Abstract
Although the multiphase chemistry of SO2 in aerosol particles is of great importance to air quality under polluted haze conditions, a fundamental understanding of the pertinent mechanisms and kinetics is lacking. In particular, there is considerable debate on the importance of NO2 in the oxidation of SO2 in aerosol particles. Here experiments with atmospherically relevant deliquesced particles at buffered pH values of 4–5 show that the effective rate constant for the reaction of NO2 with SO32− ((1.4 ± 0.5) × 1010 M−1 s−1) is more than three orders of magnitude larger than the value in dilute solutions. An interfacial reaction at the surface of aerosol particles probably drives the very fast kinetics. Our results indicate that oxidation of SO2 by NO2 at aerosol surfaces may be an important source of sulfate aerosols under polluted haze conditions.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data are available in the main paper and the Supplementary Information files. Source data files are provided for Figs. 1, 2 and 4 and for Extended Data Figs. 1–6. Source data are provided with this paper.
References
Su, H., Cheng, Y. & Pöschl, U. New multiphase chemical processes influencing atmospheric aerosols, air quality, and climate in the Anthropocene. Acc. Chem. Res. 53, 2034–2043 (2020).
Wang, G. et al. Persistent sulfate formation from London fog to Chinese haze. Proc. Natl Acad. Sci. USA 113, 13630–13635 (2016).
Wang, Y. et al. Enhanced sulfate formation during China’s severe winter haze episode in January 2013 missing from current models. J. Geophys. Res. Atmos. https://doi.org/10.1002/2013JD021426 (2014).
Cheng, Y. et al. Reactive nitrogen chemistry in aerosol water as a source of sulfate during haze events in China. Sci. Adv. https://doi.org/10.1126/sciadv.1601530 (2016).
Huang, R.-J. et al. High secondary aerosol contribution to particulate pollution during haze events in China. Nature 514, 218–222 (2014).
Guo, S. et al. Elucidating severe urban haze formation in China. Proc. Natl Acad. Sci. USA 111, 17373–17378 (2014).
Zhang, R. et al. Formation of urban fine particulate matter. Chem. Rev. 115, 3803–3855 (2015).
Zheng, B. et al. Heterogeneous chemistry: a mechanism missing in current models to explain secondary inorganic aerosol formation during the January 2013 haze episode in North China. Atmos. Chem. Phys. 15, 2031–2049 (2015).
Ye, C. et al. High H2O2 concentrations observed during haze periods during the winter in Beijing: importance of H2O2 oxidation in sulfate formation. Environ. Sci. Technol. Lett. 5, 757–763 (2018).
Hung, H.-M., Hsu, M.-N. & Hoffmann, M. R. Quantification of SO2 oxidation on interfacial surfaces of acidic micro-droplets: implication for ambient sulfate formation. Environ. Sci. Technol. 52, 9079–9086 (2018).
Liu, T., Clegg, S. L. & Abbatt, J. P. D. Fast oxidation of sulfur dioxide by hydrogen peroxide in deliquesced aerosol particles. Proc. Natl Acad. Sci. USA 117, 1354–1359 (2020).
Li, L., Hoffmann, M. R. & Colussi, A. J. Role of nitrogen dioxide in the production of sulfate during Chinese haze-aerosol episodes. Environ. Sci. Technol. 52, 2686–2693 (2018).
Gen, M., Zhang, R., Huang, D. D., Li, Y. & Chan, C. K. Heterogeneous oxidation of SO2 in sulfate production during nitrate photolysis at 300 nm: effect of pH, relative humidity, irradiation intensity, and the presence of organic compounds. Environ. Sci. Technol. 53, 8757–8766 (2019).
Wang, X. et al. Atmospheric photosensitization: a new pathway for sulfate formation. Environ. Sci. Technol. 54, 3114–3120 (2020).
Zhang, F. et al. An unexpected catalyst dominates formation and radiative forcing of regional haze. Proc. Natl Acad. Sci. USA 117, 3960–3966 (2020).
Ma, T. et al. Contribution of hydroxymethanesulfonate (HMS) to severe winter haze in the North China Plain. Atmos. Chem. Phys. 20, 5887–5897 (2020).
Li, J. et al. Stable sulfur isotopes revealed a major role of transition-metal ion-catalyzed SO2 oxidation in haze episodes. Environ. Sci. Technol. 54, 2626–2634 (2020).
Ge, S. et al. Abundant NH3 in China enhances atmospheric HONO production by promoting the heterogeneous reaction of SO2 with NO2. Environ. Sci. Technol. 53, 14339–14347 (2019).
Song, S. et al. Fine-particle pH for Beijing winter haze as inferred from different thermodynamic equilibrium models. Atmos. Chem. Phys. 18, 7423–7438 (2018).
Guo, H., Weber, R. J. & Nenes, A. High levels of ammonia do not raise fine particle pH sufficiently to yield nitrogen oxide-dominated sulfate production. Sci Rep. 7, 12109 (2017).
Liu, M. et al. Fine particle pH during severe haze episodes in northern China. Geophys. Res. Lett. 44, 5213–5221 (2017).
Lee, Y. & Schwartz, S. E. in Precipitation Scavenging, Dry Deposition and Resuspension (eds Pruppacher, H. R., Semonin, R. G. and Slinn, W. G. N.) Vol. 1, 453–470 (Elsevier, 1983).
Clifton, C. L., Altstein, N. & Huie, R. E. Rate constant for the reaction of nitrogen dioxide with sulfur(IV) over the pH range 5.3–13. Environ. Sci. Technol. 22, 586–589 (1988).
Bzdek, B. R., Reid, J. P. & Cotterell, M. I. Open questions on the physical properties of aerosols. Commun. Chem. 3, 105 (2020).
Chen, T. et al. Enhancement of aqueous sulfate formation by the coexistence of NO2/NH3 under high ionic strengths in aerosol water. Environ. Pollut. 252, 236–244 (2019).
Wang, G. et al. Particle acidity and sulfate production during severe haze events in China cannot be reliably inferred by assuming a mixture of inorganic salts. Atmos. Chem. Phys. 18, 10123–10132 (2018).
Wang, W. et al. Sulfate formation is dominated by manganese-catalyzed oxidation of SO2 on aerosol surfaces during haze events. Nat. Commun. 12, 1993 (2021).
Finlayson-Pitts, B. J., Wingen, L. M., Sumner, A. L., Syomin, D. & Ramazan, K. A. The heterogeneous hydrolysis of NO2 in laboratory systems and in outdoor and indoor atmospheres: an integrated mechanism. Phys. Chem. Chem. Phys. 5, 223–242 (2003).
Yang, J. et al. Unraveling a new chemical mechanism of missing sulfate formation in aerosol haze: gaseous NO2 with aqueous HSO3–/SO32–. J. Am. Chem. Soc. 141, 19312–19320 (2019).
Wexler, A. S. & Clegg, S. L. Atmospheric aerosol models for systems including the ions H+, NH4+, Na+, SO42−, NO3−, Cl−, Br−, and H2O. J. Geophys. Res. Atmos. 107, ACH 14-11–ACH 14-14 (2002).
Aerosol Thermodynamics Model (E-AIM, 2021); http://www.aim.env.uea.ac.uk/aim/aim.php
Canagaratna, M. R. et al. Chemical and microphysical characterization of ambient aerosols with the aerodyne aerosol mass spectrometer. Mass Spectrom. Rev. 26, 185–222 (2007).
Drexler, C., Elias, H., Fecher, B. & Wannowius, K. J. Kinetic investigation of sulfur(IV) oxidation by peroxo compounds R–OOH in aqueous solution. Fresenius J. Anal. Chem. 340, 605–615 (1991).
Oblath, S. B., Markowitz, S. S., Novakov, T. & Chang, S. G. Kinetics of the initial reaction of nitrite ion in bisulfite solutions. J. Phys. Chem. 86, 4853–4857 (1982).
Oblath, S. B., Markowitz, S. S., Novakov, T. & Chang, S. G. Kinetics of the formation of hydroxylamine disulfonate by reaction of nitrite with sulfites. J. Phys. Chem. 85, 1017–1021 (1981).
Takeuchi, H., Ando, M. & Kizawa, N. Absorption of nitrogen oxides in aqueous sodium sulfite and bisulfite solutions. Ind. Eng. Chem. Proc. Des. Dev. 16, 303–308 (1977).
Spindler, G. et al. Wet annular denuder measurements of nitrous acid: laboratory study of the artefact reaction of NO2 with S(IV) in aqueous solution and comparison with field measurements. Atmos. Environ. 37, 2643–2662 (2003).
Shen, C. H. & Rochelle, G. T. Nitrogen dioxide absorption and sulfite oxidation in aqueous sulfite. Environ. Sci. Technol. 32, 1994–2003 (1998).
Mertes, S. & Wahner, A. Uptake of nitrogen dioxide and nitrous acid on aqueous surfaces. J. Phys. Chem. 99, 14000–14006 (1995).
Jayne, J. T., Davidovits, P., Worsnop, D. R., Zahniser, M. S. & Kolb, C. E. Uptake of sulfur dioxide(G) by aqueous surfaces as a function of pH: the effect of chemical reaction at the interface. J. Phys. Chem. 94, 6041–6048 (1990).
Yabushita, A. et al. Anion-catalyzed dissolution of NO2 on aqueous microdroplets. J. Phys. Chem. A 113, 4844–4848 (2009).
Murdachaew, G., Varner, M. E., Phillips, L. F., Finlayson-Pitts, B. J. & Gerber, R. B. Nitrogen dioxide at the air–water interface: trapping, absorption, and solvation in the bulk and at the surface. Phys. Chem. Chem. Phys. 15, 204–212 (2013).
Shao, J. et al. Heterogeneous sulfate aerosol formation mechanisms during wintertime Chinese haze events: air quality model assessment using observations of sulfate oxygen isotopes in Beijing. Atmos. Chem. Phys. 19, 6107–6123 (2019).
Wang, J. et al. Fast sulfate formation from oxidation of SO2 by NO2 and HONO observed in Beijing haze. Nat. Commun. 11, 2844 (2020).
Song, S. et al. Thermodynamic modeling suggests declines in water uptake and acidity of inorganic aerosols in Beijing winter haze events during 2014/2015–2018/2019. Environ. Sci. Technol. Lett. 6, 752–760 (2019).
Xue, J. et al. Efficient control of atmospheric sulfate production based on three formation regimes. Nat. Geosci. 12, 977–982 (2019).
Zheng, G. et al. Multiphase buffer theory explains contrasts in atmospheric aerosol acidity. Science 369, 1374–1377 (2020).
Matthew, B. M., Middlebrook, A. M. & Onasch, T. B. Collection efficiencies in an Aerodyne aerosol mass spectrometer as a function of particle phase for laboratory generated aerosols. Aerosol Sci. Technol. 42, 884–898 (2008).
Roscioli, J. R., Zahniser, M. S., Nelson, D. D., Herndon, S. C. & Kolb, C. E. New approaches to measuring sticky molecules: improvement of instrumental response times using active passivation. J. Phys. Chem. A 120, 1347–1357 (2016).
Wang, N., Jorga, S. D., Pierce, J. R., Donahue, N. M. & Pandis, S. N. Particle wall-loss correction methods in smog chamber experiments. Atmos. Meas. Tech. 11, 6577–6588 (2018).
Pathak, R. K., Stanier, C. O., Donahue, N. M. & Pandis, S. N. Ozonolysis of α-pinene at atmospherically relevant concentrations: temperature dependence of aerosol mass fractions (yields). J. Geophys. Res. Atmos. https://doi.org/10.1029/2006JD007436 (2007).
Hildebrandt, L., Donahue, N. M. & Pandis, S. N. High formation of secondary organic aerosol from the photo-oxidation of toluene. Atmos. Chem. Phys. 9, 2973–2986 (2009).
Acknowledgements
T.L. thanks the National Natural Science Foundation of China projects (92044301, 21806108), the National Key R&D Program of China (2016YFC0202000) and Dengfeng Project of Nanjing University for funding. T.L. and J.P.D.A. thank the Natural Sciences and Engineering Research Council (RPGIN-05972, BCPIR-537926) for funding, and J.G. Murphy and M. Davis for the loan of the TILDAS NH3 monitor and helpful discussions.
Author information
Authors and Affiliations
Contributions
T.L. and J.P.D.A. designed the research project. T.L. performed the research. T.L. and J.P.D.A. analysed data. T.L. and J.P.D.A. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Yafang Cheng, Jianzhen Yu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Time evolution of gas-phase and aerosol-phase species for a typical ammonium nitrate seed aerosol experiment.
Concentrations of wall-loss corrected NO3- (wNO3-) and SO42- (wSO42-) were derived by correcting SO42- concentrations using the wall-loss rate of aerosol NO3- before adding NH3 into the chamber.
Extended Data Fig. 2 Time evolution of gas-phase and aerosol-phase species for the HONO control experiment.
Concentrations of wall-loss corrected SO42- were derived by correcting SO42- concentrations using a wall-loss rate of NO3- before adding NH3 into the chamber. HONO was added as an oxidant instead of NO2 by bubbling a mixed solution of 10 ml 10 mM NaNO2 and 25 ml 50 mM H2SO4 for 15 min.
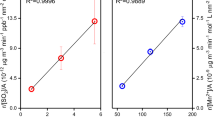
Extended Data Fig. 3 The fit of kexp vs 1/aH+ displayed in log-scale.
AN and MA represent ammonium nitrate and malonic acid experiments, respectively. The fit is based on equation (2) in the main text.
Extended Data Fig. 4 Dependence of kexp on the activity of hydrogen ion.
The grey line represents a regression fitting with equation (2) in the main text, resulting in a negligible value of \({{{\mathrm{k}}}}_{{{{\mathrm{NO}}}}_2 + {{{\mathrm{HSO}}}}_3^ - }\). The black and red dashed lines represent the results for \({{{\mathrm{k}}}}_{{{{\mathrm{NO}}}}_2 + {{{\mathrm{HSO}}}}_3^ - }\) of 3.0 × 106 M-1 s-1 and 1.5 × 107 M-1 s-1, respectively.
Extended Data Fig. 5 Time evolution of gas-phase and aerosol-phase species for control experiments without adding SO2.
a, malonic acid, b, ammonium nitrate. Concentrations of wall-loss corrected NO3- for malonic acid and ammonium nitrate experiments were derived by correcting NO3- concentrations using wall-loss rates of organics and NO3- before adding NH3 into the chamber, respectively.
Extended Data Fig. 6 Average mass-based size distributions.
Average mass-based size distributions of organics, nitrate, and sulfate for typical a malonic acid and b ammonium nitrate seed aerosol experiments. The average mass-based size distributions are shown because the time series data are quite noisy. Nevertheless, the distributions appear similar throughout the course of the experiment.
Supplementary information
Supplementary Information
Supplementary Tables 1–3.
Source data
Source Data Fig. 1
Statistical Source Data for Fig. 1
Source Data Fig. 2
Statistical Source Data for Fig. 2
Source Data Fig. 4
Statistical Source Data for Fig. 4
Source Data Extended Data Fig. 1
Statistical Source Data for Extended Data Fig. 1
Source Data Extended Data Fig. 2
Statistical Source Data for Extended Data Fig. 2
Source Data Extended Data Fig. 3
Statistical Source Data for Extended Data Fig. 3
Source Data Extended Data Fig. 4
Statistical Source Data for Extended Data Fig. 4
Source Data Extended Data Fig. 5
Statistical Source Data for Extended Data Fig. 5
Source Data Extended Data Fig. 6
Statistical Source Data for Extended Data Fig. 6
Rights and permissions
About this article
Cite this article
Liu, T., Abbatt, J.P.D. Oxidation of sulfur dioxide by nitrogen dioxide accelerated at the interface of deliquesced aerosol particles. Nat. Chem. 13, 1173–1177 (2021). https://doi.org/10.1038/s41557-021-00777-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00777-0
This article is cited by
-
Elucidating HONO formation mechanism and its essential contribution to OH during haze events
npj Climate and Atmospheric Science (2023)
-
Increasing contribution of nighttime nitrogen chemistry to wintertime haze formation in Beijing observed during COVID-19 lockdowns
Nature Geoscience (2023)
-
An interfacial role for NO2
Nature Chemistry (2021)