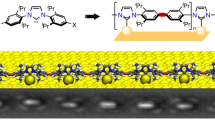
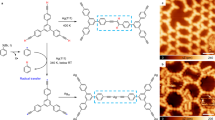
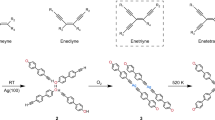
Abstract
Element–element double bonds of group 14 elements can be formed in solution, but generally only by applying harsh reductive conditions using sterically highly shielded tetryl halides as precursors. The two-dimensional confinement in surface-assisted polymerization represents a valuable alternative to access such reactive compounds, as it allows shielding of the labile entities without requiring bulky residues and catalytic activation of the reactive groups. Here, we demonstrate Si–Si bond formation in on-surface chemistry. Polymerization upon multiple Si–H bond dissociation and subsequent Si–Si bond formation was achieved on Au(111) and Cu(111) surfaces by using two different monomers, each containing two silicon functional groups (CH3SiH2 or SiH3) attached to an aromatic backbone, leading to polymeric disilenes that interact with the surface. A combination of experimental and theoretical studies corroborates the formation of covalent Si–Si bonds between the long, highly ordered polymer chains with high diastereoselectivity. The reactive Si=Si bonds formally generated via double dehydrogenative coupling are stabilized via covalent Si–surface interaction.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Supplementary Information is available in the online version of the paper. Reprints and permissions information is available online at www.nature.com/reprints. Data supporting the findings of this work are available within this paper or its Supplementary Information and also from all corresponding authors upon reasonable request. Converged adsorption geometries of all investigated structures can be found in a separate .zip file.
References
West, R., Fink, M. J. & Michl, J. Tetramesityldisilene, a stable compound containing a silicon-silicon double bond. Science 214, 1343–1344 (1981).
Masamune, S., Murakami, S., Snow, J. T., Tobita, H. & Williams, D. J. Molecular structure of tetrakis(2,6-diethylphenyl)disilene. Organometallics 3, 333–334 (1984).
Kira, M., Maruyama, T., Kabuto, C., Ebata, K. & Sakurai, H. Stable tetrakis(trialkylsilyl)disilenes; synthesis, X-ray structures, and UV/VIS spectra. Angew. Chem. Int. Ed. 33, 1489–1491 (1994).
Tokitoh, N., Suzuki, H., Okazaki, R. & Ogawa, K. Synthesis, structure, and reactivity of extremely hindered disilenes: the first example of thermal dissociation of a disilene into a silylene. J. Am. Chem. Soc. 115, 10428–10429 (1993).
Weidenbruch, M. Silylenes and disilenes: examples of low coordinated silicon compounds. Coord. Chem. Rev. 130, 275–300 (1994).
Kaiser, K. et al. An sp-hybridized molecular carbon allotrope, cyclo[18]carbon. Science 365, 1299–1301 (2019).
Gille, M., Grill, L. & Hecht, S. Bottom-up zu molekularen nanostrukturen. Nachr. Chem. 60, 986–990 (2012).
Held, P. A., Fuchs, H. & Studer, A. Covalent-bond formation via on-surface chemistry. Chem. Eur. J. 23, 5874–5892 (2017).
Wang, T. & Zhu, J. Confined on-surface organic synthesis: strategies and mechanisms. Surf. Sci. Rep. 74, 97–140 (2019).
Clair, S. & de Oteyza, D. G. Controlling a chemical coupling reaction on a surface: tools and strategies for on-surface synthesis. Chem. Rev. 119, 4717–4776 (2019).
Grill, L. & Hecht, S. Covalent on-surface polymerization. Nat. Chem. 12, 115–130 (2020).
Pigot, M. & Dumur, F. Molecular engineering in 2D surface covalent organic frameworks: towards next generation of molecular tectons - a mini review. Synth. Met. 260, 116265 (2020).
Grill, L. et al. Nano-architectures by covalent assembly of molecular building blocks. Nat. Nanotechnol. 2, 687–691 (2007).
Cai, J. et al. Atomically precise bottom-up fabrication of graphene nanoribbons. Nature 466, 470–473 (2010).
Shekhirev, M., Zahl, P. & Sinitskii, A. Phenyl functionalization of atomically precise graphene nanoribbons for engineering interribbon interactions and graphene nanopores. ACS Nano 12, 8662–8669 (2018).
Zhong, D. et al. Linear alkane polymerization on a gold surface. Science 334, 213–216 (2011).
Sun, K. et al. Surface-assisted alkane polymerization: investigation on structure–reactivity relationship. J. Am. Chem. Soc. 140, 4820–4825 (2018).
Zhang, X. et al. Coordination-controlled C–C coupling products via ortho-site C–H activation. ACS Nano 13, 1385–1393 (2019).
Rogers, C. et al. Closing the nanographene gap: Surface‐assisted synthesis of peripentacene from 6,6′‐bipentacene precursors. Angew. Chem. Int. Ed. 54, 15143–15146 (2015).
Gao, H.-Y. et al. Glaser coupling at metal surfaces. Angew. Chem. Int. Ed. 52, 4024–4028 (2013).
Sun, Q., Zhang, C., Kong, H., Tan, Q. & Xu, W. On-surface aryl–aryl coupling via selective C–H activation. Chem. Commun. 50, 11825–11828 (2014).
Wiengarten, A. et al. Surface-assisted dehydrogenative homocoupling of porphine molecules. J. Am. Chem. Soc. 136, 9346–9354 (2014).
Sun, Q. et al. On-surface formation of two-dimensional polymer via direct C–H activation of metal phthalocyanine. Chem. Commun. 51, 2836–2839 (2015).
Basagni, A. et al. Stereoselective photopolymerization of tetraphenylporphyrin derivatives on Ag(110) at the sub-monolayer level. Chem. Eur. J. 20, 14296–14304 (2014).
Matena, M. et al. On-surface synthesis of a two-dimensional porous coordination network: Unraveling adsorbate interactions. Phys. Rev. B 90, 125408 (2014).
del Árbol, N. R. et al. On-surface bottom-up synthesis of azine derivatives displaying strong acceptor behavior. Angew. Chem. Int. Ed. 57, 8582–8586 (2018).
Held, P. A. et al. On-surface domino reactions: Glaser coupling and dehydrogenative coupling of a biscarboxylic acid to form polymeric bisacylperoxides. Angew. Chem. Int. Ed. 55, 9777–9782 (2016).
Gao, H. et al. Intermolecular on-surface σ-bond metathesis. J. Am. Chem. Soc. 139, 7012–7019 (2017).
Kawai, S. et al. Diacetylene linked anthracene oligomers synthesized by one-shot homocoupling of trimethylsilyl on Cu(111). ACS Nano 12, 8791–8797 (2018).
Zhang, L. et al. On-surface activation of trimethylsilyl-terminated alkynes on coinage metal surfaces. ChemPhysChem 20, 2382–2393 (2019).
Ekström, U., Ottosson, H. O. & Norman, P. Characterization of the chemisorption of methylsilane on a Au(111) surface from the silicon K- and L-edge spectra: a theoretical study using the four-component static exchange approximation. J. Phys. Chem. C 111, 13846–13850 (2007).
Diller, K. et al. Polyphenylsilole multilayers - an insight from X-ray electron spectroscopy and density functional theory. Phys. Chem. Chem. Phys. 17, 31117–31124 (2015).
Parrill, T. M. & Chung, Y. W. Surface analysis of cubic silicon carbide (001). Surf. Sci. 243, 96–112 (1991).
Hijikata, Y., Yaguchi, H., Yoshikawa, M. & Yoshida, S. Composition analysis of SiO2/SiC interfaces by electron spectroscopic measurements using slope-shaped oxide films. Appl. Surf. Sci 184, 161–166 (2001).
Lee, S., Makan, S., Banaszak Holl, M. M. & McFeely, F. R. Synthetic control of solid/solid interfaces: Analysis of three new silicon/silicon oxide interfaces by soft X-ray photoemission. J. Am. Chem. Soc. 116, 11819–11826 (1994).
Mesarwi, A. & Ignatiev, A. X-ray photoemission study of Y-promoted oxidation of the Si(100) surface. Surf. Sci. 244, 15–21 (1991).
Moulder, J. F., Stickle, W. F., Sobol, P. E. & Bomben, K. D. Handbook of X-ray Photoelectron Spectroscopy: a Reference Book of Standard Spectra for Identification and Interpretation of XPS Data. 56–57 (Physical Electronics Division, Perkin-Elmer Corporation, 1992).
Lin, G.-R. et al. Finite silicon atom diffusion induced size limitation on self-assembled silicon quantum dots in silicon-rich silicon carbide. J. Electrochem. Soc. 159, K35–K41 (2012).
Tsai, H. S., Hsiao, C. H., Chen, C. W., Ouyang, H. & Liang, J. H. Synthesis of nonepitaxial multilayer silicene assisted by ion implantation. Nanoscale 8, 9488–9492 (2016).
Mönig, H. et al. Quantitative assessment of intermolecular interactions by atomic force microscopy imaging using copper oxide tips. Nat. Nanotechnol. 13, 371–375 (2018).
Mönig, H. Copper-oxide tip functionalization for submolecular atomic force microscopy. Chem. Commun. 54, 9874–9888 (2018).
Sekiguchi, A., Kinjo, R. & Ichinohe, M. A stable compound containing a silicon-silicon triple bond. Science 305, 1755–1757 (2004).
Yesilpinar, D. et al. High resolution noncontact atomic force microscopy imaging with oxygen-terminated copper tips at 78 K. Nanoscale 12, 2961–2965 (2020).
Liu, L. et al. α-Diazo ketones in on-surface chemistry. J. Am. Chem. Soc. 140, 6000–6005 (2018).
Campbell, C. T. The degree of rate control: a powerful tool for catalysis research. ACS Catal. 7, 2770 (2017).
Hammond, G. S. A correlation of reaction rates. J. Am. Chem. Soc. 77, 334–338 (1955).
Nilson, A., Pettersson, L. G. M. & Nørskov, J. K. Chemical Bonding at Surfaces and Interfaces 1st edn, Ch. 3 (Elsevier, 2008).
Cao, K., Füchsel, G., Kleyn, A. W. & Juurlink, L. B. F. Hydrogen adsorption and desorption from Cu(111) and Cu(211). Phys. Chem. Chem. Phys. 20, 22477–22488 (2018).
Anger, G., Winkler, A. & Rendulic, K. D. Adsorption and desorption kinetics in the systems H2/Cu(111), H2/Cu(110) and H2/Cu(100). Surf. Sci. 220, 1–17 (1989).
Zhao, M. & Anderson, A. B. Theory of hydrogen deposition and evolution on Cu(111) electrodes. J. Electrochem. Soc. 164, H691–H695 (2017).
Björk, J. in On-surface Synthesis II (eds de Oteyza, D. G. & Rogero, C.) 19–34 (Springer, 2018).
Svane, K. L. & Hammer, B. Thermodynamic aspects of dehydrogenation reactions on noble metal surfaces. J. Chem. Phys. 141, 174705 (2014).
Matsuo, T. & Hayakawa, N. π-Electron systems containing Si=Si double bonds. Sci. Technol. Adv. Mat. 19, 108–129 (2018).
Sasamori, T., Yuasa, A., Hosoi, Y., Furukawa, Y. & Tokitoh, N. 1,2-Bis(ferrocenyl)disilene: a multistep redox system with an Si=Si double bond. Organometallics 27, 3325–3327 (2008).
Kobayashi, M. et al. Air-stable, room-temperature emissive disilenes with π-extended aromatic groups. J. Am. Chem. Soc. 132, 15162–15163 (2010).
Li, L. et al. Coplanar oligo(p-phenylenedisilenylene)s as Si=Si analogues of oligo(p-phenylenevinylene)s: evidence for extended π-conjugation through the carbon and silicon π-frameworks. J. Am. Chem. Soc. 137, 15026–15035 (2015).
Bejan, I. & Scheschkewitz, D. Two Si–Si double bonds connected by a phenylene bridge. Angew. Chem. Int. Ed. 46, 5784–5786 (2007).
Fukazawa, A., Li, Y., Yamaguchi, S., Tsuji, H. & Tamao, K. Coplanar oligo(p-phenylenedisilenylene)s based on the octaethyl-substituted s-hydrindacenyl groups. J. Am. Chem. Soc. 129, 14164–14165 (2007).
Tamao, K., Kobayashi, M., Matsuo, T., Furukawa, S. & Tsuji, H. The first observation of electroluminescence from di(2-naphthyl)disilene, an Si=Si double bond-containing π-conjugated compound. Chem. Commun. 48, 1030–1032 (2012).
Obeid, N. M. et al. (Oligo)aromatic species with one or two conjugated Si=Si bonds: near-IR emission of anthracenyl-bridged tetrasiladiene. Dalton Trans. 46, 8839–8848 (2017).
Ulman, A. Formation and structure of self-assembled monolayers. Chem. Rev. 96, 1533–1554 (1996).
Schreiber, F. Structure and growth of self-assembling monolayers. Prog. Surf. Sci. 65, 151–256 (2000).
Love, J. C., Estroff, L. A., Kriebel, J. K., Nuzzo, R. G. & Whitesides, G. M. Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem. Rev. 105, 1103–1169 (2005).
Elemans, J. A. A. W., Lei, S. & De Feyter, S. Molecular and supramolecular networks on surfaces: from two-dimensional crystal engineering to reactivity. Angew. Chem. Int. Ed. 48, 7298–7332 (2009).
McCreery, R. L., Yana, H. & Bergrena, A. J. A critical perspective on molecular electronic junctions: there is plenty of room in the middle. Phys. Chem. Chem. Phys. 15, 1065–1081 (2013).
Nicholson, K. T., Zhang, K. Z. & Banaszak Holl, M. M. Chemisorption of H8Si8O12 clusters on gold: a novel Si–H bond activation. J. Am. Chem. Soc. 121, 3232–3233 (1999).
Fadeev, A. Y. & McCarthy, T. J. A new route to covalently attached monolayers: reaction of hydridosilanes with titanium and other metal surfaces. J. Am. Chem. Soc. 121, 12184–12185 (1999).
Klare, H. F. T. et al. Cooperative catalytic activation of Si−H bonds by a polar Ru−S bond: regioselective low-temperature C−H silylation of indoles under neutral conditions by a Friedel−Crafts mechanism. J. Am. Chem. Soc. 133, 3312–3315 (2011).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab-initio total energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996) ; erratum 78, 1396 (1997).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Methfessel, M. & Paxton, A. T. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 40, 3616–3621 (1989).
Neugebauer, J. & Scheffler, M. Adsorbate-substrate and adsorbate-adsorbate interactions of Na and K adlayers on Al(111). Phys. Rev. B 46, 16067–16080 (1992).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Becke, A. D. & Johnson, E. R. A density-functional model of the dispersion interaction. J. Chem. Phys. 123, 154101 (2005).
Johnson, E. R. & Becke, A. D. A post-Hartree–Fock model of intermolecular interactions. J. Chem. Phys. 123, 024101 (2005).
Johnson, E. R. & Becke, A. D. A post-Hartree–Fock model of intermolecular interactions: inclusion of higher-order corrections. J. Chem. Phys. 124, 174104 (2006).
Larsen, A. H. et al. The atomic simulation environment - a Python library for working with atoms. J. Phys. Condens. Matter 29, 273002 (2017).
Bahn, S. R. & Jacobsen, K. W. An object-oriented scripting interface to a legacy electronic structure code. Comput. Sci. Eng. 4, 56–66 (2002).
Straumanis, M. E. & Yu, L. S. Lattice parameters, densities, expansion coefficients and perfection of structure of Cu and of Cu–In α phase. Acta Cryst A25, 676–682 (1969).
Kittel, C. Introduction to Solid State Physics 7th edn (Wiley, 1996).
Hanwell, M. D. et al. Avogadro: an advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 4, 17 (2012).
Furche, F. et al. Turbomole. Wiley Interdiscip. Rev. Comput. Mol. Sci. 4, 91–100 (2014).
Schäfer, A., Horn, H. & Ahlrichs, R. Fully optimized contracted Gaussian basis sets for atoms Li to Kr. J. Chem. Phys. 97, 2571–2577 (1992).
Eichkorn, K., Treutler, O., Öhm, H., Häser, M. & Ahlrichs, R. Auxiliary basis sets to approximate Coulomb potentials. Chem. Phys. Lett. 240, 283–290 (1995).
Eichkorn, K., Weigend, F., Treutler, O. & Ahlrichs, R. Auxiliary basis sets for main row atoms and transition metals and their use to approximate Coulomb potentials. Theor. Chem. Acc. 97, 119–124 (1997).
Tersoff, J. & Hamann, D. R. Theory and application for the scanning tunneling microscope. Phys. Rev. Lett. 50, 1998–2001 (1983).
Blum, V. et al. Ab initio molecular simulations with numeric atom-centered orbitals. Comput. Phys. Commun. 180, 2175–2196 (2009).
Humphrey, W., Dalke, A. & Schulten, K. VMD - visual molecular dynamics. J. Molec. Graphics 14, 33–38 (1996).
Horcas, I. et al. WSXM: a software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 78, 013705 (2007).
Acknowledgements
We gratefully acknowledge financial support from the Deutsche Forschungsgemeinschaft (DFG) within the collaborative research centres SFB 858 (projects B02, Z01) and TRR61 (projects B03 and B07). H.M. and H.F. further thank the DFG for support through projects MO 2345/4-1 and FU 299/19. H.-Y.G. acknowledges financial support from NSFC within Grant 21972104 and ‘1000-Youth Talents Plan’. We thank D. Yesilpinar for technical support during the AFM measurements.
Author information
Authors and Affiliations
Contributions
L.L. performed the STM experiments. H. Kong participated in the STM experiments of BDS 3. H. Klaasen performed organic synthesis. M.C.W. performed theoretical studies. B.S.L., L.L. and A.T. performed XPS experiments and B.S.L. and L.L. performed the nc-AFM experiments, both experiments were supervised by H.M. All authors discussed the results. H.-Y.G., J.N., H.F. and A.S. supervised the project and designed the experiments. L.L., H. Klaasen, M.C.W., A.T., H.-Y.G. and A.S. wrote the manuscript with suggestions from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks the, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Chemical synthesis, Supplementary Figs. 1–30, Tables 1–3, Discussion and NMR spectra of new compounds.
Supplementary Data
Zip-file containing the converged adsorption geometries of all investigated structures as .txt files.
Rights and permissions
About this article
Cite this article
Liu, L., Klaasen, H., Witteler, M.C. et al. Polymerization of silanes through dehydrogenative Si–Si bond formation on metal surfaces. Nat. Chem. 13, 350–357 (2021). https://doi.org/10.1038/s41557-021-00651-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00651-z
This article is cited by
-
Universal inter-molecular radical transfer reactions on metal surfaces
Nature Communications (2024)
-
On-surface synthesis of disilabenzene-bridged covalent organic frameworks
Nature Chemistry (2023)
-
Selective activation of four quasi-equivalent C–H bonds yields N-doped graphene nanoribbons with partial corannulene motifs
Nature Communications (2022)