Abstract
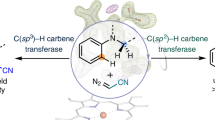
Chiral amines can be made by insertion of a carbene into an N–H bond using two-catalyst systems that combine a transition metal-based carbene-transfer catalyst and a chiral proton-transfer catalyst to enforce stereocontrol. Haem proteins can effect carbene N–H insertion, but asymmetric protonation in an active site replete with proton sources is challenging. Here we describe engineered cytochrome P450 enzymes that catalyse carbene N–H insertion to prepare biologically relevant α-amino lactones with high activity and enantioselectivity (up to 32,100 total turnovers, >99% yield and 98% e.e.). These enzymes serve as dual-function catalysts, inducing carbene transfer and promoting the subsequent proton transfer with excellent stereoselectivity in a single active site. Computational studies uncover the detailed mechanism of this new-to-nature enzymatic reaction and explain how active-site residues accelerate this transformation and provide stereocontrol.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data necessary to support the paper’s conclusions are available in the main text and the Supplementary Information. X-ray crystal structures of 3e (CCDC 2065484) and 3l (CCDC 2065489) are available free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. Plasmids encoding the enzymes reported in this study are available for research purposes from F.H.A. under a material transfer agreement with the California Institute of Technology. Source data are provided with this paper.
References
Hili, R. & Yudin, A. K. Making carbon–nitrogen bonds in biological and chemical synthesis. Nat. Chem. Biol. 2, 284–287 (2006).
Froidevaux, V., Negrell, C., Caillol, S., Pascault, J.-P. & Boutevin, B. Biobased amines: from synthesis to polymers; present and future. Chem. Rev. 116, 14181–14224 (2016).
Bariwal, J. & Van der Eycken, E. C–N bond forming cross-coupling reactions: an overview. Chem. Soc. Rev. 42, 9283–9303 (2013).
Kim, J. E., Choi, S., Balamurugan, M., Jang, J. H. & Nam, K. T. Electrochemical C–N bond formation for sustainable amine synthesis. Trends Chem. 2, 1004–1019 (2020).
Kohls, H., Steffen-Munsberg, F. & Höhne, M. Recent achievements in developing the biocatalytic toolbox for chiral amine synthesis. Curr. Opin. Chem. Biol. 19, 180–192 (2014).
Hauer, B. Embracing nature’s catalysts: a viewpoint on the future of biocatalysis. ACS Catal. 10, 8418–8427 (2020).
Wu, S., Snajdrova, R., Moore, J. C., Baldenius, K. & Bornscheuer, U. T. Biocatalysis: enzymatic synthesis for industrial applications. Angew. Chem. Int. Ed. 60, 88–119 (2021).
Doyle, M. P. Catalytic methods for metal carbene transformations. Chem. Rev. 86, 919–939 (1986).
Liu, B., Zhu, S.-F., Zhang, W., Chen, C. & Zhou, Q.-L. Highly enantioselective insertion of carbenoids into N−H bonds catalyzed by copper complexes of chiral spiro bisoxazolines. J. Am. Chem. Soc. 129, 5834–5835 (2007).
Lee, E. C. & Fu, G. C. Copper-catalyzed asymmetric N−H insertion reactions: couplings of diazo compounds with carbamates to generate α-amino acids. J. Am. Chem. Soc. 129, 12066–12067 (2007).
Hou, Z. et al. Highly enantioselective insertion of carbenoids into N–H bonds catalyzed by copper(I) complexes of binol derivatives. Angew. Chem. Int. Ed. 49, 4763–4766 (2010).
Arredondo, V., Hiew, S. C., Gutman, E. S., Premachandra, I. D. U. A. & Van Vranken, D. L. Enantioselective palladium-catalyzed carbene insertion into the N−H bonds of aromatic heterocycles. Angew. Chem. Int. Ed. 56, 4156–4159 (2017).
Wang, Z. J., Peck, N. E., Renata, H. & Arnold, F. H. Cytochrome P450-catalyzed insertion of carbenoids into N–H bonds. Chem. Sci. 5, 598–601 (2014).
Sreenilayam, G. & Fasan, R. Myoglobin-catalyzed intermolecular carbene N–H insertion with arylamine substrates. Chem. Commun. 51, 1532–1534 (2015).
Steck, V., Carminati, D. M., Johnson, N. R. & Fasan, R. Enantioselective synthesis of chiral amines via biocatalytic carbene N–H insertion. ACS Catal. 10, 10967–10977 (2020).
Steck, V., Sreenilayam, G. & Fasan, R. Selective functionalization of aliphatic amines via myoglobin-catalyzed carbene N–H insertion. Synlett 31, 224–229 (2020).
Coelho, P. S., Brustad, E. M., Kannan, A. & Arnold, F. H. Olefin cyclopropanation via carbene transfer catalyzed by engineered cytochrome P450 enzymes. Science 339, 307–310 (2013).
Zhang, R. K. et al. Enzymatic assembly of carbon–carbon bonds via iron-catalysed sp3 C–H functionalization. Nature 565, 67–72 (2019).
Liu, Z. & Arnold, F. H. New-to-nature chemistry from old protein machinery: carbene and nitrene transferases. Curr. Opin. Biotechnol. 69, 43–51 (2021).
Chen, K. & Arnold, F. H. Engineering new catalytic activities in enzymes. Nat. Catal. 3, 203–213 (2020).
Ren, Y.-Y., Zhu, S.-F. & Zhou, Q.-L. Chiral proton-transfer shuttle catalysts for carbene insertion reactions. Org. Biomol. Chem. 16, 3087–3094 (2018).
Li, M.-L., Yu, J.-H., Li, Y.-H., Zhu, S.-F. & Zhou, Q.-L. Highly enantioselective carbene insertion into N–H bonds of aliphatic amines. Science 366, 990–994 (2019).
Sharon, D. A., Mallick, D., Wang, B. & Shaik, S. Computation sheds insight into iron porphyrin carbenes’ electronic structure, formation, and N–H insertion reactivity. J. Am. Chem. Soc. 138, 9597–9610 (2016).
Pavlović, D. et al. Synthesis and structure–activity relationships of α-amino-γ-lactone ketolides: a novel class of macrolide antibiotics. ACS Med. Chem. Lett. 5, 1133–1137 (2014).
DeAngelis, A., Dmitrenko, O. & Fox, J. M. Rh-catalyzed intermolecular reactions of cyclic α-diazocarbonyl compounds with selectivity over tertiary C–H bond migration. J. Am. Chem. Soc. 134, 11035–11043 (2012).
Sattely, E. S., Meek, S. J., Malcolmson, S. J., Schrock, R. R. & Hoveyda, A. H. Design and stereoselective preparation of a new class of chiral olefin metathesis catalysts and application to enantioselective synthesis of quebrachamine: catalyst development inspired by natural product synthesis. J. Am. Chem. Soc. 131, 943–953 (2009).
Chen, K., Zhang, S.-Q., Brandenberg, O. F., Hong, X. & Arnold, F. H. Alternate heme ligation steers activity and selectivity in engineered cytochrome P450-catalyzed carbene-transfer reactions. J. Am. Chem. Soc. 140, 16402–16407 (2018).
Zhou, A. Z., Chen, K. & Arnold, F. H. Enzymatic lactone-carbene C–H insertion to build contiguous chiral centers. ACS Catal. 10, 5393–5398 (2020).
Chen, K. et al. Engineered cytochrome c-catalyzed lactone-carbene B–H insertion. Synlett 30, 378–382 (2019).
Coelho, P. S. et al. A serine-substituted P450 catalyzes highly efficient carbene transfer to olefins in vivo. Nat. Chem. Biol. 9, 485–487 (2013).
Brandenberg, O. F., Chen, K. & Arnold, F. H. Directed evolution of a cytochrome P450 carbene transferase for selective functionalization of cyclic compounds. J. Am. Chem. Soc. 141, 8989–8995 (2019).
Garcia-Borràs, M. et al. Origin and control of chemoselectivity in cytochrome c catalyzed carbene transfer into Si–H and N–H bonds. J. Am. Chem. Soc. 143, 7114–7123 (2021).
Khade, R. L. & Zhang, Y. Catalytic and biocatalytic iron porphyrin carbene formation: effects of binding mode, carbene substituent, porphyrin substituent, and protein axial ligand. J. Am. Chem. Soc. 137, 7560–7563 (2015).
Kalita, S., Shaik, S., Kisan, H. K. & Dubey, K. D. A paradigm shift in the catalytic cycle of P450: the preparatory choreography during O2 binding and origins of the necessity for two protonation pathways. ACS Catal. 10, 11481–11492 (2020).
Fisher, D. J. & Hayes, A. L. Mode of action of the systemic fungicides furalaxyl, metalaxyl and ofurace. Pestic. Sci. 13, 330–339 (1982).
Kunz, W. & Kristinsson, H. Bildung von 4-, 5- und 6gliedrigen heterocyclen durch ambidoselektive ringschlüsse von enolat-ionen. Helv. Chim. Acta 62, 872–881 (1979).
Acknowledgements
This work was supported by the National Science Foundation Division of Molecular and Cellular Biosciences (grant 2016137 to F.H.A.), the US Army Research Office Institute for Collaborative Biotechnologies (cooperative agreement W911NF-19-2-0026 to F.H.A.), the Spanish Ministry of Science and Innovation MICINN (grant PID2019-111300GA-I00 to M.G.-B.) and the Generalitat de Catalunya AGAUR Beatriu de Pinós H2020 MSCA-Cofund (2018-BP-00204 project to M.G.-B.). K.C. thanks the Resnick Sustainability Institute at Caltech for fellowship support. The computer resources at MinoTauro and the Barcelona Supercomputing Center BSC-RES are acknowledged (RES-QSB-2020-2-0016). We thank D. C. Miller, S. Brinkmann-Chen, R. Lal and T. Zeng for helpful discussions and comments on the manuscript. We further thank M. Shahgholi for high-resolution mass spectrometry analysis and M. K. Takase for X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
Z.L. and K.C. conceived and designed the overall project with F.H.A. providing guidance. Z.L. and A.Z.Z. designed and performed the initial screening of haem proteins and the substrate scope study. C.C.-T. and M.G.-B. carried out the computational studies. Z.L., K.C., M.G.-B. and F.H.A. wrote the manuscript with the input of all authors.
Corresponding authors
Ethics declarations
Competing interests
K.C., Z.L. and A.Z.Z. are inventors on a US Patent Application (invention title, Diverse Carbene Transferase Enzyme Catalysts Derived from a P450 Enzyme; application no., 17/200,394) filed by the California Institute of Technology, which covers lactone-carbene N–H insertion with engineered P450 enzymes. The patent was filed on 12 March 2021. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Sabine Flitsch, Sason Shaik and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–21, Tables 1–17, experimental data, procedural details, synthesis and characterization data, NMR spectra, X-ray crystallographic data and computational modelling data.
Supplementary Data 1
crystallographic data of compound 3e; CCDC 2065484.
Supplementary Data 2
crystallographic data of compound 3l; CCDC 2065489.
Source data
Source Data Fig. 2
Statistical source data of 40 previously reported variants.
Source Data Fig. 4
Statistical source data of substrate scope.
Source Data Fig. 5
Statistical source data of OD test.
Rights and permissions
About this article
Cite this article
Liu, Z., Calvó-Tusell, C., Zhou, A.Z. et al. Dual-function enzyme catalysis for enantioselective carbon–nitrogen bond formation. Nat. Chem. 13, 1166–1172 (2021). https://doi.org/10.1038/s41557-021-00794-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00794-z
This article is cited by
-
Construction of Au/Cu hierarchically organized particles with dual-functional enzyme-like activity
Science China Materials (2023)
-
Chemoselective carbene insertion into the N−H bonds of NH3·H2O
Nature Communications (2022)