Abstract
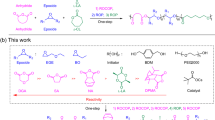
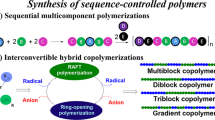
Controlling monomer sequence and dispersity in synthetic macromolecules is a major goal in polymer science as both parameters determine materials’ properties and functions. However, synthetic approaches that can simultaneously control both sequence and dispersity remain experimentally unattainable. Here we report a simple, one pot and rapid synthesis of sequence-controlled multiblocks with on-demand control over dispersity while maintaining a high livingness, and good agreement between theoretical and experimental molecular weights and quantitative yields. Key to our approach is the regulation in the activity of the chain transfer agent during a controlled radical polymerization that enables the preparation of multiblocks with gradually ascending (Ɖ = 1.16 → 1.60), descending (Ɖ = 1.66 → 1.22), alternating low and high dispersity values (Ɖ = 1.17 → 1.61 → 1.24 → 1.70 → 1.26) or any combination thereof. We further demonstrate the potential of our methodology through the synthesis of highly ordered pentablock, octablock and decablock copolymers, which yield multiblocks with concurrent control over both sequence and dispersity.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data and are available within this article and its Supplementary Information. Source data are provided with this paper.
References
Lutz, J.-F., Ouchi, M., Liu, D. R. & Sawamoto, M. Sequence-controlled polymers. Science 341, 1238149 (2013).
Ouchi, M., Badi, N., Lutz, J.-F. & Sawamoto, M. Single-chain technology using discrete synthetic macromolecules. Nat. Chem. 3, 917–924 (2011).
Lutz, J. F. Defining the field of sequence‐controlled polymers. Macromol. Rapid Commun. 38, 1700582 (2017).
De Neve, J., Haven, J. J., Maes, L. & Junkers, T. Sequence-definition from controlled polymerization: the next generation of materials. Polym. Chem. 9, 4692–4705 (2018).
Haven, J. J. et al. Elements of RAFT navigation: RAFT 20 years later: RAFT-synthesis of uniform, sequence-defined (co)polymers. ACS Symp. Ser. 1284, 77–103 (2018).
Bates, F. S. et al. Multiblock polymers: panacea or Pandora’s box? Science 336, 434–440 (2012).
Parkatzidis, K., Wang, H. S., Truong, N. P. & Anastasaki, A. Recent developments and future challenges in controlled radical polymerization: a 2020 update. Chem 6, 1575–1588 (2020).
Merrifield, R. B. Solid phase peptide synthesis. I. The synthesis of a tetrapeptide. J. Am. Chem. Soc. 85, 2149–2154 (1963).
Zamfir, M. & Lutz, J.-F. Ultra-precise insertion of functional monomers in chain-growth polymerizations. Nat. Commun. 3, 1138 (2012).
McHale, R., Patterson, J. P., Zetterlund, P. B. & O’Reilly, R. K. Biomimetic radical polymerization via cooperative assembly of segregating templates. Nat. Chem. 4, 491–497 (2012).
Schoonen, L. & van Hest, J. C. Compartmentalization approaches in soft matter science: from nanoreactor development to organelle mimics. Adv. Mater. 28, 1109–1128 (2016).
Pfeifer, S. & Lutz, J.-F. A facile procedure for controlling monomer sequence distribution in radical chain polymerizations. J. Am. Chem. Soc. 129, 9542–9543 (2007).
Nakatani, K., Ogura, Y., Koda, Y., Terashima, T. & Sawamoto, M. Sequence-regulated copolymers via tandem catalysis of living radical polymerization and in situ transesterification. J. Am. Chem. Soc. 134, 4373–4383 (2012).
Pfeifer, S., Zarafshani, Z., Badi, N. & Lutz, J.-F. Liquid-phase synthesis of block copolymers containing sequence-ordered segments. J. Am. Chem. Soc. 131, 9195–9197 (2009).
Dong, R. et al. Sequence-defined multifunctional polyethers via liquid-phase synthesis with molecular sieving. Nat. Chem. 11, 136–145 (2019).
Beyer, V. P., Kim, J. & Becer, C. R. Synthetic approaches for multiblock copolymers. Polym. Chem. 11, 1271–1291 (2020).
Elacqua, E. et al. Supramolecular multiblock copolymers featuring complex secondary structures. J. Am. Chem. Soc. 139, 12240–12250 (2017).
Heiler, C., Offenloch, J. T., Blasco, E. & Barner-Kowollik, C. Photochemically induced folding of single chain polymer nanoparticles in water. ACS Macro Lett. 6, 56–61 (2017).
Zhang, J. et al. Synthesis of sequence-controlled multiblock single chain nanoparticles by a stepwise folding–chain extension–folding process. Macromolecules 49, 8933–8942 (2016).
Guimarães, T. R. et al. Nano-engineered multiblock copolymer nanoparticles via reversible addition–fragmentation chain transfer emulsion polymerization. Macromolecules 52, 2965–2974 (2019).
Anastasaki, A. et al. One‐pot synthesis of ABCDE multiblock copolymers with hydrophobic, hydrophilic, and semi‐fluorinated segments. Angew. Chem. Int. Ed. 56, 14483–14487 (2017).
Barbon, S. M. et al. Elucidating the effect of sequence and degree of polymerization on antimicrobial properties for block copolymers. Polym. Chem. 11, 84–90 (2020).
Kuroki, A. et al. Sequence control as a powerful tool for improving the selectivity of antimicrobial polymers. ACS Appl. Mater. Inter. 9, 40117–40126 (2017).
Judzewitsch, P. R., Nguyen, T. K., Shanmugam, S., Wong, E. H. & Boyer, C. Towards sequence‐controlled antimicrobial polymers: effect of polymer block order on antimicrobial activity. Angew. Chem. 130, 4649–4654 (2018).
Zhang, J. et al. Effect of hydrophilic monomer distribution on self‐assembly of a pH‐responsive copolymer: spheres, worms and vesicles from a single copolymer composition. Angew. Chem. Int. Ed. 59, 2–8 (2020).
Chin, S. M., He, H., Konkolewicz, D. & Matyjaszewski, K. Synthesis of triblock and multiblock methacrylate polymers and self‐assembly of stimuli responsive triblock polymers. J. Polym. Sci. Pol. Chem. 52, 2548–2555 (2014).
Shanmugam, S. & Boyer, C. Stereo-, temporal and chemical control through photoactivation of living radical polymerization: synthesis of block and gradient copolymers. J. Am. Chem. Soc. 137, 9988–9999 (2015).
Xu, J., Jung, K., Atme, A., Shanmugam, S. & Boyer, C. A robust and versatile photoinduced living polymerization of conjugated and unconjugated monomers and its oxygen tolerance. J. Am. Chem. Soc. 136, 5508–5519 (2014).
Soeriyadi, A. H., Boyer, C., Nyström, F., Zetterlund, P. B. & Whittaker, M. R. High-order multiblock copolymers via iterative Cu(0)-mediated radical polymerizations (SET-LRP): toward biological precision. J. Am. Chem. Soc. 133, 11128–11131 (2011).
Boyer, C., Derveaux, A., Zetterlund, P. B. & Whittaker, M. R. Synthesis of multi-block copolymer stars using a simple iterative Cu(0)-mediated radical polymerization technique. Polym. Chem. 3, 117–123 (2012).
Boyer, C., Soeriyadi, A. H., Zetterlund, P. B. & Whittaker, M. R. Synthesis of complex multiblock copolymers via a simple iterative Cu(0)-mediated radical polymerization approach. Macromolecules 44, 8028–8033 (2011).
Engelis, N. G. et al. Sequence-controlled methacrylic multiblock copolymers via sulfur-free RAFT emulsion polymerization. Nat. Chem. 9, 171–178 (2017).
Anastasaki, A. et al. Photoinduced sequence-control via one pot living radical polymerization of acrylates. Chem. Sci. 5, 3536–3542 (2014).
Zhang, Q. et al. Sequence‐controlled multi‐block glycopolymers to inhibit DC‐SIGN‐gp120 binding. Angew. Chem. Int. Ed. 52, 4435–4439 (2013).
Chuang, Y.-M., Ethirajan, A. & Junkers, T. Photoinduced sequence-controlled copper-mediated polymerization: synthesis of decablock copolymers. ACS Macro Lett. 3, 732–737 (2014).
Wenn, B., Martens, A., Chuang, Y.-M., Gruber, J. & Junkers, T. Efficient multiblock star polymer synthesis from photo-induced copper-mediated polymerization with up to 21 arms. Polym. Chem. 7, 2720–2727 (2016).
Debuigne, A., Detrembleur, C., Jérôme, C. & Junkers, T. Straightforward synthesis of symmetrical multiblock copolymers by simultaneous block extension and radical coupling reactions. Macromolecules 46, 8922–8931 (2013).
Gody, G., Maschmeyer, T., Zetterlund, P. B. & Perrier, S. Rapid and quantitative one-pot synthesis of sequence-controlled polymers by radical polymerization. Nat. Commun. 4, 2505 (2013).
Gody, G., Barbey, R., Danial, M. & Perrier, S. Ultrafast RAFT polymerization: multiblock copolymers within minutes. Polym. Chem. 6, 1502–1511 (2015).
Clothier, G. K. et al. Exploitation of the nanoreactor concept for efficient synthesis of multiblock copolymers via macroRAFT-mediated emulsion polymerization. ACS Macro Lett. 8, 989–995 (2019).
Gody, G., Maschmeyer, T., Zetterlund, P. B. & Perrier, S. B. Pushing the limit of the RAFT process: multiblock copolymers by one-pot rapid multiple chain extensions at full monomer conversion. Macromolecules 47, 3451–3460 (2014).
Anastasaki, A. et al. Photoinduced synthesis of α,ω-telechelic sequence-controlled multiblock copolymers. Macromolecules 48, 1404–1411 (2015).
Gentekos, D. T., Sifri, R. J. & Fors, B. P. Controlling polymer properties through the shape of the molecular-weight distribution. Nat. Rev. Mater. 4, 761–774 (2019).
Whitfield, R. et al. Tailoring polymer dispersity and shape of molecular weight distributions: methods and applications. Chem. Sci. 10, 8724–8734 (2019).
Junkers, T. Polymers in the blender. Macromol. Chem. Phys. 221, 2000234 (2020).
Liu, D., Sponza, A. D., Yang, D. & Chiu, M. Modulating polymer dispersity with light: cationic polymerization of vinyl ethers using photochromic initiators. Angew. Chem. Int. Ed. 58, 16210–16216 (2019).
Liu, X., Wang, C. G. & Goto, A. Polymer dispersity control by organocatalyzed living radical polymerization. Angew. Chem. 131, 5654–5659 (2019).
Gentekos, D. T., Dupuis, L. N. & Fors, B. P. Beyond dispersity: deterministic control of polymer molecular weight distribution. J. Am. Chem. Soc. 138, 1848–1851 (2016).
Gentekos, D. T. et al. Exploiting molecular weight distribution shape to tune domain spacing in block copolymer thin films. J. Am. Chem. Soc. 140, 4639–4648 (2018).
Kottisch, V., Gentekos, D. T. & Fors, B. P. ‘Shaping’ the future of molecular weight distributions in anionic polymerization. ACS Macro Lett. 5, 796–800 (2016).
Nadgorny, M. et al. Manipulation of molecular weight distribution shape as a new strategy to control processing parameters. Macromol. Rapid Commun. 38, 1700352 (2017).
Wang, Z. et al. Control of dispersity and grafting density of particle brushes by variation of ATRP catalyst concentration. ACS Macro Lett. 8, 859–864 (2019).
Plichta, A., Zhong, M., Li, W., Elsen, A. M. & Matyjaszewski, K. Tuning dispersity in diblock copolymers using ARGET ATRP. Macromol. Chem. Phys. 213, 2659–2668 (2012).
Whitfield, R., Parkatzidis, K., Truong, N. P., Junkers, T. & Anastasaki, A. Tailoring polymer dispersity by raft polymerization: a versatile approach. Chem 6, 1340–1352 (2020).
Parkatzidis, K. et al. Tailoring polymer dispersity by mixing chain transfer agents in PET-RAFT polymerization. Polym. Chem. 11, 4968–4972 (2020).
Rolland, M., Truong, N. P., Whitfield, R. & Anastasaki, A. Tailoring polymer dispersity in photoinduced iron-catalyzed ATRP. ACS Macro Lett. 9, 459–463 (2020).
Whitfield, R., Parkatzidis, K., Rolland, M., Truong, N. P. & Anastasaki, A. Tuning dispersity by photoinduced atom transfer radical polymerisation: monomodal distributions with ppm copper concentration. Angew. Chem. Int. Ed. 58, 13323–13328 (2019).
Benaglia, M. et al. Polystyrene-block-poly (vinyl acetate) through the use of a switchable RAFT agent. Macromolecules 42, 9384–9386 (2009).
Benaglia, M. et al. Universal (switchable) RAFT agents. J. Am. Chem. Soc. 131, 6914–6915 (2009).
Moad, G., Keddie, D., Guerrero‐Sanchez, C., Rizzardo, E. & Tang, S.H. Advances in switchable RAFT polymerization. Macromol. Symp. 350, 34–42 (2015).
Keddie, D. J., Guerrero-Sanchez, C., Moad, G., Rizzardo, E. & Thang, S. H. Switchable reversible addition–fragmentation chain transfer (RAFT) polymerization in aqueous solution, N,N-dimethylacrylamide. Macromolecules 44, 6738–6745 (2011).
Fraenkel, G. & Franconi, C. Protonation of amides 1. J. Am. Chem. Soc. 82, 4478–4483 (1960).
Farona, M., Ayers, W., Ramsey, B. & Graselli, J. Acrylamide and N,N-dimethylacrylamide complexes. II. N-protonation and N-bonding to some transition metal perchlorates and tetrafluouroborates. Inorg. Chim. Acta 3, 503–507 (1969).
Yadav, V. et al. Dispersity control in atom transfer radical polymerizations through addition of phenylhydrazine. Polym. Chem. 9, 4332–4342 (2018).
Harrisson, S. The chain length distribution of an ideal reversible deactivation radical polymerization. Polymers 10, 887 (2018).
Gody, G., Zetterlund, P. B., Perrier, S. & Harrisson, S. The limits of precision monomer placement in chain growth polymerization. Nat. Commun. 7, 10514 (2016).
Kurov, G., Afonin, A., Svyatkina, L., Dmitrieva, L. & Pal’chuk, E. Structure and hydrolytic activity of N-vinyl derivatives of phenothiazine, carbazole and acridone. Russ. Chem. B 36, 403–405 (1987).
Harrisson, S. et al. RAFT polymerization of vinyl esters: synthesis and applications. Polymers 6, 1437–1488 (2014).
Perrier, S. B. 50th anniversary perspective: RAFT polymerization—a user guide. Macromolecules 50, 7433–7447 (2017).
Acknowledgements
A.A. acknowledges ETH Zurich (Switzerland) for financial support. N.P.T. acknowledges the award of a DECRA Fellowship from the ARC (DE180100076). We acknowledge M. Rolland for her artistic contribution in the conceptual figure. Finally, we acknowledge L. Bigler and U. Stadler (University of Zurich) for access to matrix-assisted desorption ionization time-of-flight mass spectrometry.
Author information
Authors and Affiliations
Contributions
A.A. conceived the initial idea and managed the overall project; M.-N.A., N.P.T. and A.A. designed the experiments; M.-N.A performed the vast majority of the experiments and analysed the data with input from A.A., N.P.T. and R.W.; M.-N.A. and A.A. co-wrote the manuscript with input from N.P.T. and R.W. During the revisions, S.H., T.J. and D.W. were added as co-authors. D.W. and T.J. performed the electrospray ionization–mass spectrometry measurements and the Predici simulations and S.H. conducted the calculations for the percentage of defective chains. All the authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Graeme Moad, Shigeru Yamago and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–40, Schemes 1–3, Tables 1–28. Supplementary discussion and experimental procedures.
Source data
Source Data Fig. 2
NMR and GPC source data.
Source Data Fig. 3
NMR and GPC source data.
Source Data Fig. 4
NMR and GPC source data.
Source Data Fig. 5
NMR and GPC source data.
Rights and permissions
About this article
Cite this article
Antonopoulou, MN., Whitfield, R., Truong, N.P. et al. Concurrent control over sequence and dispersity in multiblock copolymers. Nat. Chem. 14, 304–312 (2022). https://doi.org/10.1038/s41557-021-00818-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00818-8
This article is cited by
-
An acidic route for radical polymerizations
Nature Synthesis (2024)
-
Acid-triggered radical polymerization of vinyl monomers
Nature Synthesis (2024)
-
Organoboron chemistry towards controlled and precise polymer synthesis
Science China Chemistry (2023)
-
Dispersity Regulation in Photo-controlled Radical Polymerization by Merging Aryl Sulfonyl Chloride Initiators and Mixed Disulfide Agents
Chemical Research in Chinese Universities (2023)