Abstract
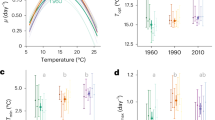
The cyanobacterium Trichodesmium fixes as much as half of the nitrogen (N2) that supports tropical open-ocean biomes, but its growth is frequently limited by iron (Fe) availability1,2. How future ocean warming may interact with this globally widespread Fe limitation of Trichodesmium N2 fixation is unclear3. Here, we show that the optimum growth temperature of Fe-limited Trichodesmium is ~5 °C higher than for Fe-replete cells, which results in large increases in growth and N2 fixation under the projected warmer Fe-deplete sea surface conditions. Concurrently, the cellular Fe content decreases as temperature rises. Together, these two trends result in thermally driven increases of ~470% in Fe-limited cellular iron use efficiencies (IUEs), defined as the molar quantity of N2 fixed by Trichodesmium per unit time per mole of cellular Fe (mol N2 fixed h–1 mol Fe–1), which enables Trichodesmium to much more efficiently leverage the scarce available Fe supplies to support N2 fixation. Modelling these results in the context of the IPCC representative concentration pathway (RCP) 8.5 global warming scenario4 predicts that IUEs of N2 fixers could increase by ~76% by 2100, and largely alleviate the prevailing Fe limitation across broad expanses of the tropical Pacific and Indian Oceans. Thermally enhanced cyanobacterial IUEs could increase future global marine N2 fixation by ~22% over the next century, and thus profoundly alter the biology and biogeochemistry of open-ocean ecosystems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Capone, D. G., Zehr, J. P., Paerl, H. W., Bergman, B. & Carpenter, E. J. Trichodesmium, a globally significant marine cyanobacterium. Science 276, 1221–1229 (1997).
Sohm, J. A., Webb, E. A. & Capone, D. G. Emerging patterns of marine nitrogen fixation. Nat. Rev. Microbiol. 9, 499–508 (2011).
Hutchins, D. A. & Boyd, P. Marine phytoplankton and the changing ocean iron cycle. Nat. Clim. Change 6, 1072–1079 (2016).
IPCC Climate Change 2013: The Physical Science Basis (eds Stocker, T. F. et al.) (Cambridge Univ. Press, 2013).
Breitbarth, E., Oschilles, A. & LaRoche, J. L. Physiological constraints on the global distribution of Trichodesmium—effect of temperature on diazotrophy. Biogeosciences 4, 53–61 (2007).
Fu, F.-X. et al. Differing responses of marine N2-fixers to warming and consequences for future diazotroph community structure. Aquat. Microb. Ecol. 72, 33–46 (2014).
Yvon-Durocher, G. et al. Reconciling the temperature dependence of respiration across timescales and ecosystem types. Nature 487, 472–476 (2012).
Fu, F. X., Warner, M. E., Zhang, Y., Feng, Y. & Hutchins, D. A. Effects of increased temperature and CO2 on photosynthesis, growth, and elemental ratios in marine Synechococcus and Prochlorococcus (Cyanobacteria). J. Phycol. 43, 485–496 (2007).
Sunda, W. G. & Huntsman, S. A. Interactive effects of light and temperature on iron limitation in a marine diatom: implications for marine productivity and carbon cycling. Limnol. Oceanogr. 56, 1475–1488 (2011).
Boyd, P. et al. Physiological responses of a Southern Ocean diatom to complex future ocean conditions. Nat. Clim. Change 6, 207–213 (2016).
Kustka, A., Sañudo-Wilhelmy, S. A., Carpenter, E. J., Capone, D. G. & Raven, J. A. A revised estimate of the iron use efficiency of nitrogen fixation, with special reference to the marine cyanobacterium Trichodesmium (Cyanophyta). J. Phycol. 39, 12–25 (2003).
Sañudo-Wilhelmy, S. A. et al. Phosphorus limitation of nitrogen fixation by Trichodesmium in the central Atlantic Ocean. Nature 411, 66–69 (2001).
Walworth, N. G. et al. Mechanisms of increased Trichodesmium fitness under iron and phosphorus co-limitation in the present and future ocean. Nat. Commun. 7, 12081 (2016).
Xu, K., Fu, F.-X. & Hutchins, D. A. Comparative responses of two dominant Antarctic phytoplankton taxa to interactions between ocean acidification, warming, irradiance, and iron availability. Limnol. Oceanogr. 59, 919–931 (2014).
Toseland, A. et al. The impact of temperature on marine phytoplankton resource allocation and metabolism. Nat. Clim. Change 3, 979–984 (2013).
Fasham, M. J. R., Ducklow, H. W. & McKelvie, S. M. A nitrogen-based model of plankton dynamics in the oceanic mixed layer. J. Mar. Res. 48, 591–639 (1990).
Geider, R. J., MacIntyre, H. L. & Kana, T. M. Dynamic model of phytoplankton growth and acclimation: responses of the balanced growth rate and the chlorophyll a:carbon ratio to light, nutrient-limitation and temperature. Mar. Ecol. Progr. Ser. 148, 187–200 (1997).
Moore, J. K., Lindsay, K., Doney, S. C., Long, M. C. & Misumi, K. Marine ecosystem dynamics and biogeochemical cycling in the Community Earth System Model [CESM1(BGC)]: comparison of the 1990s with the 2090s under the RCP4.5 and RCP8.5 scenarios. J. Clim. 26, 9291–9312 (2013).
Behrenfeld, M. J. & Kolber, Z. S. Widespread iron limitation of phytoplankton in the South Pacific. Ocean. Sci. 283, 840–843 (1999).
Dutkiewicz, S., Ward, B. A., Monteiro, F. M. & Follows, M. J. Interconnection of nitrogen fixers and iron in the Pacific Ocean: theory and numerical simulations. Glob. Biogeochem. Cycles 26, GB1012 (2012).
Carpenter, E. J. and Capone, D. G. in Nitrogen in the Marine Environment 2nd edn (eds Capone, D. G. et al.) Ch. 4 (Academic, London, 2008).
Hutchins, D. A., Fu, F.-X., Webb, E. A., Walworth, N. & Tagliabue, A. Taxon-specific responses of marine nitrogen fixers to elevated carbon dioxide concentrations. Nat. Geosci. 6, 790–795 (2013).
Hutchins, D. A. et al. Irreversibly increased nitrogen fixation in Trichodesmium experimentally adapted to elevated carbon dioxide. Nat. Commun. 6, 8155 (2015).
Fu, F.-X. et al. Interactions between changing pCO2, N2 fixation, and Fe limitation in the marine unicellular cyanobacterium Crocosphaera. Limnol. Oceanogr. 53, 2472–2484 (2008).
Doney, S. C. et al. Climate change impacts on marine ecosystems. Annu. Rev. Mar. Sci. 4, 11–37 (2012).
Boyce, D. G., Lewis, M. R., M. R. & Worm, M. R. Global phytoplankton decline over the past century. Nature 466, 591–596 (2010).
Behrenfeld, M. J. et al. Revaluating ocean warming impacts on global phytoplankton. Nat. Clim. Change 6, 323–330 (2016).
Acknowledgements
This study was supported by US National Science Foundation grants OCE 1657757, OCE 1638804, OCE 1538525, OCE 1260233 and OCE 1260490 and National Natural Science Foundation of China grants 31470171 and 31770033.
Author information
Authors and Affiliations
Contributions
H.B.J., D.A.H. and F.-X.F. contributed to conceiving and planning the experiments, H.B.J., F.-X.F., P.P.Q., X.-W.W. and Z.Z. performed the lab experiments, P.P.G. and S.A.S.-W. contributed analytical work, S.-R.C. and N.M.L. contributed modelling work, H.B.J. and D.A.H. contributed to writing the paper and all of the authors contributed comments, revisions and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods and Supplementary Methods’s References, Supplementary Figures 1–15, Supplementary Tables 1–6
Rights and permissions
About this article
Cite this article
Jiang, HB., Fu, FX., Rivero-Calle, S. et al. Ocean warming alleviates iron limitation of marine nitrogen fixation. Nature Clim Change 8, 709–712 (2018). https://doi.org/10.1038/s41558-018-0216-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-018-0216-8
This article is cited by
-
Global patterns and predictors of C:N:P in marine ecosystems
Communications Earth & Environment (2022)
-
Molecular mechanisms underlying iron and phosphorus co-limitation responses in the nitrogen-fixing cyanobacterium Crocosphaera
The ISME Journal (2022)
-
The marine nitrogen cycle: new developments and global change
Nature Reviews Microbiology (2022)
-
Nanomolar phosphate supply and its recycling drive net community production in the subtropical North Pacific
Nature Communications (2021)
-
Transient exposure to novel high temperatures reshapes coastal phytoplankton communities
The ISME Journal (2020)