Abstract
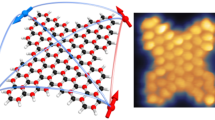
Nanographenes with zigzag edges are predicted to manifest non-trivial π-magnetism resulting from the interplay of concurrent electronic effects, such as hybridization of localized frontier states and Coulomb repulsion between valence electrons. This provides a chemically tunable platform to explore quantum magnetism at the nanoscale and opens avenues towards organic spintronics. The magnetic stability in nanographenes is thus far greatly limited by the weak magnetic exchange coupling, which remains below the room-temperature thermal energy. Here, we report the synthesis of large rhombus-shaped nanographenes with zigzag peripheries on gold and copper surfaces. Single-molecule scanning probe measurements show an emergent magnetic spin singlet ground state with increasing nanographene size. The magnetic exchange coupling in the largest nanographene (C70H22, containing five benzenoid rings along each edge), determined by inelastic electron tunnelling spectroscopy, exceeds 100 meV or 1,160 K, which outclasses most inorganic nanomaterials and survives on a metal electrode.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Additional STM/STS data and theoretical calculations, materials and methods, solution synthetic procedures and characterization data of chemical compounds (X-ray diffraction, NMR spectroscopy and high-resolution mass spectrometry) are available in the Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 1978171 (3) and 1978172 (4). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. Source data are provided with this paper.
Code availability
The tight-binding calculations were performed using a custom-made Python program available on the GitHub repository (https://github.com/eimrek/tb-mean-field-hubbard).
References
Yazyev, O. V. Emergence of magnetism in graphene materials and nanostructures. Rep. Prog. Phys. 73, 056501 (2010).
Liu, S. & Langenaeker, W. Hund’s multiplicity rule: a unified interpretation. Theor. Chem. Acc. 110, 338–344 (2003).
Gryn’ova, G., Coote, M. L. & Corminboeuf, C. Theory and practice of uncommon molecular electronic configurations. Wiley Interdiscip. Rev. Comput. Mol. Sci. 5, 440–459 (2015).
Yeh, C.-N. & Chai, J.-D. Role of Kekulé and non-Kekulé structures in the radical character of alternant polycyclic aromatic hydrocarbons: a TAO-DFT study. Sci. Rep. 6, 30562 (2016).
Das, A., Müller, T., Plasser, F. & Lischka, H. Polyradical character of triangular non-Kekulé structures, zethrenes, p-quinodimethane-linked bisphenalenyl, and the Clar goblet in comparison: an extended multireference study. J. Phys. Chem. A 120, 1625–1636 (2016).
Nakada, K., Fujita, M., Dresselhaus, G. & Dresselhaus, M. S. Edge state in graphene ribbons: nanometer size effect and edge shape dependence. Phys. Rev. B 54, 17954–17961 (1996).
Fernández-Rossier, J. & Palacios, J. J. Magnetism in graphene nanoislands. Phys. Rev. Lett. 99, 177204 (2007).
Ezawa, M. Metallic graphene nanodisks: electronic and magnetic properties. Phys. Rev. B 76, 245415 (2007).
Konishi, A. et al. Synthesis and characterization of teranthene: a singlet biradical polycyclic aromatic hydrocarbon having Kekulé structures. J. Am. Chem. Soc. 132, 11021–11023 (2010).
Konishi, A. et al. Synthesis and characterization of quarteranthene: elucidating the characteristics of the edge state of graphene nanoribbons at the molecular level. J. Am. Chem. Soc. 135, 1430–1437 (2013).
Wang, S. et al. Giant edge state splitting at atomically precise graphene zigzag edges. Nat. Commun. 7, 11507 (2016).
Huang, R. et al. Higher order π-conjugated polycyclic hydrocarbons with open-shell singlet ground state: nonazethrene versus nonacene. J. Am. Chem. Soc. 138, 10323–10330 (2016).
Zeng, W. et al. Superoctazethrene: an open-shell graphene-like molecule possessing large diradical character but still with reasonable stability. J. Am. Chem. Soc. 140, 14054–14058 (2018).
Mishra, S. et al. On-surface synthesis of super-heptazethrene. Chem. Commun. 56, 7467–7470 (2020).
Li, J. et al. Single spin localization and manipulation in graphene open-shell nanostructures. Nat. Commun. 10, 200 (2019).
Mishra, S. et al. Topological frustration induces unconventional magnetism in a nanographene. Nat. Nanotechnol. 15, 22–28 (2020).
Mishra, S. et al. Topological defect-induced magnetism in a nanographene. J. Am. Chem. Soc. 142, 1147–1152 (2020).
Li, J. et al. Uncovering the triplet ground state of triangular graphene nanoflakes engineered with atomic precision on a metal surface. Phys. Rev. Lett. 124, 177201 (2020).
Mishra, S. et al. Collective all-carbon magnetism in triangulene dimers. Angew. Chem. Int. Ed. 59, 12041–12047 (2020).
Ajayakumar, M. R. et al. Toward full zigzag-edged nanographenes: peri-tetracene and its corresponding circumanthracene. J. Am. Chem. Soc. 140, 6240–6244 (2018).
Mishra, S. et al. Tailoring bond topologies in open-shell graphene nanostructures. ACS Nano 12, 11917–11927 (2018).
Rogers, C. et al. Closing the nanographene gap: surface-assisted synthesis of peripentacene from 6,6′-bipentacene precursors. Angew. Chem. Int. Ed. 54, 15143–15146 (2015).
Morita, Y., Suzuki, S., Sato, K. & Takui, T. Synthetic organic spin chemistry for structurally well-defined open-shell graphene fragments. Nat. Chem. 3, 197–204 (2011).
Goto, K. et al. A stable neutral hydrocarbon radical: synthesis, crystal structure and physical properties of 2,5,8-tri-tert-butyl-phenalenyl. J. Am. Chem. Soc. 121, 1619–1620 (1999).
Inoue, J. et al. The first detection of a Clar’s hydrocarbon, 2,6,10-tri-tert-butyltriangulene: a ground-state triplet of non-Kekulé polynuclear benzenoid hydrocarbon. J. Am. Chem. Soc. 123, 12702–12703 (2001).
Pavliček, N. et al. Synthesis and characterization of triangulene. Nat. Nanotechnol. 12, 308–311 (2017).
Mishra, S. et al. Synthesis and characterization of π-extended triangulene. J. Am. Chem. Soc. 141, 10621–10625 (2019).
Su, J. et al. Atomically precise bottom-up synthesis of π-extended [5]triangulene. Sci. Adv. 5, eaav7717 (2019).
Broene, R. D. & Diederich, F. The synthesis of circumanthracene. Tetrahedron Lett. 32, 5227–5230 (1991).
Gu, Y., Wu, X., Gopalakrishna, T. Y., Phan, H. & Wu, J. Graphene-like molecules with four zigzag edges. Angew. Chem. Int. Ed. 57, 6541–6545 (2018).
Gu, Y. et al. peri-Acenoacenes. Chem. Commun. 55, 5567–5570 (2019).
Chen, Q., Schollmeyer, D., Müllen, K. & Narita, A. Synthesis of circumpyrene by alkyne benzannulation of brominated dibenzo[hi,st]ovalene. J. Am. Chem. Soc. 141, 19994–19999 (2019).
Hirjibehedin, C. F., Lutz, C. P. & Heinrich, A. J. Spin coupling in engineered atomic structures. Science 312, 1021–1024 (2006).
Gross, L., Mohn, F., Moll, N., Liljeroth, P. & Meyer, G. The chemical structure of a molecule resolved by atomic force microscopy. Science 325, 1110–1114 (2009).
Kichin, G., Weiss, C., Wagner, C., Tautz, F. S. & Temirov, R. Single molecule and single atom sensors for atomic resolution imaging of chemically complex surfaces. J. Am. Chem. Soc. 133, 16847–16851 (2011).
Peljhan, S. & Kokalj, A. DFT study of gas-phase adsorption of benzotriazole on Cu(111), Cu(100), Cu(110), and low coordinated defects thereon. Phys. Chem. Chem. Phys. 13, 20408–20417 (2011).
Simonov, K. A. et al. From graphene nanoribbons on Cu(111) to nanographene on Cu(110): critical role of substrate structure in the bottom-up fabrication strategy. ACS Nano 9, 8997–9011 (2015).
Khanna, S. K. & Lambe, J. Inelastic electron tunneling spectroscopy. Science 220, 1345–1351 (1983).
Ternes, M. Spin excitations and correlations in scanning tunneling spectroscopy. New J. Phys. 17, 063016 (2015).
Ovchinnikov, A. A. Multiplicity of the ground state of large alternant organic molecules with conjugated bonds. Theoret. Chim. Acta 47, 297–304 (1978).
Lieb, E. H. Two theorems on the Hubbard model. Phys. Rev. Lett. 62, 1201–1204 (1989).
Agapito, L. A., Kioussis, N. & Kaxiras, E. Electric-field control of magnetism in graphene quantum dots: ab initio calculations. Phys. Rev. B 82, 201411 (2010).
Ganguly, S., Kabir, M. & Saha-Dasgupta, T. Magnetic and electronic crossovers in graphene nanoflakes. Phys. Rev. B 95, 174419 (2017).
Golor, M., Koop, C., Lang, T. C., Wessel, S. & Schmidt, M. J. Magnetic correlations in short and narrow graphene armchair nanoribbons. Phys. Rev. Lett. 111, 085504 (2013).
Ortiz, R. et al. Exchange rules for diradical π-conjugated hydrocarbons. Nano Lett. 19, 5991–5997 (2019).
Yang, L., Park, C.-H., Son, Y.-W., Cohen, M. L. & Louie, S. G. Quasiparticle energies and band gaps in graphene nanoribbons. Phys. Rev. Lett. 99, 186801 (2007).
Neaton, J. B., Hybertsen, M. S. & Louie, S. G. Renormalization of molecular electronic levels at metal–molecule interfaces. Phys. Rev. Lett. 97, 216405 (2006).
van der Lit, J. et al. Suppression of electron–vibron coupling in graphene nanoribbons contacted via a single atom. Nat. Commun. 4, 2023 (2013).
Majzik, Z. et al. Studying an antiaromatic polycyclic hydrocarbon adsorbed on different surfaces. Nat. Commun. 9, 1198 (2018).
Schuler, B. et al. Reversible Bergman cyclization by atomic manipulation. Nat. Chem. 8, 220–224 (2016).
Oberg, J. C. et al. Control of single-spin magnetic anisotropy by exchange coupling. Nat. Nanotechnol. 9, 64–68 (2014).
Wehling, T. O. et al. Strength of effective Coulomb interactions in graphene and graphite. Phys. Rev. Lett. 106, 236805 (2011).
Schüler, M., Rösner, M., Wehling, T. O., Lichtenstein, A. I. & Katsnelson, M. I. Optimal Hubbard models for materials with nonlocal Coulomb interactions: graphene, silicene and benzene. Phys. Rev. Lett. 111, 036601 (2013).
Landauer, R. Irreversibility and heat generation in the computing process. IBM J. Res. Dev. 5, 183–191 (1961).
Agarwal, H., Pramanik, S. & Bandyopadhyay, S. Single spin universal Boolean logic gate. New J. Phys. 10, 015001 (2008).
Wang, W. L., Yazyev, O. V., Meng, S. & Kaxiras, E. Topological frustration in graphene nanoflakes: magnetic order and spin logic devices. Phys. Rev. Lett. 102, 157201 (2009).
Savary, L. & Balents, L. Quantum spin liquids: a review. Rep. Prog. Phys. 80, 016502 (2016).
Choi, D.-J. et al. Colloquium: atomic spin chains on surfaces. Rev. Mod. Phys. 91, 041001 (2019).
Horcas, I. et al. WSXM: a software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 78, 013705 (2007).
Tran, V.-T., Saint-Martin, J., Dollfus, P. & Volz, S. Third nearest neighbor parameterized tight binding model for graphene nano-ribbons. AIP Adv. 7, 075212 (2017).
Scalettar, R. T. An Introduction to the Hubbard Hamiltonian, lecture notes, Autumn School on Correlated Electrons: Quantum Materials: Experiments and Theory (Forschungszentrum Jülich, 2016).
Hutter, J., Iannuzzi, M., Schiffmann, F. & VandeVondele, J. CP2K: atomistic simulations of condensed matter systems. Wiley Interdiscip. Rev. Comput. Mol. Sci. 4, 15–25 (2014).
VandeVondele, J. et al. QUICKSTEP: Fast and accurate density functional calculations using a mixed Gaussian and plane waves approach. Comput. Phys. Commun. 167, 103–128 (2005).
Pickett, W. E. Pseudopotential methods in condensed matter applications. Comput. Phys. Rep. 9, 115–197 (1989).
VandeVondele, J. & Hutter, J. Gaussian basis sets for accurate calculations on molecular systems in gas and condensed phases. J. Chem. Phys. 127, 114105 (2007).
Goedecker, S., Teter, M. & Hutter, J. Separable dual-space Gaussian pseudopotentials. Phys. Rev. B 54, 1703–1710 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Gaspari, R. et al. s-orbital continuum model accounting for the tip shape in simulated scanning tunneling microscope images. Phys. Rev. B 84, 125417 (2011).
Talirz, L. Toolkit using the Atomistic Simulation Environment (ASE) (2015); https://github.com/ltalirz/asetk
Tersoff, J. D. & Hamann, D. R. Theory of the scanning tunneling microscope. Phys. Rev. B 31, 805–813 (1985).
Tersoff, J. D. Method for the calculation of scanning tunneling microscope images and spectra. Phys. Rev. B 40, 11990–11993 (1989).
Wilhelm, J., Del Ben, M. & Hutter, J. GW in the Gaussian and plane waves scheme with application to linear acenes. J. Chem. Theory Comput. 12, 3623–3635 (2016).
Kharche, N. & Meunier, V. Width and crystal orientation dependent band gap renormalization in substrate-supported graphene nanoribbons. J. Phys. Chem. Lett. 7, 1526–1533 (2016).
Pizzi, G., Cepellotti, A., Sabatini, R., Marzari, N. & Kozinsky, B. AiiDA: automated interactive infrastructure and database for computational science. Comput. Mater. Sci. 111, 218–230 (2016).
Acknowledgements
We thank D. Passerone for fruitful discussions, D. Schollmeyer for single-crystal X-ray analysis and L. Rotach for technical support. This work was supported by the Swiss National Science Foundation (grant nos. 200020-182015 and IZLCZ2-170184), NCCR MARVEL funded by the Swiss National Science Foundation (grant no. 51NF40-182892), the European Union’s Horizon 2020 research and innovation programme (grant no. 785219, Graphene Flagship Core 2), the Office of Naval Research (N00014-18-1-2708), the Max Planck Society, Ministry of Science and Innovation of Spain (grant nos. PID2019-106114GB-I00 and PID2019-109539GB), Generalitat Valenciana and Fondo Social Europeo (grant no. ACIF/2018/175), MINECO-Spain (grant no. MAT2016-78625) and Portuguese FCT (grant no. UTAPEXPL/ NTec/0046/2017). Computational support from the Swiss Supercomputing Center (CSCS) under project ID s904 is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
R.F., P.R., A.N. and K.M. conceived the project. Q.C. and X.Y. synthesized and characterized the precursor molecules. S.M. performed the STM experiments. S.M. analysed the data with contributions from M.D.G. K.E. and C.A.P. performed the DFT and GW calculations. R.O., J.F.-R., J.C.S.-G., O.G. and S.M. performed the tight-binding and Hubbard calculations. All authors contributed to discussing the results and writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Christopher Ehlert and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–32, notes 1 and 2, containing experimental STM/STS data, theoretical calculations, synthetic procedures and solution characterization data (X-ray diffraction, NMR spectroscopy and high-resolution mass spectrometry), general methods and materials and references.
Supplementary Data 1
Crystal structure of 3, CCDC 1978171.
Supplementary Data 2
Crystal structure of 4, CCDC 1978172.
Supplementary Data 3
Python script to simulate MFH-LDOS maps of the SOMOs and SUMOs of [5]-rhombene with the mean-field Hubbard tight-binding code available at https://github.com/eimrek/tb-mean-field-hubbard. Note that the simulations are performed with nearest-neighbour hopping, with U/t1 = 1.1. The geometry of the system and output images are included.
Supplementary Data 4
Source data for Supplementary Figs. 4a,b and 7.
Source data
Source Data Fig. 4
Source data for Fig. 4a,b,d
Rights and permissions
About this article
Cite this article
Mishra, S., Yao, X., Chen, Q. et al. Large magnetic exchange coupling in rhombus-shaped nanographenes with zigzag periphery. Nat. Chem. 13, 581–586 (2021). https://doi.org/10.1038/s41557-021-00678-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00678-2
This article is cited by
-
Highly entangled polyradical nanographene with coexisting strong correlation and topological frustration
Nature Chemistry (2024)
-
Intelligent synthesis of magnetic nanographenes via chemist-intuited atomic robotic probe
Nature Synthesis (2024)
-
Uncovering the magnetic response of open-shell graphene nanostructures on metallic surfaces at different doping levels
Science China Physics, Mechanics & Astronomy (2024)
-
Synthesis of polyacene by using a metal–organic framework
Nature Synthesis (2023)
-
Synthesis of monolayer and persistent bilayer graphene fragments by using a radical-mediated coupling approach
Nature Synthesis (2023)