Abstract
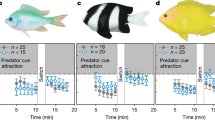
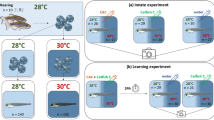
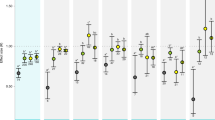
Survival of marine fishes that are exposed to elevated near-future CO2 levels is threatened by their altered responses to sensory cues. Here we demonstrate a physiological and molecular mechanism in the olfactory system that helps to explain altered behaviour under elevated CO2. We combine electrophysiology measurements and transcriptomics with behavioural experiments to investigate how elevated CO2 affects the olfactory system of European sea bass (Dicentrarchus labrax). When exposed to elevated CO2 (approximately 1,000 µatm), fish must be up to 42% closer to an odour source for detection, compared with current CO2 levels (around 400 µatm), decreasing their chances of detecting food or predators. Compromised olfaction correlated with the suppression of the transcription of genes involved in synaptic strength, cell excitability and wiring of the olfactory system in response to sustained exposure to elevated CO2 levels. Our findings complement the previously proposed impairment of γ-aminobutyric acid receptors, and indicate that both the olfactory system and central brain function are compromised by elevated CO2 levels.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Velez, Z. et al. Identification, release and olfactory detection of bile salts in the intestinal fluid of the Senegalese sole (Solea senegalensis). J. Comp. Physiol. A 195, 691–698 (2009).
Yacoob, S. Y. & Browman, H. I. Olfactory and gustatory sensitivity to some feed-related chemicals in the Atlantic halibut (Hippoglossus hippoglossus). Aquaculture 263, 303–309 (2007).
Munday, P. L. et al. Ocean acidification impairs olfactory discrimination and homing ability of a marine fish. Proc. Natl Acad. Sci. USA 106, 1848–1852 (2009).
Munday, P. L. et al. Replenishment of fish populations is threatened by ocean acidification. Proc. Natl Acad. Sci. USA 107, 12930–12934 (2010).
Yambe, H. et al. L-kynurenine, an amino acid identified as a sex pheromone in the urine of ovulated female masu salmon. Proc. Natl Acad. Sci. USA 103, 15370–15374 (2006).
Arvedlund, M., McCormick, M. I., Fautin, D. G. & Bildsøe, M. Host recognition and possible imprinting in the anemonefish Amphiprion melanopus (Pisces: Pomacentridae). Mar. Ecol. Prog. Ser. 188, 207–218 (1999).
Arvedlund, M. & Takemura, A. The importance of chemical environmental cues for juvenile Lethrinus nebulosus Forsskål (Lethrinidae, Teleostei) when settling into their first benthic habitat. J. Exp. Mar. Biol. Ecol. 338, 112–122 (2006).
Atema, J., Kingsford, M. J. & Gerlach, G. Larval reef fish could use odour for detection, retention and orientation to reefs. Mar. Ecol. Prog. Ser. 241, 151–160 (2002).
Gerlach, G., Atema, J., Kingsford, M. J., Black, K. P. & Miller-Sims, V. Smelling home can prevent dispersal of reef fish larvae. Proc. Natl Acad. Sci. USA 104, 858–863 (2007).
Vrieze, L. A. & Sorensen, P. W. Laboratory assessment of the role of a larval pheromone and natural stream odor in spawning stream localization by migratory sea lamprey (Petromyzon marinus). Can. J. Fish. Aquat. Sci. 58, 2374–2385 (2001).
Hamilton, T. J., Holcombe, A. & Tresguerres, M. CO2-induced ocean acidification increases anxiety in rockfish via alteration of GABAA receptor functioning. Proc. R. Soc. B 281, 20132509 (2014).
Jutfelt, F., Bresolin de Souza, K., Vuylsteke, A. & Sturve, J. Behavioural disturbances in a temperate fish exposed to sustained high-CO2 levels. PLoS ONE 8, e65825 (2013).
Ferrari, M. C. O. et al. Effects of ocean acidification on learning in coral reef fishes. PLoS ONE 7, e31478 (2012).
Dixson, D. L., Jennings, A. R., Atema, J. & Munday, P. L. Odor tracking in sharks is reduced under future ocean acidification conditions. Glob. Change Biol. 21, 1454–1462 (2015).
Green, L. & Jutfelt, F. Elevated carbon dioxide alters the plasma composition and behaviour of a shark. Biol. Lett. 10, 20140538 (2014).
Nilsson, G. E. et al. Near-future carbon dioxide levels alter fish behaviour by interfering with neurotransmitter function. Nat. Clim. Change 2, 201–204 (2012).
Chivers, D. P. et al. Impaired learning of predators and lower prey survival under elevated CO2: a consequence of neurotransmitter interference. Glob. Change Biol. 20, 515–522 (2013).
Watson, S.-A. et al. Marine mollusc predator-escape behaviour altered by near-future carbon dioxide levels. Proc. R. Soc. B 281, 20132377 (2014).
Heuer, R. M., Welch, M. J., Rummer, J. L., Munday, P. L. & Grosell, M. Altered brain ion gradients following compensation for elevated CO2 are linked to behavioural alterations in a coral reef fish. Sci. Rep. 6, 33216 (2016).
Leduc, A. O. H. C., Munday, P. L., Brown, G. E. & Ferrari, M. C. O. Effects of acidification on olfactory-mediated behaviour in freshwater and marine ecosystems: a synthesis. Phil. Trans. R. Soc. B 368, 20120447 (2013).
Fariña, A. C. et al. Lophius in the world: a synthesis on the common features and life strategies. ICES J. Mar. Sci. 65, 1272–1280 (2008).
Hara, T. The diversity of chemical stimulation in fish olfaction and gustation. Rev. Fish Biol. Fish. 4, 1–35 (1994).
Buchinger, T. J., Li, W. & Johnson, N. S. Bile salts as semiochemicals in fish. Chem. Senses 39, 647–654 (2014).
Leduc, A. O. H. C. et al. Ambient pH and the response to chemical alarm cues in juvenile Atlantic salmon: mechanisms of reduced behavioral responses. Trans. Am. Fish. Soc. 139, 117–128 (2010).
Lönnstedt, O. M. & McCormick, M. I. Chemical alarm cues inform prey of predation threat: the importance of ontogeny and concentration in a coral reef fish. Anim. Behav. 82, 213–218 (2011).
Frade, P., Hubbard, P. C., Barata, E. N. & Canario, A. V. M. Olfactory sensitivity of the Mozambique tilapia to conspecific odours. J. Fish Biol. 61, 1239–1254 (2002).
Hubbard, P. C., Barata, E. N. & Canário, A. V. M. Olfactory sensitivity of the gilthead seabream (Sparus auratus L) to conspecific body fluids. J. Chem. Ecol. 29, 2481–2498 (2003).
Haas, B. J. et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 8, 1494–1512 (2013).
Li, B. & Dewey, C. N. RSEM: accurate transcript quantification from RNA-seq data with or without a reference genome. BMC Bioinformatics 12, 323 (2011).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Malenka, R. C. & Nicoll, A. R. Long-term potentiation--a decade of progress? Science 285, 1870–1874 (1999).
Zhang, J. J., Okutani, F., Inoue, S. & Kaba, H. Activation of the mitogen-activated protein kinase/extracellular signal-regulated kinase signaling pathway leading to cyclic AMP response element-binding protein phosphorylation is required for the long-term facilitation process of aversive olfactory learning in young rats. Neuroscience 121, 9–16 (2003).
Deigweiher, K., Koschnick, N., Pörtner, H.-O. & Lucassen, M. Acclimation of ion regulatory capacities in gills of marine fish under environmental hypercapnia. Am. J. Physiol. 295, R1660–R1670 (2008).
Hashiguchi, Y., Furuta, Y. & Nishida, M. Evolutionary patterns and selective pressures of odorant/pheromone receptor gene families in teleost fishes. PLoS ONE 3, e4083 (2008).
Dubacq, C., Fouquet, C. & Trembleau, A. Making scent of the presence and local translation of odorant receptor mRNAs in olfactory axons. Dev. Neurobiol. 74, 259–268 (2014).
Benhaïm, D. et al. Early life behavioural differences in wild caught and domesticated sea bass (Dicentrarchus labrax). Appl. Anim. Behav. Sci. 141, 79–90 (2012).
Ferrari, M. C. O. et al. Intrageneric variation in antipredator responses of coral reef fishes affected by ocean acidification: implications for climate change projections on marine communities. Glob. Change Biol. 17, 2980–2986 (2011).
Cripps, I. L., Munday, P. L. & McCormick, M. I. Ocean acidification affects prey detection by a predatory reef fish. PLoS ONE 6, e22736 (2011).
Duteil M. et al. European sea bass show behavioural resilience to near-future ocean acidification. R. Soc. Open Sci. 3, 160656 (2016).
Munday, P. L. et al. Elevated CO2 affects the behavior of an ecologically and economically important coral reef fish. Mar. Biol. 160, 2137–2144 (2013).
Ou, M. et al. Responses of pink salmon to CO2-induced aquatic acidification. Nat. Clim. Change 5, 950–955 (2015).
Roggatz, C. C., Lorch, M., Hardege, J. D. & Benoit, D. M. Ocean acidification affects marine chemical communication by changing structure and function of peptide signalling molecules. Glob. Change Biol. 22, 3914–3926 (2016).
Schunter, C. et al. Molecular signatures of transgenerational response to ocean acidification in a species of reef fish. Nat. Clim. Change 6, 1014–1018 (2016).
Brauner, C. J. et al. Limited extracellular but complete intracellular acid–base regulation during short-term environmental hypercapnia in the armoured catfish, Liposarcus pardalis. J. Exp. Biol. 207, 3381–3390 (2004).
Esbaugh, A., Heuer, R. & Grosell, M. Impacts of ocean acidification on respiratory gas exchange and acid–base balance in a marine teleost, Opsanus beta. J. Comp. Physiol. B 182, 921–934 (2012).
Munday, P. L., Cheal, A. J., Dixson, D. L., Rummer, J. L. & Fabricius, K. E. Behavioural impairment in reef fishes caused by ocean acidification at CO2 seeps. Nat. Clim. Change 4, 487–492 (2014).
Welch, M. J., Watson, S.-A., Welsh, J. Q., McCormick, M. I. & Munday, P. L. Effects of elevated CO2 on fish behaviour undiminished by transgenerational acclimation. Nat. Clim. Change 4, 1086–1089 (2014).
Poulton, D. A., Porteus, C. S. & Simpson, S. D. Combined impacts of elevated CO2 and anthropogenic noise on European sea bass (Dicentrarchus labrax). ICES J. Mar. Sci. 74, 1230–1236 (2017).
Chivers, D., Puttlitz, M. & Blaustein, A. Chemical alarm signaling by reticulate sculpins, Cottus perplexus. Environ. Biol. Fish. 57, 347–352 (2000).
Field, D. et al. Open software for biologists: from famine to feast. Nat. Biotechnol. 24, 801–803 (2006).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Crusoe, M. R. et al. The khmer software package: enabling efficient nucleotide sequence analysis. F1000Research 4, 900 (2015).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Yates, A. et al. Ensembl 2016. Nucleic Acids Res. 44, D710–D716 (2016).
Buchfink, B., Xie, C. & Huson, D. H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods 12, 59–60 (2015).
Huson, D. H., Mitra, S., Ruscheweyh, H.-J., Weber, N. & Schuster, S. C. Integrative analysis of environmental sequences using MEGAN4. Genome Res. 21, 1552–1560 (2011).
Huang, D. W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4, 44–57 (2008).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 29, e45 (2001).
Filby, A. L. & Tyler, C. R. Appropriate ‘housekeeping’ genes for use in expression profiling the effects of environmental estrogens in fish. BMC Mol. Biol. 8, 10 (2007).
Hagey, L. R., Møller, P. R., Hofmann, A. F. & Krasowski, M. D. Diversity of bile salts in fish and amphibians: evolution of a complex biochemical pathway. Physiol. Biochem. Zool. 83, 308–321 (2010).
Ballantyne, J. S. Jaws: the inside story. The metabolism of elasmobranch fishes. Comp. Biochem. Physiol.B 118, 703–742 (1997).
Huertas, M. et al. Olfactory sensitivity to bile fluid and bile salts in the European eel (Anguilla anguilla), goldfish (Carassius auratus) and Mozambique tilapia (Oreochromis mossambicus) suggests a ‘broad range’ sensitivity not confined to those produced by conspecifics alone. J. Exp. Biol. 213, 308–317 (2010).
Zhang, C., Brown, S. & Hara, T. Biochemical and physiological evidence that bile acids produced and released by lake char (Salvelinus namaycush) function as chemical signals. J. Comp. Physiol. B 171, 161–171 (2001).
Hubbard, P., Barata, E. N., Ozório, R. A., Valente, L. P. & Canário, A. M. Olfactory sensitivity to amino acids in the blackspot sea bream (Pagellus bogaraveo): a comparison between olfactory receptor recording techniques in seawater. J. Comp. Physiol. A 197, 839–849 (2011).
Hubbard, P. C., Barata, E. N. & Canario, A. V. Olfactory sensitivity to changes in environmental [Ca2+] in the marine teleost Sparus aurata. J. Exp. Biol. 203, 3821–3829 (2000).
Velez, Z., Hubbard, P. C., Barata, E. N. & Canário, A. V. M. Olfactory transduction pathways in the Senegalese sole Solea senegalensis. J. Fish Biol. 83, 501–514 (2013).
Dickson, A. G., Sabine, C. L. & Christian, J. R. (eds) Guide to Best Practices for Ocean CO 2 Measurements PICES Special Publication 3 (North Pacific Marine Science Organization, 2007).
Meredith, T. L., Caprio, J. & Kajiura, S. M. Sensitivity and specificity of the olfactory epithelia of two elasmobranch species to bile salts. J. Exp. Biol. 215, 2660–2667 (2012).
Acknowledgements
We thank L. Hagey and A. Hofmann (UCSD) for their gift of cyprinol sulfate and scymnol sulfate, the Aquatic Research Centre (ARC) staff at the University of Exeter for their assistance with fish husbandry and experimental setup, B. Verbruggen for helpful bioinformatics advice and L. Salisbury for help with tissue sampling. This study was supported by grants from Association of European Marine Biology Laboratories (227799), the Natural Environment Research Council (R.W.W.; NE/H017402/1), the Biotechnology and Biological Sciences Research Council (R.W.W.; BB/D005108/1), Fundação para a Ciência e Tecnologia (Portuguese Science Ministry) (UID/Multi/04326/2013) and a Royal Society Newton International Fellowship to C.S.P. C.S.P. is also a beneficiary of a Starting Grant from AXA.
Author information
Authors and Affiliations
Contributions
C.S.P. and R.W.W. designed the behavioural experiments. C.S.P. performed the experiments and analysed those data; C.S.P., P.C.H., A.V.M.C. and R.W.W. designed the electrophysiology study, C.S.P. and P.C.H. performed the electrophysiology experiments. C.S.P., T.M.U.W., R.v.A. and E.M.S. designed the transcriptomics experiments, C.S.P. performed the experiments and constructed the libraries. C.S.P. performed the bioinformatics analysis and interpreted the results with help from T.M.U.W., R.v.A. and E.M.S. All authors contributed to and provided feedback on various drafts of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures 1–8, Supplementary tables 1–13, Supplementary References
Supplementary Data 1
Lists of differentially expressed genes in the olfactory epithelium and olfactory bulb at 2 and 7 days of exposure to control and high CO2
Rights and permissions
About this article
Cite this article
Porteus, C.S., Hubbard, P.C., Uren Webster, T.M. et al. Near-future CO2 levels impair the olfactory system of a marine fish. Nature Clim Change 8, 737–743 (2018). https://doi.org/10.1038/s41558-018-0224-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-018-0224-8
This article is cited by
-
Response of CRH system in brain and gill of marine medaka to seawater acidification
Fish Physiology and Biochemistry (2024)
-
Does mutualism provide additional indirect benefits? Behavioral indicators of chemical communication in a temporally dynamic fish-mussel mutualism
Behavioral Ecology and Sociobiology (2024)
-
Neuromolecular responses in disrupted mutualistic cleaning interactions under future environmental conditions
BMC Biology (2023)
-
Transgenerational exposure to ocean acidification impacts the hepatic transcriptome of European sea bass (Dicentrarchus labrax)
BMC Genomics (2023)
-
The extensive transgenerational transcriptomic effects of ocean acidification on the olfactory epithelium of a marine fish are associated with a better viral resistance
BMC Genomics (2022)