Abstract
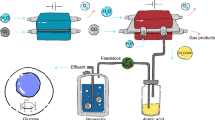
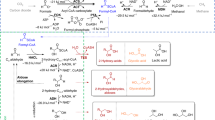
Living systems provide a promising approach to chemical synthesis, having been optimized by evolution to convert renewable carbon sources, such as glucose, into an enormous range of small molecules. However, a large number of synthetic structures can still be difficult to obtain solely from cells, such as unsubstituted hydrocarbons. In this work, we demonstrate the use of a dual cellular–heterogeneous catalytic strategy to produce olefins from glucose using a selective hydrolase to generate an activated intermediate that is readily deoxygenated. Using a new family of iterative thiolase enzymes, we genetically engineered a microbial strain that produces 4.3 ± 0.4 g l−1 of fatty acid from glucose with 86% captured as 3-hydroxyoctanoic and 3-hydroxydecanoic acids. This 3-hydroxy substituent serves as a leaving group that enables heterogeneous tandem decarboxylation–dehydration routes to olefinic products on Lewis acidic catalysts without the additional redox input required for enzymatic or chemical deoxygenation of simple fatty acids.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated and analysed during this study are included in the published article, source data files or supporting information files. All plasmids and strains generated in this study are available by request. Source data are provided with this paper.
References
Nielsen, J. & Keasling, J. D. Engineering cellular metabolism. Cell 164, 1185–1197 (2016).
Woolston, B. M., Edgar, S. & Stephanopoulos, G. Metabolic engineering: past and future. Annu. Rev. Chem. Biomol. Eng. 4, 259–288 (2013).
Lee, J. W. et al. Systems metabolic engineering of microorganisms for natural and non-natural chemicals. Nat. Chem. Biol. 8, 536–546 (2012).
Liao, J. C., Mi, L., Pontrelli, S. & Luo, S. Fuelling the future: microbial engineering for the production of sustainable biofuels. Nat. Rev. Micro. 14, 288–304 (2016).
Schirmer, A., Rude, M. A., Li, X., Popova, E. & Del Cardayre, S. B. Microbial biosynthesis of alkanes. Science 329, 559–562 (2010).
Rude, M. A. et al. Terminal olefin (1-alkene) biosynthesis by a novel P450 fatty acid decarboxylase from Jeotgalicoccus species. Appl. Environ. Microbiol. 77, 1718–1727 (2011).
Rui, Z. et al. Microbial biosynthesis of medium-chain 1-alkenes by a nonheme iron oxidase. Proc. Natl Acad. Sci. USA 111, 18237–18242 (2014).
Rui, Z., Harris, N. C., Zhu, X., Huang, W. & Zhang, W. Discovery of a family of desaturase-like enzymes for 1-alkene biosynthesis. ACS Catal. 5, 7091–7094 (2015).
Gu, L. et al. Polyketide decarboxylative chain termination preceded by O-sulfonation in curacin A biosynthesis. J. Am. Chem. Soc. 131, 16033–16035 (2009).
Christenson, J. K. et al. OleB from bacterial hydrocarbon biosynthesis is a β-lactone decarboxylase that shares key features with haloalkane dehalogenases. Biochemistry 56, 5278–5287 (2017).
Bernard, A. et al. Reconstitution of plant alkane biosynthesis in yeast demonstrates that Arabidopsis ECERIFERUM1 and ECERIFERUM3 are core components of a very-long-chain alkane synthesis complex. Plant Cell 24, 3106–3118 (2012).
Bond-Watts, B. B., Bellerose, R. J. & Chang, M. C. Y. Enzyme mechanism as a kinetic control element for designing synthetic biofuel pathways. Nat. Chem. Biol. 7, 222–227 (2011).
Shen, C. R. et al. Driving forces enable high-titer anaerobic 1-butanol synthesis in Escherichia coli. Appl. Environ. Microbiol. 77, 2905–2915 (2011).
Rudroff, F. et al. Opportunities and challenges for combining chemo- and biocatalysis. Nat. Catal. 1, 12–22 (2018).
Abdelrahman, O. A. et al. Renewable isoprene by sequential hydrogenation of itaconic acid and dehydra-decyclization of 3-methyl-tetrahydrofuran. ACS Catal. 7, 1428–1431 (2017).
Suastegui, M. et al. Combining metabolic engineering and electrocatalysis: application to the production of polyamides from sugar. Angew. Chem. Int. Ed. 55, 2368–2373 (2016).
Choi, Y. J. & Lee, S. Y. Microbial production of short-chain alkanes. Nature 502, 571–574 (2013).
Steen, E. J. et al. Microbial production of fatty-acid-derived fuels and chemicals from plant biomass. Nature 463, 559–562 (2010).
Lennen, R. M., Braden, D. J., West, R. M., Dumesic, J. A. & Pfleger, B. F. A process for microbial hydrocarbon synthesis: overproduction of fatty acids in Escherichia coli and catalytic conversion to alkanes. Biotechnol. Bioeng. 106, 193–202 (2010).
Yan, Q. et al. Metabolic engineering of β-oxidation to leverage thioesterases for production of 2-heptanone, 2-nonanone and 2-undecanone. Metab. Eng. 61, 335–343 (2020).
Blazeck, J., Liu, L., Knight, R. & Alper, H. S. Heterologous production of pentane in the oleaginous yeast Yarrowia lipolytica. J. Biotechnol. 165, 184–194 (2013).
Kim, H. M., Chae, T. U., Choi, S. Y., Kim, W. J. & Lee, S. Y. Engineering of an oleaginous bacterium for the production of fatty acids and fuels. Nat. Chem. Biol. 15, 721–729 (2019).
Magnuson, K., Jackowski, S., Rock, C. O. & Cronan, J. E. Regulation of fatty-acid biosynthesis in Escherichia coli. Microbiol. Rev. 57, 522–542 (1993).
Dellomonaco, C., Clomburg, J. M., Miller, E. N. & Gonzalez, R. Engineered reversal of the β-oxidation cycle for the synthesis of fuels and chemicals. Nature 476, 355–359 (2011).
Sheppard, M. J., Kunjapur, A. M. & Prather, K. L. J. Modular and selective biosynthesis of gasoline-range alkanes. Metab. Eng. 33, 28–40 (2016).
Haapalainen, A. M., Meriläinen, G. & Wierenga, R. K. The thiolase superfamily: condensing enzymes with diverse reaction specificities. Trends Biochem. Sci. 31, 64–71 (2006).
Spratt, S. K., Black, P. N., Ragozzino, M. M. & Nunn, W. D. Cloning, mapping, and expression of genes involved in the fatty acid-degradative multienzyme complex of Escherichia coli. J. Bacteriol. 158, 535–542 (1984).
Atkinson, H. J., Morris, J. H., Ferrin, T. E. & Babbitt, P. C. Using sequence similarity networks for visualization of relationships across diverse protein superfamilies. PLoS ONE 4, e4345 (2009).
Wittkop, T. et al. Partitioning biological data with transitivity clustering. Nat. Methods 7, 419–420 (2010).
Masamune, S., Walsh, C. T., Sinskey, A. J. & Peoples, O. Poly-(R)-3-hydroxybutyrate (PHB) biosynthesis: mechanistic studies on the biological Claisen condensation catalyzed by β-ketoacyl thiolase. Pure Appl. Chem. 61, 303–312 (1989).
Slater, S. et al. Multiple β-ketothiolases mediate poly(β-hydroxyalkanoate) copolymer synthesis in Ralstonia eutropha. J. Bacteriol. 180, 1979–1987 (1998).
Teufel, R. et al. Bacterial phenylalanine and phenylacetate catabolic pathway revealed. Proc. Natl Acad. Sci. USA 107, 14390–14395 (2010).
Toomey, R. E. & Wakil, S. J. Studies on the mechanism of fatty acid synthesis XV. Preparation and general properties of β-ketoacyl acyl carrier protein reductase from Escherichia coli. Biochim. Biophys. Acta Lipids Lipid Metab. 116, 189–197 (1966).
Park, S. J. & Lee, S. Y. Identification and characterization of a new enoyl coenzyme A hydratase involved in biosynthesis of medium-chain-length polyhydroxyalkanoates in recombinant Escherichia coli. J. Bacteriol. 185, 5391–5397 (2003).
Campbell, J. W. & Cronan, J. E. The enigmatic Escherichia coli fadE gene is yafH. J. Bacteriol. 184, 3759–3764 (2002).
Engemann, C. et al. Identification and functional characterization of genes and corresponding enzymes involved in carnitine metabolism of Proteussp. Arch. Microbiol. 183, 176–189 (2005).
Bond-Watts, B. B., Weeks, A. M. & Chang, M. C. Y. Biochemical and structural characterization of the trans-enoyl-CoA reductase from Treponema denticola. Biochemistry 51, 6827–6837 (2012).
Pollard, M. R., Anderson, L., Fan, C., Hawkins, D. J. & Davies, H. M. A specific acyl-ACP thioesterase implicated in medium-chain fatty acid production in immature cotyledons of Umbellularia californica. Arch. Biochem. Biophys. 284, 306–312 (1991).
Dehesh, K., Edwards, P., Hayes, T., Cranmer, A. M. & Fillatti, J. Two novel thioesterases are key determinants of the bimodal distribution of acyl chain length of Cuphea palustris seed oil. Plant Physiol. 110, 203–210 (1996).
Grisewood, M. J. et al. Computational redesign of acyl-ACP thioesterase with improved selectivity toward medium-chain-length fatty acids. ACS Catal. 7, 3837–3849 (2017).
Jing, F. et al. Phylogenetic and experimental characterization of an acyl-ACP thioesterase family reveals significant diversity in enzymatic specificity and activity. BMC Biochem. 12, 44 (2011).
Campbell, J. W., Morgan-Kiss, R. M. & Cronan, J. E. A new Escherichia coli metabolic competency: growth on fatty acids by a novel anaerobic β-oxidation pathway. Mol. Microbiol. 47, 793–805 (2003).
McMurry, L. M., Oethinger, M. & Levy, S. B. Triclosan targets lipid synthesis. Nature 394, 531–532 (1998).
Mäki-Arvela, P., Kubickova, I., Snåre, M., Eränen, K. & Murzin, D. Y. Catalytic deoxygenation of fatty acids and their derivatives. Energy Fuels 21, 30–41 (2007).
Tanzawa, T. & Schwartz, J. Catalytic conversion of β-hydroxy carboxylic acids to olefins by tungsten(VI) complexes: a new acyl group transfer catalyst. Organometallics 9, 3026–3027 (1990).
Wang, D., Hakim, S. H., Martin Alonso, D. & Dumesic, J. A. A highly selective route to linear alpha olefins from biomass-derived lactones and unsaturated acids. Chem. Commun. 49, 7040–7043 (2013).
Chatterjee, A., Hopen Eliasson, S. H. & Jensen, V. R. Selective production of linear α-olefins via catalytic deoxygenation of fatty acids and derivatives. Catal. Sci. Technol. 8, 1487–1499 (2018).
Chen, C. S. H. & Bridger, R. F. Shape-selective oligomerization of alkenes to near-linear hydrocarbons by zeolite catalysis. J. Catal. 161, 687–693 (1996).
O’Connor, C. T. & Kojima, M. Alkene oligomerization. Catal. Today 6, 329–349 (1990).
Abdelrahman, O. A. et al. Biomass-derived butadiene by dehydra-decyclization of tetrahydrofuran. ACS Sustain. Chem. Eng. 5, 3732–3736 (2017).
Acknowledgements
This work was funded by the generous support of the National Science Foundation through a CAREER Award (029504-003) to M.C.Y.C. and the Center for Sustainable Polymers, a National Science Foundation-supported Center for Chemical Innovation (CHE-1901635). H.S. acknowledges support from the Camille and Henry Dreyfus Postdoctoral Program in Environmental Chemistry. Z.Q.W. also acknowledges the generous support from the Research Foundation for the State University of New York (71272-ZQW).
Author information
Authors and Affiliations
Contributions
Z.Q.W., H.S., N.H., D.S.P., P.J.D. and M.C.Y.C designed the research, Z.Q.W., H.S., E.J.K., N.H., D.S.P. and Y.M. carried out the experiments, Z.Q.W., H.S., E.J.K., N.H., D.S.P., G.K., P.J.D. and M.C.Y.C analysed the data and Z.Q.W., H.S., G.K., P.J.D. and M.C.Y.C wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Stephen Wallace and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary materials, methods, figures, tables, and references.
Supplementary Data 1
Raw data for Supplementary Fig. S2C.
Supplementary Data 2
Raw data for Supplementary Fig. S3.
Supplementary Data 3
Raw data for Supplementary Fig. S4.
Supplementary Data 4
Raw data for Supplementary Fig. S5.
Supplementary Data 5
Raw data for Supplementary Fig. S6.
Supplementary Data 6
Raw data for Supplementary Fig. S7.
Supplementary Data 7
Raw data for Supplementary Fig. S8.
Supplementary Data 8
Raw data for Supplementary Fig. S9.
Source data
Source Data Fig. 3
Raw data for Fig. 3.
Source Data Fig. 4
Raw data for Fig. 4.
Source Data Fig. 5
Raw data for Fig. 5.
Rights and permissions
About this article
Cite this article
Wang, Z.Q., Song, H., Koleski, E.J. et al. A dual cellular–heterogeneous catalyst strategy for the production of olefins from glucose. Nat. Chem. 13, 1178–1185 (2021). https://doi.org/10.1038/s41557-021-00820-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00820-0
This article is cited by
-
The non-mevalonate pathway requires a delicate balance of intermediates to maximize terpene production
Applied Microbiology and Biotechnology (2024)
-
Two steps to sustainable polymers
Nature Chemistry (2021)