Abstract
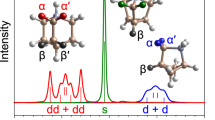
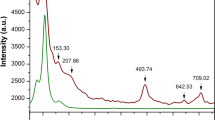
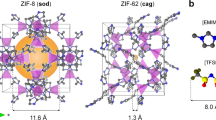
Several organic–inorganic hybrid materials from the metal–organic framework (MOF) family have been shown to form stable liquids at high temperatures. Quenching then results in the formation of melt-quenched MOF glasses that retain the three-dimensional coordination bonding of the crystalline phase. These hybrid glasses have intriguing properties and could find practical applications, yet the melt-quench phenomenon has so far remained limited to a few MOF structures. Here we turn to hybrid organic–inorganic perovskites—which occupy a prominent position within materials chemistry owing to their functional properties such as ion transport, photoconductivity, ferroelectricity and multiferroicity—and show that a series of dicyanamide-based hybrid organic–inorganic perovskites undergo melting. Our combined experimental–computational approach demonstrates that, on quenching, they form glasses that largely retain their solid-state inorganic–organic connectivity. The resulting materials show very low thermal conductivities (~0.2 W m−1 K−1), moderate electrical conductivities (10−3–10−5 S m−1) and polymer-like thermomechanical properties.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Representative input files for the molecular dynamics simulations are available online in our data repository at https://github.com/fxcoudert/citable-data. The experimental data that support the findings of this study (characterization and analytical data for both crystalline and glass materials, structural refinements, a.c. electrical conductivity experiments and thermal conductivity measurements, source data for all experimental Supplementary figures) are available as Supplementary Information and in Symplectic Elements with the identifier https://doi.org/10.17863/CAM.63032.
References
Mitzi, D. B. Introduction: Perovskites. Chem. Rev. 119, 3033–3035 (2019).
Li, W. et al. Chemically diverse and multifunctional hybrid organic–inorganic perovskites. Nat. Rev. Mater. 2, 16099 (2017).
Li, W., Stroppa, A., Wang, Z.-M. & Gao, S. Hybrid Organic-Inorganic Perovskites (Wiley, 2020).
Burschka, J. et al. Sequential deposition as a route to high-performance perovskite-sensitized solar cells. Nature 499, 316–319 (2013).
Lee, M. M. et al. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 338, 643–647 (2012).
Jain, P. et al. Multiferroic behavior associated with an order–disorder hydrogen bonding transition in metal–organic frameworks (MOFs) with the perovskite ABX3 architecture. J. Am. Chem. Soc. 131, 13625–13627 (2009).
Wu, Y. et al. [Am]Mn(H2POO)3: a new family of hybrid perovskites based on the hypophosphite ligand. J. Am. Chem. Soc. 139, 16999–17002 (2017).
Bermudez-Garcia, J. M. et al. Role of temperature and pressure on the multisensitive multiferroic dicyanamide framework [TPrA][Mn(dca)3] with perovskite-like structure. Inorg. Chem. 54, 11680–11687 (2015).
Saparov, B. & Mitzi, D. B. Organic–inorganic perovskites: structural versatility for functional materials design. Chem. Rev. 116, 4558–4596 (2016).
Egger, D. A., Rappe, A. M. & Kronik, L. Hybrid organic–inorganic perovskites on the move. Acc. Chem. Res. 49, 573–581 (2016).
Wang, Y. H. et al. Pressure-induced phase transformation, reversible amorphization, and anomalous visible light response in organolead bromide perovskite. J. Am. Chem. Soc. 137, 11144–11149 (2015).
Ou, T. J. et al. Visible light response, electrical transport, and amorphization in compressed organolead iodine perovskites. Nanoscale 8, 11426–11431 (2016).
Mitzi, D. B. Synthesis, crystal structure, and optical and thermal properties of (C4H9NH3)2MI4 (M=Ge, Sn, Pb). Chem. Mater. 8, 791–800 (1996).
Gaillac, R. et al. Liquid metal–organic frameworks. Nat. Mater. 16, 1149–1154 (2017).
Bermudez-Garcia, J. M. et al. Multiple phase and dielectric transitions on a novel multi-sensitive [TPrA][M(dca)3] (M: Fe2+, Co2+ and Ni2+) hybrid inorganic–organic perovskite family. J. Mater. Chem. C 4, 4889–4898 (2016).
Umeyama, D. et al. Reversible solid-to-liquid phase transition of coordination polymer crystals. J. Am. Chem. Soc. 137, 864–870 (2015).
Bermudez-Garcia, J. M. et al. A simple in situ synthesis of magnetic M@CNTs by thermolysis of the hybrid perovskite [TPrA][M(dca)3]. New J. Chem. 41, 3124–3133 (2017).
Qiao, A. et al. A metal–organic framework with ultrahigh glass-forming ability. Sci. Adv. 4, eaao6827 (2018).
Pell, A. J., Pintacuda, G. & Grey, C. P. Paramagnetic NMR in solution and the solid state. Prog. Nucl. Magn. Reson. Spectrosc. 111, 1–271 (2019).
Ishii, Y., Wickramasinghe, N. P. & Chimon, S. A new approach in 1D and 2D 13C high-resolution solid-state NMR spectroscopy of paramagnetic organometallic complexes by very fast magic-angle spinning. J. Am. Chem. Soc. 125, 3438–3439 (2003).
Kervern, G., Pintacuda, G. & Emsley, L. Fast adiabatic pulses for solid-state NMR of paramagnetic systems. Chem. Phys. Lett. 435, 157–162 (2007).
Gougeon, R. et al. High-resolution solid-state nuclear magnetic resonance study of the tetrapropylammonium template in a purely siliceous MFI-type zeolite. Magn. Reson. Chem. 36, 415–421 (1998).
Hing, A. W., Vega, S. & Schaefer, J. Transferred-echo double-resonance NMR. J. Magn. Reson. 96, 205–209 (1992).
Tucker, M. C. et al. Hyperfine fields at the Li site in LiFePO4-type olivine materials for lithium rechargeable batteries: a 7Li MAS NMR and SQUID study. J. Am. Chem. Soc. 124, 3832–3833 (2002).
Tauber, K., Dani, A. & Yuan, J. Y. Covalent cross-linking of porous poly(ionic liquid) membrane via a triazine network. Acs Macro. Lett. 6, 1–5 (2017).
Kroke, E. et al. Tri-s-triazine derivatives. Part I. From trichloro-tri-s-triazine to graphitic C3N4 structures. New J. Chem. 26, 508–512 (2002).
Silverstein, R. M., Webster, F. X., Kiemle, D. J. & Bryce, D. L. Spectrometric Identification of Organic Compounds 8th edn (Wiley, 2014).
Orgel, L. E. Spectra of transition-metal complexes. J. Chem. Phys. 23, 1004–1014 (1955).
Bo, S. H. et al. Thin-film and bulk investigations of LiCoBO3 as a Li-ion battery cathode. ACS Appl. Mater. Interfaces 6, 10840–10848 (2014).
Schlueter, J. A., Manson, J. L. & Geiser, U. Structural and magnetic diversity in tetraalkylammonium salts of anionic M[N(CN)2]3- (M = Mn and Ni) three-dimensional coordination polymers. Inorg. Chem. 44, 3194–3202 (2005).
Chakravarty, C., Debenedetti, P. G. & Stillinger, F. H. Lindemann measures for the solid-liquid phase transition. J. Chem. Phys. 126, 204508 (2007).
Leo, C. J., Rao, G. V. S. & Chowdari, B. V. R. Fast ion conduction in the Li-analogues of Nasicon, Li1 + x [(Ta1 – x Gex)Al](PO4)3. J. Mater. Chem. 12, 1848–1853 (2002).
Chowdari, B. V. R., Yoo, H.-L., Choi, G. M. & Lee, J.-H. Solid State Ionics: The Science and Technology of Ions in Motion (World Scientific, 2004).
Snyder, G. J. & Toberer, E. S. Complex thermoelectric materials. Nat. Mater. 7, 105–114 (2008).
Bell, L. E. Cooling, heating, generating power, and recovering waste heat with thermoelectric systems. Science 321, 1457–1461 (2008).
Pisoni, A. et al. Ultra-low thermal conductivity in organic–inorganic hybrid perovskite CH3NH3PbI3. J. Phys. Chem. Lett. 5, 2488–2492 (2014).
Ye, T. et al. Ultra-high Seebeck coefficient and low thermal conductivity of a centimeter-sized perovskite single crystal acquired by a modified fast growth method. J. Mater. Chem. C 5, 1255–1260 (2017).
Gunatilleke, W. D. C. B. et al. Thermal conductivity of a perovskite-type metal–organic framework crystal. Dalton Trans. 46, 13342–13344 (2017).
Huang, B. L. et al. Thermal conductivity of a metal-organic framework (MOF-5): Part II. Measurement. Int. J. Heat Mass Transfer 50, 405–411 (2007).
Cui, B. Y. et al. Thermal conductivity of ZIF-8 thin-film under ambient gas pressure. ACS Appl. Mater. Interfaces 9, 28139–28143 (2017).
Bansal, N. P. & Doremus, R. H. Thermal Conductivity (Elsevier, 1986).
Debenedetti, P. G. & Stillinger, F. H. Supercooled liquids and the glass transition. Nature 410, 259–267 (2001).
Angell, C. A. Formation of glasses from liquids and biopolymers. Science 267, 1924–1935 (1995).
Bennett, T. D. et al. Melt-quenched glasses of metal–organic frameworks. J. Am. Chem. Soc. 138, 3484–3492 (2016).
Bansal, N. P. & Doremus, R. H. Electrical Conductivity (Elsevier, 1986).
Wu, Y. et al. Hypophosphite hybrid perovskites: a platform for unconventional tilts and shifts. Chem. Commun. 54, 3751–3754 (2018).
Gaultois, M. W. et al. Data-driven review of thermoelectric materials: performance and resource considerations. Chem. Mater. 25, 2911–2920 (2013).
Russ, B. et al. Organic thermoelectric materials for energy harvesting and temperature control. Nat. Rev. Mater. 1, 16050 (2016).
Menard, K. P. & Menard, N. R. in Encyclopedia of Polymer Science and Technology 1–33 (Wiley, 2015).
CES EduPack 2019 Software (Granta Design, 2019).
Bermudez-Garcia, J. M. et al. Giant barocaloric tunability in [(CH3CH2CH2)4N]Cd[N(CN)2]3 hybrid perovskite. J. Mater. Chem. C 6, 9867–9874 (2018).
Bermudez-Garcia, J. M. et al. Giant barocaloric effect in the ferroic organic-inorganic hybrid [TPrA][Mn(dca)3] perovskite under easily accessible pressures. Nat. Commun. 8, 15715 (2017).
Frentzel-Beyme, L. et al. Porous purple glass – a cobalt imidazolate glass with accessible porosity from a meltable cobalt imidazolate framework. J. Mat. Chem. A 7, 985–990 (2019).
Li, G., Lee-Sullivan, P. & Thring, R. W. Determination of activation energy for glass transition of an epoxy adhesive using dynamic mechanical analysis. J. Therm. Anal. Calorim. 60, 377–390 (2000).
Thurber, K. R. & Tycko, R. Measurement of sample temperatures under magic-angle spinning from the chemical shift and spin-lattice relaxation rate of 79Br in KBr powder. J. Magn. Reson. 196, 84–87 (2009).
Morcombe, C. R. & Zilm, K. W. Chemical shift referencing in MAS solid state NMR. J. Magn. Reson. 162, 479–486 (2003).
Fuller, M. P. & Griffiths, P. R. Diffuse reflectance measurements by infrared Fourier transform spectrometry. Anal. Chem. 50, 1906–1910 (1978).
Bain, G. A. & Berry, J. F. Diamagnetic corrections and Pascal’s constants. J. Chem. Educ. 85, 532–536 (2008).
Soper, A. K. GudrunN and GudrunX: Programs for Correcting Raw Neutron and X-ray Diffraction Data to Differential Scattering Cross Section. Technical Report RAL-TR-2011-013 (2011).
Soper, A. K. & Barney, E. R. Extracting the pair distribution function from white-beam X-ray total scattering data. J. Appl. Crystallogr. 44, 714–726 (2011).
VandeVondele, J. et al. QUICKSTEP: fast and accurate density functional calculations using a mixed Gaussian and plane waves approach. Comput. Phys. Commun. 167, 103–128 (2005).
To, T. et al. Fracture toughness of a metal–organic framework glass. Nat. Commun. 11, 2593 (2020).
Acknowledgements
B.K.S. thanks the Royal Society and the Science and Engineering Research Board of India (SERB) for their combined support in the form of a Newton International Fellowship (NIF\R1\180163). T.D.B. thanks the Royal Society for a University Research Fellowship (UF150021) and a research grant (RG94426), and the University of Canterbury Te Whare Wānanga o Waitaha, New Zealand for a University of Cambridge Visiting Canterbury Fellowship. T.D.B. and L.N.M. also thank the Leverhulme Trust for a Philip Leverhulme Prize. The EPSRC is acknowledged for a Doctoral Training Studentship to A.R.H., a PhD studentship award to A.F.S. under the industrial CASE scheme along with Johnson Matthey PLC (JM11106), and a PhD studentship award to A.P. via the National Productivity Investment Fund (EP/R51231X/1) and grant EP/K039687/1. M.D. and F.-X.C. acknowledge financial support from the Agence Nationale de la Recherche under the project “MATAREB” (ANR-18-CE29-0009-01) and access to high-performance computing platforms provided by GENCI grant A0070807069. A.D. acknowledges DST-INSPIRE (IF160050), Government of India, for awarding his fellowship. S.K.S. gratefully thanks DST, Government of India, for the infrastructural facilities. B.K.S. acknowledges R. Cornell (University of Cambridge) for his help with performing the dynamic-mechanical measurements. F.B. thanks K. J. Sanders (McMaster University) for assistance with the SHAPs pulses and K. Luzyanin (University of Liverpool) for collecting the liquid-state NMR data. J.M.B.-G. acknowledges Xunta de Galicia for a postdoctoral fellowship. X.M. is grateful for support from the Royal Society. M.F.T. would like to thank Corning Incorporated for project funding. We acknowledge the assistance of L. Longley (University of Cambridge) with calorimetric data analysis. The authors gratefully acknowledge the provision of synchrotron access to Beamline I15-1 (EE20038) at the Diamond Light Source, Rutherford Appleton Laboratory, UK.
Author information
Authors and Affiliations
Contributions
B.K.S. and T.D.B. designed the project. A.R.H., A.P. and F.B. performed all NMR experiments and analysed the data. S.M. collected the HRMS data and analysed them with F.B. The electrical conductivity measurements were performed by B.K.S. and A.D. The electrical conductivity data were analysed by B.K.S. and S.K.S. The X-ray total scattering data were collected by T.D.B., A.F.S., M.F.T., L.N.M., D.A.K., D.S.K. and P.A.C. The total scattering data were analysed by B.K.S., D.A.K., A.F.S. and T.D.B. Interpretation and analysis of calorimetric data were aided by J.M.B.-G. and X.M. Molecular simulations were performed by M.D. and F.-X.C. who also analysed the data. B.K.S. collected and analysed all other data. All authors participated in manuscript writing, led by T.D.B. and B.K.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Maria Senaris-Rodriguez and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary text, Figs. 1–60 and Tables 1–4.
Rights and permissions
About this article
Cite this article
Shaw, B.K., Hughes, A.R., Ducamp, M. et al. Melting of hybrid organic–inorganic perovskites. Nat. Chem. 13, 778–785 (2021). https://doi.org/10.1038/s41557-021-00681-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00681-7
This article is cited by
-
Desolvation of metal complexes to construct metal–organic framework glasses
Nature Synthesis (2023)
-
Nested order-disorder framework containing a crystalline matrix with self-filled amorphous-like innards
Nature Communications (2022)
-
Construction and nanotribological study of a glassy covalent organic network on surface
Nano Research (2022)
-
A glass act
Nature Chemistry (2021)