspiration equation. In Proc. Watershed Management and Operations Management 2000 (eds Flug, M., Frevert, D. & Watkins, D. W. Jr) 1–11 (American Society of Civil Engineering, 2000).
Aschonitis, V., Demertzi, K., Papamichail, D., Colombani, N. & Mastrocicco, M. Revisiting the Priestley–Taylor method for the assessment of reference crop evapotranspiration in Italy. J. Agrometeorol. 20, 5–18 (2015).
Aschonitis, V. G. et al. High-resolution global grids of revised Priestley–Taylor and Hargreaves–Samani coefficients for assessing ASCE-standardized reference crop evapotranspiration and solar radiation. Earth Syst. Sci. Data. 9, 615–638 (2017).
Itenfisu, D., Elliott, R. L., Allen, R. G. & Walter, I. A. Comparison of reference evapotranspiration calculations as part of the ASCE standardization effort. J. Irrig. Drain. Eng. 129, 440–448 (2003).
Zhang, K. et al. Vegetation greening and climate change promote multidecadal rises of global land evapotranspiration. Sci. Rep. 5, 15956 (2015).
Acknowledgements
The authors thank R. Johnson, D. Andreasen and G. Kahl for assistance with soil analyses. Soil sample preparation and analyses were conducted at the Stable Isotope and Organic Geochemistry Laboratory at Washington State University, Vancouver. This work was, in part, financially supported by National Research Initiative grant no. 2007–35107–18429 and from the USDA National Institute of Food and Agriculture grant no. 2017–05483. Soil samples were provided by NEON, which is a programme sponsored by the National Science Foundation and operated under a cooperative agreement with Battelle Memorial Institute.
Author information
Authors and Affiliations
Contributions
M.G.K. conceived of the study, designed and executed soil sample analyses, as well as global soil C and climate data set analyses. M.G.K. wrote the manuscript, to which both authors contributed substantial interpretation, discussion and text.
Corresponding author
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures 1–3
Rights and permissions
About this article
Cite this article
Kramer, M.G., Chadwick, O.A. Climate-driven thresholds in reactive mineral retention of soil carbon at the global scale. Nature Clim Change 8, 1104–1108 (2018). https://doi.org/10.1038/s41558-018-0341-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-018-0341-4
This article is cited by
-
Land-use induced soil carbon stabilization at the expense of rock derived nutrients: insights from pristine Andean soils
Scientific Reports (2023)
-
Coupled iron cycling and organic matter transformation across redox interfaces
Nature Reviews Earth & Environment (2023)
-
Integrating terrestrial and aquatic ecosystems to constrain estimates of land-atmosphere carbon exchange
Nature Communications (2023)
-
Sphagnum increases soil’s sequestration capacity of mineral-associated organic carbon via activating metal oxides
Nature Communications (2023)
-
Effect of mineral soil constituents on carbon characteristics of peatlands in aeolian environments of Iceland
Wetlands Ecology and Management (2023)