Abstract
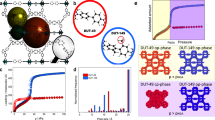
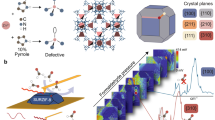
Competitive water adsorption can have a significant impact on metal–organic framework performance properties, ranging from occupying active sites in catalytic reactions to co-adsorbing at the most favourable adsorption sites in gas separation and storage applications. In this study, we investigate, for a metal–organic framework that is stable after moisture exposure, what are the reversible, loading-dependent structural changes that occur during water adsorption. Herein, a combination of in situ synchrotron powder and single-crystal diffraction, infrared spectroscopy and molecular modelling analysis was used to understand the important role of loading-dependent water effects in a water stable metal–organic framework. Through this analysis, insights into changes in crystallographic lattice parameters, water siting information and water-induced defect structure as a response to water loading were obtained. This work shows that, even in stable metal–organic frameworks that maintain their porosity and crystallinity after moisture exposure, important molecular-level structural changes can still occur during water adsorption due to guest–host interactions such as water-induced bond rearrangements.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data supporting the claims and findings of this paper are available within the Supplementary Information or are available upon request from the corresponding author. Crystallographic data for all structures reported in this paper have been deposited with the Cambridge Crystallographic Data Centre (CCDC), CCDC numbers 1864840 (DMOF-TM_crystal 09_Flow_01), 1864836 (DMOF-TM_crystal 09_Flow_02), 1864833 (DMOF-TM_crystal 09_Flow_03), 1864842 (DMOF-TM_crystal 09_Flow_04), 1864834 (DMOF-TM_crystal 09_Flow_05), 1864835 (DMOF-TM_crystal 09_Flow_06), 1864838 (DMOF-TM_crystal 09_Flow_07), 1864837 (DMOF-TM_crystal 09_Flow_08), 1864839 (DMOF-TM_crystal 09, static) and 1864841 (DMOF-1, crystal 15, static; where crystal 09 and crystal 15 were both selected from the same batch). Copies of the crystallographic data can be obtained at https://www.ccdc.cam.ac.uk/structures/ free of charge. A SCXRD structure of the ‘activated’ DMOF-TM structure could not be obtained; unit cell parameters and atomic positions were obtained by Rietveld refinement, refined to the SCXRD data from this study (P4/nbm) and also to the structure previously reported for this material (P4/mmm).
References
Zhou, H.-C., Long, J. R. & Yaghi, O. M. Introduction to metal–organic frameworks. Chem. Rev. 112, 673–674 (2012).
Furukawa, H., Cordova, K. E., O’Keeffe, M. & Yaghi, O. M. The chemistry and applications of metal–organic frameworks. Science 341, 1230444 (2013).
Kreno, L. E. et al. Metal–organic framework materials as chemical sensors. Chem. Rev. 112, 1105–1125 (2012).
Stassen, I. et al. An updated roadmap for the integration of metal–organic frameworks with electronic devices and chemical sensors. Chem. Soc. Rev. 46, 3185–3241 (2017).
Nilsson, A. & Pettersson, L. G. M. The structural origin of anomalous properties of liquid water. Nat. Commun. 6, 8998 (2015).
Burtch, N. C., Jasuja, H. & Walton, K. S. Water stability and adsorption in metal–organic frameworks. Chem. Rev. 114, 10575–10612 (2014).
Bosch, M., Zhang, M. & Zhou, H. Increasing the stability of metal–organic frameworks. Adv. Chem. 2014, 1–8 (2014).
Howarth, A. J. et al. Chemical, thermal and mechanical stabilities of metal–organic frameworks. Nat. Rev. Mater. 1, 15018 (2016).
Canivet, J., Fateeva, A., Guo, Y., Coasne, B. & Farrusseng, D. Water adsorption in MOFs: fundamentals and applications. Chem. Soc. Rev. 43, 5594–5617 (2014).
Horike, S., Shimomura, S. & Kitagawa, S. Soft porous crystals. Nat. Chem. 1, 695–704 (2009).
Schneemann, A. et al. Flexible metal–organic frameworks. Chem. Soc. Rev. 43, 6062–6096 (2014).
Zeng, M. H. et al. Apical ligand substitution, shape recognition, vapor-adsorption phenomenon, and microcalorimetry for a pillared bilayer porous framework that shrinks or expands in crystal-to-crystal manners upon change in the cobalt(II) coordination environment. Inorg. Chem. 48, 7070–7079 (2009).
Bennett, T. D., Cheetham, A. K., Fuchs, A. H. & Coudert, F.-X. Interplay between defects, disorder and flexibility in metal–organic frameworks. Nat. Chem. 9, 11–16 (2017).
Sholl, D. S. & Lively, R. P. Defects in metal–organic frameworks: challenge or opportunity? J. Phys. Chem. Lett. 6, 3437–3444 (2015).
Fang, Z., Bueken, B., De Vos, D. E. & Fischer, R. A. Defect-engineered metal–organic frameworks. Angew. Chem. Int. Ed. 54, 7234–7254 (2015).
Trickett, C. A. et al. Definitive molecular level characterization of defects in UiO-66 crystals. Angew. Chem. Int. Ed. 54, 11162–11167 (2015).
Chun, H., Dybtsev, D. N., Kim, H. & Kim, K. Synthesis, X-ray crystal structures, and gas sorption properties of pillared square grid nets based on paddle-wheel motifs: implications for hydrogen storage in porous materials. Chem. Eur. J. 11, 3521–3529 (2005).
Dybtsev, D. N., Chun, H. & Kim, K. Rigid and flexible: a highly porous metal–organic framework with unusual guest-dependent dynamic behavior. Angew. Chem. Int. Ed. 43, 5033–5036 (2004).
Burtch, N. C. & Walton, K. S. Modulating adsorption and stability properties in pillared metal−organic frameworks: a model system for understanding ligand effects. Acc. Chem. Res. 48, 2850–2857 (2015).
Jasuja, H., Burtch, N. C., Huang, Y., Cai, Y. & Walton, K. S. Kinetic water stability of an isostructural family of zinc-based pillared metal–organic frameworks. Langmuir 29, 633–642 (2013).
Jasuja, H., Jiao, Y., Burtch, N. C., Huang, Y. & Walton, K. S. Synthesis of cobalt-, nickel -, copper -, and zinc-based, water-stable, pillared metal−organic frameworks. Langmuir 30, 14300–14307 (2014).
Lu, W. et al. Tuning the structure and function of metal–organic frameworks via linker design. Chem. Soc. Rev. 43, 5561–5593 (2014).
Pecharsky, V. K. & Zavalij, P. Y. Fundamentals of Powder Diffraction and Structural Characterization of Materials Vol. 69 (Springer, 2009).
Mittemeijer, E. J. & Welzel, U. The “state of the art” of the diffraction analysis of crystallite size and lattice strain. Z. Kristallogr. 223, 552–560 (2008).
Gor, G. Y., Huber, P. & Bernstein, N. Adsorption-induced deformation of nanoporous materials—a review. Appl. Phys. Rev. 4, 011303 (2017).
Pawley, G. S. Unit-cell refinement from powder diffraction scans. J. Appl. Crystallogr. 14, 357–361 (1981).
Rietveld, H. M. A profile refinement method for nuclear and magnetic structures. J. Appl. Cryst. 2, 65–71 (1969).
Ravikovitch, P. I. & Neimark, A. V. Density functional theory model of adsorption deformation. Langmuir 22, 10864–10868 (2006).
Moggach, S. A., Bennett, T. D. & Cheetham, A. K. The effect of pressure on ZIF-8: increasing pore size with pressure and the formation of a high-pressure phase at 1.47 GPa. Angew. Chem. Int. Ed. 121, 7221–7223 (2009).
Hobday, C. L. et al. Understanding the adsorption process in ZIF-8 using high pressure crystallography and computational modelling. Nat. Commun. 9, 1429 (2018).
Burtch, N. C. et al. Understanding DABCO nanorotor dynamics in isostructural metal–organic frameworks. J. Phys. Chem. Lett. 6, 812–816 (2015).
Cox, J. M. et al. Solvent exchange in a metal–organic framework single crystal monitored by dynamic in situ X-ray diffraction. Acta Crystallogr. B 73, 669–674 (2017).
Cox, J. M., Walton, I. M., Benson, C. A., Chen, Y.-S. & Benedict, J. B. A versatile environmental control cell for in situ guest exchange single-crystal diffraction. J. Appl. Crystallogr. 48, 578–581 (2015).
Yakovenko, A. A. et al. Study of guest molecules in metal–organic frameworks by powder X-ray diffraction: analysis of difference envelope density. Cryst. Growth Des. 14, 5397–5407 (2014).
Chen, Y.-P., Liu, Y., Liu, D., Bosch, M. & Zhou, H.-C. Direct measurement of adsorbed gas redistribution in metal–organic frameworks. J. Am. Chem. Soc. 137, 2919–2930 (2015).
Tan, J. C. & Cheetham, A. K. Mechanical properties of hybrid inorganic–organic framework materials: establishing fundamental structure–property relationships. Chem. Soc. Rev. 40, 1059–1080 (2011).
Burtch, N. C., Heinen, J., Bennett, T. D., Dubbeldam, D. & Allendorf, M. D. Mechanical properties in metal–organic frameworks: emerging opportunities and challenges for device functionality and technological applications. Adv. Mater. 30, 1704124 (2018).
Petkov, P. S. et al. Conformational isomerism controls collective flexibility in metal–organic framework DUT-8 (Ni). Phys. Chem. Chem. Phys. 21, 674–680 (2019).
Chupas, P. J. et al. A versatile sample-environment cell for non-ambient X-ray scattering experiments. Appl. Crystallogr. 41, 822–824 (2008).
Toby, B. H. & Von Dreele, R. B. GSAS-II: the genesis of a modern open-source all purpose crystallography software package. J. Appl. Crystallogr. 46, 544–549 (2013).
Dubbeldam, D., Calero, S., Ellis, D. E. & Snurr, R. Q. RASPA: molecular simulation software for adsorption and diffusion in flexible nanoporous materials. Mol. Simul. 42, 81–101 (2016).
Burtch, N. C., Dubbeldam, D. & Walton, K. S. Investigating water and framework dynamics in pillared MOFs. Mol. Simul. 41, 1379–1387 (2015).
Gaillac, R., Pullumbi, P. & Coudert, F.-X. ELATE: an open-source online application for analysis and visualization of elastic tensors. J. Phys. Condens. Matter 28, 275201 (2016).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Acknowledgements
This work was supported as a part of the Center for Understanding and Control of Acid Gas-Induced Evolution of Materials for Energy, an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, Basic Energy Sciences under award no. DE-SC0012577. Use of the Advanced Photon Source was supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences, under contract no. DE-AC02-06CH11357. N.C.B. acknowledges support from the National Science Foundation Graduate Research Fellowship and the Graduate Research Opportunities Worldwide (GROW) award under grant no. DGE-1148903. D.D. acknowledges support from the Netherlands Research Council for Chemical Sciences through a VIDI grant and the Dutch Research Council (NWO) Exacte Wetenschappen (Physical Sciences) for the use of supercomputer facilities with financial support from the NWO. NSF’s ChemMatCARS Sector 15 is supported by the Divisions of Chemistry (CHE) and Materials Research (DMR), National Science Foundation, under grant no. NSF/CHE- 1834750. Use of the Advanced Photon Source, an Office of Science User Facility operated for the US Department of Energy Office of Science by Argonne National Laboratory, was supported by the US Department of Energy under contract no. DE-AC02-06CH11357.
Author information
Authors and Affiliations
Contributions
All authors contributed extensively to the work presented in this paper. N.C.B., C.R.M., Y.J. and J.T.H. performed the powder synthesis and adsorption characterization work, and C.R.M. led the synchrotron diffraction analysis with help from N.C.B., A.A.Y. and W.X. The computational studies were performed by N.C.B., J.H. and D.D. SCXRD experiments and analysis were led by I.M.W. with assistance from Y.-S.C. and J.T.H. IR spectroscopy experiments and analysis were performed by J.T.H. and I.M.W. The manuscript was written by N.C.B., I.M.W and K.S.W. with input from the other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Sample synthesis and preparation; characterization; molecular dynamics simulations; theoretical defects calculations; Supplementary Figures 1–46; references 1–33.
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 1; CCDC reference 1864840
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 1; CCDC reference 1864840
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 2; CCDC reference 1864836
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 2; CCDC reference 1864836
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 3; CCDC reference 1864833
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 3; CCDC reference 1864833
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 4; CCDC reference 1864842
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 4; CCDC reference 1864842
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 5; CCDC reference 1864834
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 5; CCDC reference 1864834
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 6; CCDC reference 1864835
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 6; CCDC reference 1864835
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 7; CCDC reference 1864838
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 7; CCDC reference 1864838
Crystallographic data
CIF for DMOF-TM, crystal 09, Flow 8; CCDC reference 1864837
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Flow 8; CCDC reference 1864837
Crystallographic data
CIF for DMOF-TM, crystal 09, Static; CCDC reference 1864839
Crystallographic data
Structure factors for DMOF-TM, crystal 09, Static; CCDC reference 1864839
Crystallographic data
CIF for DMOF-TM, crystal 15, Static; CCDC reference 1864841
Crystallographic data
Structure factors for DMOF-TM, crystal 15, Static; CCDC reference 1864841
Crystallographic data
CIF for the activated DMOF-TM, refined to P4/nbm
Crystallographic data
CIF for the activated DMOF-TM, refined to P4/mmm
Rights and permissions
About this article
Cite this article
Burtch, N.C., Walton, I.M., Hungerford, J.T. et al. In situ visualization of loading-dependent water effects in a stable metal–organic framework. Nat. Chem. 12, 186–192 (2020). https://doi.org/10.1038/s41557-019-0374-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0374-y
This article is cited by
-
Desolvation of metal complexes to construct metal–organic framework glasses
Nature Synthesis (2023)
-
Water-assisted hydrogen spillover in Pt nanoparticle-based metal–organic framework composites
Nature Communications (2023)
-
Crystalline hydrogen bonding of water molecules confined in a metal-organic framework
Communications Chemistry (2022)
-
Defects engineering simultaneously enhances activity and recyclability of MOFs in selective hydrogenation of biomass
Nature Communications (2022)
-
Water induced ultrathin Mo2C nanosheets with high-density grain boundaries for enhanced hydrogen evolution
Nature Communications (2022)