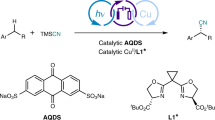
Abstract
Enantioselective catalytic processes are promoted by chiral catalysts that can execute a specific mode of catalytic reactivity, channeling the chemical reaction through a certain mechanistic pathway. Here, we show how by simply using visible light we can divert the established ionic reactivity of a chiral allyl–iridium(iii) complex to switch on completely new catalytic functions, enabling mechanistically unrelated radical-based enantioselective pathways. Photoexcitation provides the chiral organometallic intermediate with the ability to activate substrates via an electron-transfer manifold. This redox event unlocks an otherwise inaccessible cross-coupling mechanism, since the resulting iridium(ii) centre can intercept the generated radicals and undergo a reductive elimination to forge a stereogenic centre with high stereoselectivity. This photochemical strategy enables difficult-to-realize enantioselective alkyl–alkyl cross-coupling reactions between allylic alcohols and readily available radical precursors, which are not achievable under thermal activation.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data that support the findings of this study are available in the Supplementary Information (experimental procedures and characterization data). Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2021458 (4g) and 2021459 (4h). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Ojima, I. (ed.) Catalytic Asymmetric Synthesis (John Wiley & Sons, 2010).
Blaser, H. U. & Schmidt, E. (eds) Asymmetric Catalysis on Industrial Scale: Challenges, Approaches and Solutions (Wiley-VCH, 2010).
Yoon, T. P. & Jacobsen, E. N. Privileged chiral catalysts. Science 299, 1691–1693 (2003).
Walsh, P. J. & Kozlowski, M. C. (eds) Fundamental of Asymmetric Catalysis (University Science Books, 2009).
Arceo, E., Jurberg, I. D., Álvarez-Fernández, A. & Melchiorre, P. Photochemical activity of a key donor–acceptor complex can drive stereoselective catalytic α-alkylation of aldehydes. Nat. Chem. 5, 750–756 (2013).
Silvi, M., Verrier, C., Rey, Y. P., Buzzetti, L. & Melchiorre, P. Visible-light excitation of iminium ions enables the enantioselective catalytic β-alkylation of enals. Nat. Chem. 9, 868–873 (2017).
Silvi, M. & Melchiorre, P. Enhancing the potential of enantioselective organocatalysis with light. Nature 554, 41–49 (2018).
Emmanuel, M. A., Greenberg, N. R., Oblinsky, D. G. & Hyster, T. K. Accessing non-natural reactivity by irradiating nicotinamide-dependent enzymes with light. Nature 540, 414–417 (2016).
Biegasiewicz, K. F. et al. Photoexcitation of flavoenzymes enables a stereoselective radical cyclization. Science 364, 1166–1169 (2019).
Brimioulle, R. & Bach, T. Enantioselective Lewis acid catalysis of intramolecular enone [2 + 2] photocycloaddition reactions. Science 342, 840–843 (2013).
Huo, H. et al. Asymmetric photoredox transition-metal catalysis activated by visible light. Nature 515, 100–103 (2014).
Skubi, K. L. et al. Enantioselective excited-state photoreactions controlled by a chiral hydrogen-bonding iridium sensitizer. J. Am. Chem. Soc. 139, 17186–17192 (2017).
Li, Y. et al. Copper(ii)-catalyzed asymmetric photoredox reactions: enantioselective alkylation of imines driven by visible light. J. Am. Chem. Soc. 140, 15850–15858 (2018).
Chuentragool, P., Kurandina, D. & Gevorgyan, V. Catalysis with palladium complexes photoexcited by visible light. Angew. Chem. Int. Ed. 58, 11586–11598 (2019).
Torres, G. M., Liu, Y. & Arndtsen, B. A. A dual light-driven palladium catalyst: Breaking the barriers in carbonylation reactions. Science 368, 318–323 (2020).
Gandeepan, P., Koeller, J., Korvorapun, K., Mohr, J. & Ackermann, L. Visible‐light‐enabled ruthenium‐catalyzed meta‐C−H alkylation at room temperature. Angew. Chem. Int. Ed. 58, 9820–9825 (2019).
Sagadevan, A. & Greaney, M. F. meta‐Selective C−H activation of arenes at room temperature using visible light: dual‐function ruthenium catalysis. Angew. Chem. Int. Ed. 58, 9826–9830 (2019).
Kainz, Q. M. et al. Asymmetric copper-catalyzed C-N cross-couplings induced by visible light. Science 351, 681–684 (2016).
Choi, J. & Fu, G. C. Transition metal-catalyzed alkyl-alkyl bond formation: another dimension in cross-coupling chemistry. Science 356, eaaf7230 (2017).
Rössler, S. L., Petrone, D. A. & Carreira, E. M. Iridium-catalyzed asymmetric synthesis of functionally rich molecules enabled by (phosphoramidite,olefin) ligands. Acc. Chem. Res. 52, 2657–2672 (2019).
Rössler, S. L., Krautwald, S. & Carreira, E. M. Study of intermediates in iridium−(phosphoramidite,olefin)-catalyzed enantioselective allylic substitution. J. Am. Chem. Soc. 139, 3603–3606 (2017).
Defieber, C., Ariger, M. A., Moriel, P. & Carreira, E. M. Iridium-catalyzed synthesis of primary allylic amines from allylic alcohols: sulfamic acid as ammonia equivalent. Angew. Chem. Int. Ed. 46, 3139–3143 (2007).
Hartwig, J. F. & Pouy, M. J. in Iridium Catalysis (ed. Andersson, P. G.) 169–208 (Springer, 2011).
Cheng, Q. et al. Iridium-catalyzed asymmetric allylic substitution reactions. Chem. Rev. 119, 1855–1969 (2019).
Tellis, J. C., Primer, D. N. & Molander, G. A. Single-electron transmetalation in organoboron cross-coupling by photoredox/nickel dual catalysis. Science 345, 433–436 (2014).
Zuo, Z. et al. Enantioselective decarboxylative arylation of α-amino acids via the merger of photoredox and nickel catalysis. J. Am. Chem. Soc. 138, 1832–1835 (2016).
Zhang, H.-H., Zhao, J.-J. & Yu, S. Enantioselective allylic alkylation with 4-alkyl-1,4-dihydropyridines enabled by photoredox/palladium cocatalysis. J. Am. Chem. Soc. 140, 16914–16919 (2018).
Farid, S. et al. Reexamination of the Rehm–Weller data set reveals electron transfer quenching that follows a Sandros–Boltzmann dependence on free energy. J. Am. Chem. Soc. 133, 11580–11587 (2011).
Buzzetti, L., Crisenza, G. E. M. & Melchiorre, P. Mechanistic studies in photocatalysis. Angew. Chem. Int. Ed. 58, 3730–3747 (2019).
Yoshida, J., Kataoka, K., Horcajada, R. & Nagaki, A. Modern strategies in electroorganic synthesis. Chem. Rev. 108, 2265–2299 (2008).
Blouin, N. & Leclerc, M. Poly(2,7-carbazole)s: structure–property relationships. Acc. Chem. Res. 41, 1110–1119 (2008).
Głuszynska, A. Biological potential of carbazole derivatives. Eur. J. Med. Chem. 94, 405–426 (2015).
Bertrand, F. et al. α-Scission of sulfonyl radicals: a versatile process for organic synthesis. C. R. Acad. Sci. Ser. IIc 4, 547–555 (2001).
Herath, A. C. & Becker, J. Y. Kinetics of redox mediator tris(4-bromophenyl)amine in acetonitrile and ionic liquid [BMIm][PF6]: oxidation of benzyl and cyclohexyl alcohols. J. Electroanal. Chem. 619–620, 98–104 (2008).
Leifert, D. & Studer, A. The persistent radical effect in organic synthesis. Angew. Chem. Int. Ed. 59, 74–108 (2020).
Acknowledgements
This work is dedicated to the memory of Professor Kilian Muñiz. Financial support was provided by Agencia Estatal de Investigación (PID2019-106278GB-I00 and CTQ2016-75520-P), the AGAUR (Grant 2017 SGR 981) and the European Research Council (ERC-2015-CoG 681840-CATA-LUX). G.E.M.C. thanks the EU for a Horizon 2020 Marie Skłodowska-Curie Fellowship (H2020-MSCA-IF-2017, 795793). D.M. thanks H2020-MSCA-ITN-2016 (722591–PHOTOTRAIN) for a predoctoral fellowship. We thank A. Llobet and J. Holub for assistance with differential pulse voltammetry and E. E. Adán for help with X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
G.E.M.C. and P.M. conceived and supervised the project. G.E.M.C., A.F., E.G. and D.M. performed all experiments. All authors contributed to the experimental design and the interpretation of data. G.E.M.C. and P.M. directed the research and wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–24, Discussion, Tables 1 and 2, characterization of products, HPLC traces and 1H NMR and 13C NMR spectra.
Supplementary Data 1
Crystallographic data for compound 4g. CCDC 2021458.
Supplementary Data 2
Crystallographic data for compound 4h. CCDC 2021459.
Rights and permissions
About this article
Cite this article
Crisenza, G.E.M., Faraone, A., Gandolfo, E. et al. Catalytic asymmetric C–C cross-couplings enabled by photoexcitation. Nat. Chem. 13, 575–580 (2021). https://doi.org/10.1038/s41557-021-00683-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00683-5