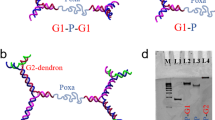
Abstract
Frank–Kasper phases, a family of ordered structures formed from particles with spherical motifs, are found in a host of materials, such as metal alloys, inorganic colloids and various types of soft matter. All the experimentally observed Frank–Kasper phases can be constructed from the basic units of three fundamental structures called the A15, C15 and Z phases. The Z phase, typically observed in metal alloys, is associated with a relatively large volume ratio between its constituents, and this constraint inhibits its formation in most self-assembled single-component soft-matter systems. We have assembled a series of nanosized shape amphiphiles that comprise a triphenylene core and six polyhedral oligomeric silsesquioxane cages grafted onto it through linkers to give a variety of unconventional structures, which include the Z phase. This structure was obtained through fine tuning of the linker lengths between the core and the peripheral polyhedral oligomeric silsesquioxane cages, and exhibits a relatively large volume asymmetry between its constituent polyhedral particle motifs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information, and/or from the corresponding author upon reasonable request.
References
Hyde, S. et al. The Language of Shape: The Role of Curvature in Condensed Matter: Physics, Chemistry and Biology (Elsevier Science, 1996).
Shechtman, D., Blech, I., Gratias, D. & Cahn, J. W. Metallic phase with long-range orientational order and no translational symmetry. Phys. Rev. Lett. 53, 1951–1953 (1984).
Frank, F. C. & Kasper, J. S. Complex alloy structures regarded as sphere packings. I. Definitions and basic principles. Acta Crystallogr. 11, 184–190 (1958).
Frank, F. C. & Kasper, J. S. Complex alloy structures regarded as sphere packings. II. Analysis and classification of representative structures. Acta Crystallogr. 12, 483–499 (1959).
Zeng, X. et al. Supramolecular dendritic liquid quasicrystals. Nature 428, 157 (2004).
Gillard, T. M., Lee, S. & Bates, F. S. Dodecagonal quasicrystalline order in a diblock copolymer melt. Proc. Natl Acad. Sci. USA 113, 5167–5172 (2016).
Keys, A. S. & Glotzer, S. C. How do quasicrystals grow? Phys. Rev. Lett. 99, 235503 (2007).
Dutour Sikiric, M., Delgado-Friedrichs, O. & Deza, M. Space fullerenes: a computer search for new Frank–Kasper structures. Acta Crystallogr. A 66, 602–615 (2010).
Shoemaker, D. P. & Shoemaker, C. B. Concerning the relative numbers of atomic coordination types in tetrahedrally close packed metal structures. Acta Crystallogr. B 42, 3–11 (1986).
Pauling, L. & Marsh, R. E. The structure of chlorine hydrate. Proc. Natl Acad. Sci. USA 38, 112–118 (1952).
Baerlocher, Ch., McCusker. L. B. & Olson, D.H. Atlas of Zeolite Framework Types 6th edn (Elsevier Science, 2007).
Lin, H. et al. Clathrate colloidal crystals. Science 355, 931–935 (2017).
Vargas, R., Mariani, P., Gulik, A. & Luzzati, V. Cubic phases of lipid-containing systems: the structure of phase Q223 (space group Pm3n). An -ray scattering study. J. Mol. Biol. 225, 137–145 (1992).
Luzzati, V. et al. Lipid polymorphism: a correction. The structure of the cubic phase of extinction symbol Fdc– consists of two types of disjointed reverse micelles embedded in a three-dimensional hydrocarbon matrix. Biochemistry 31, 279–285 (1992).
Hudson, S. D. et al. Direct visualization of individual cylindrical and spherical supramolecular dendrimers. Science 278, 449–452 (1997).
Balagurusamy, V. S. K., Ungar, G., Percec, V. & Johansson, G. Rational design of the first spherical supramolecular dendrimers self-organized in a novel thermotropic cubic liquid–crystalline phase and the determination of their shape by X-ray analysis. J. Am. Chem. Soc. 119, 1539–1555 (1997).
Ungar, G., Liu, Y., Zeng, X., Percec, V. & Cho, W.-D. Giant supramolecular liquid crystal lattice. Science 299, 1208–1211 (2003).
Dukeson, D. R. et al. Application of isomorphous replacement in the structure determination of a cubic liquid crystal phase and location of counterions. J. Am. Chem. Soc. 125, 15974–15980 (2003).
Percec, V. et al. Self-assembly of dendronized triphenylenes into helical pyramidal columns and chiral spheres. J. Am. Chem. Soc. 131, 7662–7677 (2009).
Percec, V., Imam, M. R., Peterca, M., Wilson, D. A. & Heiney, P. A. Self-assembly of dendritic crowns into chiral supramolecular spheres. J. Am. Chem. Soc. 131, 1294–1304 (2009).
Lee, S., Bluemle, M. J. & Bates, F. S. Discovery of a Frank–Kasper σ phase in sphere-forming block copolymer melts. Science 330, 349–353 (2010).
Lee, S., Leighton, C. & Bates, F. S. Sphericity and symmetry breaking in the formation of Frank–Kasper phases from one component materials. Proc. Natl Acad. Sci. USA 111, 17723–17731 (2014).
Baez-Cotto, C. M. & Mahanthappa, M. K. Micellar mimicry of intermetallic C14 and C15 laves phases by aqueous lyotropic self-assembly. ACS Nano 12, 3226–3234 (2018).
Kim, S. A., Jeong, K.-J., Yethiraj, A. & Mahanthappa, M. K. Low-symmetry sphere packings of simple surfactant micelles induced by ionic sphericity. Proc. Natl Acad. Sci. USA 114, 4072–4077 (2017).
Huang, M. et al. Selective assemblies of giant tetrahedra via precisely controlled positional interactions. Science 348, 424–428 (2015).
Yue, K. et al. Geometry induced sequence of nanoscale Frank–Kasper and quasicrystal mesophases in giant surfactants. Proc. Natl Acad. Sci. USA 113, 14195–14200 (2016).
Kim, K. et al. Thermal processing of diblock copolymer melts mimics metallurgy. Science 356, 520–523 (2017).
Kim, K. et al. Origins of low-symmetry phases in asymmetric diblock copolymer melts. Proc. Natl Acad. Sci. USA 115, 847–854 (2018).
Zhang, W.-B. et al. Molecular nanoparticles are unique elements for macromolecular science: from ‘nanoatoms’ to giant molecules. Macromolecules 47, 1221–1239 (2014).
Zhang, W. et al. Sequence-mandated, distinct assembly of giant molecules. Angew. Chem. Int. Ed. 56, 15014–15019 (2017).
Reddy, A. et al. Stable Frank–Kasper phases of self-assembled, soft matter spheres. Proc. Natl Acad. Sci. USA 115, 10233–10238 (2018).
Date, R. W. & Bruce, D. W. Shape amphiphiles: mixing rods and disks in liquid crystals. J. Am. Chem. Soc. 125, 9012–9013 (2003).
Glotzer, S. C. Some assembly required. Science 306, 419–420 (2004).
Osawa, T. et al. Wide-range 2D lattice correlation unveiled for columnarly assembled triphenylene hexacarboxylic esters. Angew. Chem. Int. Ed. 51, 7990–7993 (2012).
Vorländer, D. Die Erforschung der molekularen Gestalt mit Hilfe der kristallinischen Flüssigkeiten. Z. Phys. Chem. 105, 211–254 (1923).
Wilson, C. G., Thomas, D. K. & Spooner, F. J. The crystal structure of Zr4Al3. Acta Crystallogr. 13, 56–57 (1960).
Ishimasa, T. & Fukano, Y. Planar defect in A15 structure observed in Cr–Si fine particles. Jpn J. Appl. Phys. 22, 1092–1097 (1983).
Arita, M. & Nishida, I. Defects of A15 small particles in tungsten thin films. Surf. Rev. Lett. 3, 1191–1194 (1996).
Ziherl, P. & Kamien, R. D. Maximizing entropy by minimizing area: towards a new principle of self-organization. J. Phys. Chem. B 105, 10147–10158 (2001).
Acknowledgements
This work was supported by National Science Foundation (DMR-1408872 to S.Z.D.C. and CHE-1808115 to C.W.) and the Program for Guangdong introducing Innovative and Entrepreneurial Teams (no. 2016ZT06C322). This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. T. L. is grateful to the support by the Northern Illinois University start-up funds.
Author information
Authors and Affiliations
Contributions
Z.S., M.H. and S.Z.D.C. designed the project. Z.S., Z.G. and J.H. synthesized and characterized the samples. J.M. and C.W. contributed to the matrix-assisted laser desorption/ionization mass experiments. Z.S., X.F., T.L. and S.S. carried out the SAXS experiments. Z.S. and R.Z. carried out the TEM experiments. Z.S., C.-H.H., W.Z., T.A., S.Z.D.C. and Y.W. analysed the data. Z.S., M.H. and S.Z.D.C. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Materials; Methods; Synthesis and characterization details; Analysis of the structure; Supplementary Figs. 1–9; Supplementary Tables 1–3; Supplementary references.
Rights and permissions
About this article
Cite this article
Su, Z., Hsu, CH., Gong, Z. et al. Identification of a Frank–Kasper Z phase from shape amphiphile self-assembly. Nat. Chem. 11, 899–905 (2019). https://doi.org/10.1038/s41557-019-0330-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0330-x
This article is cited by
-
Self-assembled soft alloy with Frank–Kasper phases beyond metals
Nature Materials (2024)
-
A soft crystalline packing with no metallic analogue
Nature Materials (2024)
-
Topological dual and extended relations between networks of clathrate hydrates and Frank-Kasper phases
Nature Communications (2023)
-
Spherical Packing Superlattices in Self-Assembly of Homogenous Soft Matter: Progresses and Potentials
Chinese Journal of Polymer Science (2023)
-
Complex structures arising from the self-assembly of a simple organic salt
Nature (2021)