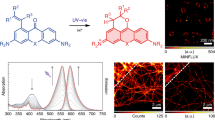
Abstract
Live-cell fluorescence nanoscopy is a powerful tool to study cellular biology on a molecular scale, yet its use is held back by the paucity of suitable fluorescent probes. Fluorescent probes based on regular fluorophores usually suffer from a low cell permeability and an unspecific background signal. Here we report a general strategy to transform regular fluorophores into fluorogenic probes with an excellent cell permeability and a low unspecific background signal. Conversion of a carboxyl group found in rhodamines and related fluorophores into an electron-deficient amide does not affect the spectroscopic properties of the fluorophore, but allows us to rationally tune the dynamic equilibrium between two different forms: a fluorescent zwitterion and a non-fluorescent, cell-permeable spirolactam. Furthermore, the equilibrium generally shifts towards the fluorescent form when the probe binds to its cellular targets. The resulting increase in fluorescence can be up to 1,000-fold. Using this simple design principle, we created fluorogenic probes in various colours for different cellular targets for wash-free, multicolour, live-cell nanoscopy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information. Additional information and files are available from the corresponding author upon reasonable request.
References
Wang, L., Frei, M. S., Salim, A. & Johnsson, K. Small-molecule fluorescent probes for live-cell super-resolution microscopy. J. Am. Chem. Soc. 141, 2770–2781 (2018).
Liu, Z., Lavis, L. D. & Betzig, E. Imaging live-cell dynamics and structure at the single-molecule level. Mol. Cell 58, 644–659 (2015).
Xue, L., Karpenko, I. A., Hiblot, J. & Johnsson, K. Imaging and manipulating proteins in live cells through covalent labeling. Nat. Chem. Biol. 11, 917–923 (2015).
Pellett, P. A. et al. Two-color STED microscopy in living cells. Biomed. Opt. Express 2, 2364–2371 (2011).
van de Linde, S., Heilemann, M. & Sauer, M. Live-cell super-resolution imaging with synthetic fluorophores. Annu. Rev. Phys. Chem. 63, 519–540 (2012).
Jones, S. A., Shim, S. H., He, J. & Zhuang, X. Fast, three-dimensional super-resolution imaging of live cells. Nat. Methods 8, 499–508 (2011).
McNeil, P. L. & Warder, E. Glass-beads load macromolecules into living cells. J. Cell Sci. 88, 669–678 (1987).
Lucy, D. B. et al. Small molecule injection into single-cell C. elegans embryos via carbon-reinforced nanopipettes. PLoS ONE 8, e75712 (2013).
Kollmannsperger, A. et al. Live-cell protein labelling with nanometre precision by cell squeezing. Nat. Commun. 7, 10372–10378 (2016).
Hori, Y. & Kikuchi, K. Protein labeling with fluorogenic probes for no-wash live-cell imaging of proteins. Curr. Opin. Chem. Biol. 17, 644–650 (2013).
Lavis, L. D. Teaching old dyes new tricks: biological probes built from fluoresceins and rhodamines. Annu. Rev. Biochem. 86, 825–843 (2017).
Lukinavicius, G. et al. A near-infrared fluorophore for live-cell super-resolution microscopy of cellular proteins. Nat. Chem. 5, 132–139 (2013).
Grimm, J. B. et al. A general method to fine-tune fluorophores for live-cell and in vivo imaging. Nat. Methods 14, 987–994 (2017).
Lukinavicius, G. et al. Fluorogenic probes for live-cell imaging of the cytoskeleton. Nat. Methods 11, 731–733 (2014).
Lukinavicius, G. et al. SiR-Hoechst is a far-red DNA stain for live-cell nanoscopy. Nat. Commun. 6, 8497–8503 (2015).
Butkevich, A. N. et al. Fluorescent rhodamines and fluorogenic carbopyronines for super-resolution STED microscopy in living cells. Angew. Chem. Int. Ed. 55, 3290–3294 (2016).
Lukinavicius, G. et al. Fluorogenic probes for multicolor imaging in living cells. J. Am. Chem. Soc. 138, 9365–9368 (2016).
Johnson, L. V., Walsh, M. L. & Chen, L. B. Localization of mitochondria in living cells with rhodamine 123. Proc. Natl Acad. Sci. USA 77, 990–994 (1980).
Poot, M. et al. Analysis of mitochondrial morphology and function with novel fixable fluorescent stains. J. Histochem. Cytochem. 44, 1363–1372 (1996).
Umezawa, K., Yoshida, M., Kamiya, M., Yamasoba, T. & Urano, Y. Rational design of reversible fluorescent probes for live-cell imaging and quantification of fast glutathione dynamics. Nat. Chem. 9, 279–286 (2017).
Umezawa, K., Kamiya, M. & Urano, Y. A reversible fluorescent probe for real-time live-cell imaging and quantification of endogenous hydropolysulfides. Angew. Chem. Int. Ed. 57, 9346–9350 (2018).
Grzybowski, M. et al. A highly photostable near-infrared labeling agent based on a phospha-rhodamine for long-term and deep imaging. Angew. Chem. Int. Ed. 57, 10137–10141 (2018).
Kim, H. N., Lee, M. H., Kim, H. J., Kim, J. S. & Yoon, J. A new trend in rhodamine-based chemosensors: application of spirolactam ring-opening to sensing ions. Chem. Soc. Rev. 37, 1465–1472 (2008).
Beija, M., Afonso, C. A. M. & Martinho, J. M. G. Synthesis and applications of rhodamine derivatives as fluorescent probes. Chem. Soc. Rev. 38, 2410–2433 (2009).
Li, H. et al. An acid catalyzed reversible ring-opening/ring-closure reaction involving a cyano-rhodamine spirolactam. Org. Biomol. Chem. 11, 1805–1809 (2013).
Ammazzalorso, A., De Filippis, B., Giampietro, L. & Amoroso, R. N-acylsulfonamides: Synthetic routes and biological potential in medicinal chemistry. Chem. Biol. Drug Des. 90, 1094–1105 (2017).
Verkade, J. M. M. et al. A polar sulfamide spacer significantly enhances the manufacturability, stability, and therapeutic index of antibody–drug conjugates. Antibodies 7, 12–23 (2018).
Keppler, A. et al. A general method for the covalent labeling of fusion proteins with small molecules in vivo. Nat. Biotechnol. 21, 86–89 (2003).
Los, G. V. et al. HaloTag: a novel protein labeling technology for cell imaging and protein analysis. ACS Chem. Biol. 3, 373–382 (2008).
Keppler, A., Pick, H., Arrivoli, C., Vogel, H. & Johnsson, K. Labeling of fusion proteins with synthetic fluorophores in live cells. Proc. Natl Acad. Sci. USA 101, 9955–9959 (2004).
Milroy, L. G. et al. Selective chemical imaging of static actin in live cells. J. Am. Chem. Soc. 134, 8480–8486 (2012).
Cai, S. X. et al. Design and synthesis of rhodamine 110 derivative and caspase-3 substrate for enzyme and cell-based fluorescent assay. Bioorg. Med. Chem. Lett. 11, 39–42 (2001).
Stagge, F., Mitronova, G. Y., Belov, V. N., Wurm, C. A. & Jakobs, S. SNAP-, CLIP- and Halo-Tag labelling of budding yeast cells. PLoS ONE 8, e78745 (2013).
Liu, Y. et al. The cation–π interaction enables a Halo-Tag fluorogenic probe for fast no-wash live cell imaging and gel-free protein quantification. Biochemistry 56, 1585–1595 (2017).
Escobedo, J. O., Rusin, O., Lim, S. & Strongin, R. M. NIR dyes for bioimaging applications. Curr. Opin. Chem. Biol. 14, 64–70 (2010).
Koide, Y. et al. Development of NIR fluorescent dyes based on Si-rhodamine for in vivo imaging. J. Am. Chem. Soc. 134, 5029–5031 (2012).
Paintrand, M., Moudjou, M., Delacroix, H. & Bornens, M. Centrosome organization and centriole architecture: their sensitivity to divalent cations. J. Struct. Biol. 108, 107–128 (1992).
Malecki, M. J. et al. Leukemia-associated mutations within the NOTCH1 heterodimerization domain fall into at least two distinct mechanistic classes. Mol. Cell. Biol. 26, 4642–4651 (2006).
Acknowledgements
We acknowledge funding from the Max Planck Society. We thank S. Jakobs for providing the U2OS Vimentin-HaloTag cells. J. Hubrich and C.-M. Gürth supported the cell culture and preparation of neurons. We are grateful to S. Pitsch for the gift of SiR700 and to L. Reymond for the gift of carbopyronine. L.W. and M.T. were supported by fellowships of the Alexander von Humboldt Foundation.
Author information
Authors and Affiliations
Contributions
All the authors discussed the results and commented on the manuscript. L.W. and K.J. designed the strategy and fluorophore structures. L.W., M.T. and L.X performed the chemical syntheses. L.W. and M.T. characterized the dyes and performed the confocal microscopy with subsequent data analysis. E.D. and J.R. performed the STED microscopy with subsequent data analysis. B.K. developed the cell lines.
Corresponding authors
Ethics declarations
Competing interests
K.J. and L.W. are inventors of the patent ‘Cell-permeable fluorogenic fluorophores’ (EP Patent Application 18210676.5, pending), which was filed by the Max Planck Society.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplementary information providing details of the experimental methods, Supplementary Figs. 1–30, Tables 1–4 and refs. 1–4.
Supplementary Video 1
No-wash real-time multicolour confocal microscopy of U2OS FlpIn Cox8-Halo-SNAP-expressing cells stained with Hoechst 33342 (0.2 μg ml–1)/MaP555-tubulin (1 μM)/MaP618-actin (500 nM)/MaP700-Halo (250 nM).
Supplementary Video 2
No-wash real-time multicolour confocal microscopy of U2OS FlpIn Cox8-Halo-SNAP-expressing cells stained with Hoechst 33342 (0.2 μg ml–1)/MaP510-Halo (250 nM)/MaP555-actin (1 μM)/SiR-Lyso (1 μM).
Supplementary Video 3
No-wash real-time multicolour confocal microscopy of U2OS FlpIn Halo-SNAP-NLS-expressing cells stained with MaP510-Halo (250 nM)/MaP555-tubulin (1 μM)/MaP618-actin (500 nM)/SiR-Lyso (1 μM).
Rights and permissions
About this article
Cite this article
Wang, L., Tran, M., D’Este, E. et al. A general strategy to develop cell permeable and fluorogenic probes for multicolour nanoscopy. Nat. Chem. 12, 165–172 (2020). https://doi.org/10.1038/s41557-019-0371-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0371-1
This article is cited by
-
A general strategy to develop fluorogenic polymethine dyes for bioimaging
Nature Chemistry (2024)
-
Bioorthogonal Reactions in Bioimaging
Topics in Current Chemistry (2024)
-
OregonFluor enables quantitative intracellular paired agent imaging to assess drug target availability in live cells and tissues
Nature Chemistry (2023)
-
A general highly efficient synthesis of biocompatible rhodamine dyes and probes for live-cell multicolor nanoscopy
Nature Communications (2023)
-
Design of a palette of SNAP-tag mimics of fluorescent proteins and their use as cell reporters
Cell Discovery (2023)