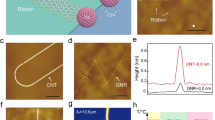
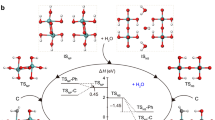
Abstract
Two-dimensional materials show a variety of promising properties, and controlling their growth is an important aspect for practical applications. To this end, active species such as hydrogen and oxygen are commonly introduced into reactors to promote the synthesis of two-dimensional materials with specific characteristics. Here, we demonstrate that fluorine can play a crucial role in tuning the growth kinetics of three representative two-dimensional materials (graphene, hexagonal boron nitride and WS2). When growing graphene by chemical vapour deposition on a copper foil, fluorine released from the decomposition of a metal fluoride placed near the copper foil greatly accelerates the growth of the graphene (up to a rate of ~200 μm s−1). Theoretical calculations show that it does so by promoting decomposition of the methane feedstock, which converts the endothermic growth process to an exothermic one. We further show that the presence of fluorine also accelerates the growth of two-dimensional hexagonal boron nitride and WS2.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information, and also from the authors upon request.
References
Xu, X. Z. et al. Ultrafast growth of single-crystal graphene assisted by a continuous oxygen supply. Nat. Nanotechnol. 11, 930–935 (2016).
Hao, Y. F. et al. The role of surface oxygen in the growth of large single-crystal graphene on copper. Science 342, 720–723 (2013).
Hao, Y. F. et al. Oxygen-activated growth and bandgap tunability of large single-crystal bilayer graphene. Nat. Nanotechnol. 11, 426–431 (2016).
Kiplinger, J. L., Richmond, T. G. & Osterberg, C. E. Activation of carbon fluorine bonds by metal-complexes. Chem. Rev. 94, 373–431 (1994).
Reichenbacher, K., Suss, H. I. & Hulliger, J. Fluorine in crystal engineering—‘the little atom that could’. Chem. Soc. Rev. 34, 22–30 (2005).
Muller, K., Faeh, C. & Diederich, F. Fluorine in pharmaceuticals: looking beyond intuition. Science 317, 1881–1886 (2007).
Purser, S., Moore, P. R., Swallow, S. & Gouverneur, V. Fluorine in medicinal chemistry. Chem. Soc. Rev. 37, 320–330 (2008).
Price, S. C. et al. Fluorine substituted conjugated polymer of medium band gap yields 7% efficiency in polymer–fullerene solar cells. J. Am. Chem. Soc. 133, 4625–4631 (2011).
Furuya, T., Kamlet, A. S. & Ritter, T. Catalysis for fluorination and trifluoromethylation. Nature 473, 470–477 (2011).
Nair, R. R. et al. Fluorographene: a two-dimensional counterpart of teflon. Small 6, 2877–2884 (2010).
Zboril, R. et al. Graphene fluoride: a stable stoichiometric graphene derivative and its chemical conversion to graphene. Small 6, 2885–2891 (2010).
Romero-Aburto, R. et al. Fluorinated graphene oxide; a new multimodal material for biological applications. Adv. Mater. 25, 5632–5637 (2013).
Castro Neto, A. H. et al. The electronic properties of graphene. Rev. Mod. Phys. 81, 109–162 (2009).
Chhowalla, M. et al. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 5, 263–275 (2013).
Giles, A. J. et al. Ultralow-loss polaritons in isotopically pure boron nitride. Nat. Mater. 17, 134–139 (2018).
Kubota, Y., Watanabe, K., Tsuda, O. & Taniguchi, T. Deep ultraviolet light-emitting hexagonal boron nitride synthesized at atmospheric pressure. Science 317, 932–934 (2007).
Novoselov, K. S. et al. A roadmap for graphene. Nature 490, 192–200 (2012).
Wang, Q. H. et al. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 7, 699–712 (2012).
Young, R. J. Two-dimensional nanocrystals: structure, properties and applications. Arab. J. Sci. Eng. 38, 1289–1304 (2013).
Fiori, G. et al. Electronics based on two-dimensional materials. Nat. Nanotechnol. 9, 768–779 (2014).
Cho, S. et al. Phase patterning for ohmic homojunction contact in MoTe2. Science 349, 625–628 (2015).
Keum, D. H. et al. Bandgap opening in few-layered monoclinic MoTe2. Nat. Phys. 11, 482–486 (2015).
Yang, H., Kim, S. W., Chhowalla, M. & Lee, Y. H. Structural and quantum-state phase transitions in van der Waals layered materials. Nat. Phys. 13, 1232–1232 (2017).
Dai, B. Y. et al. Rational design of a binary metal alloy for chemical vapour deposition growth of uniform single-layer graphene. Nat. Commun. 2, 522 (2011).
Gao, L. et al. Repeated growth and bubbling transfer of graphene with millimetre-size single-crystal grains using platinum. Nat. Commun. 3, 699 (2012).
Geng, D. C. et al. Uniform hexagonal graphene flakes and films grown on liquid copper surface. Proc. Natl Acad. Sci. USA 109, 7992–7996 (2012).
Yan, Z. et al. Toward the synthesis of wafer-scale single-crystal graphene on copper foils. ACS Nano 6, 9110–9117 (2012).
Zhou, H. L. et al. Chemical vapour deposition growth of large single crystals of monolayer and bilayer graphene. Nat. Commun. 4, 2096 (2013).
Lee, J. H. et al. Wafer-scale growth of single-crystal monolayer graphene on reusable hydrogen-terminated germanium. Science 344, 286–289 (2014).
Gao, L. B. et al. Face-to-face transfer of wafer-scale graphene films. Nature 505, 190–194 (2014).
Babenko, V. et al. Rapid epitaxy-free graphene synthesis on silicidated polycrystalline platinum. Nat. Commun. 6, 7536 (2015).
Nguyen, V. L. et al. Seamless stitching of graphene domains on polished copper (111) foil. Adv. Mater. 27, 1376–1382 (2015).
Wu, T. R. et al. Fast growth of inch-sized single-crystalline graphene from a controlled single nucleus on Cu–Ni alloys. Nat. Mater. 15, 43–47 (2016).
Wang, Z. J. et al. Stacking sequence and interlayer coupling in few-layer graphene revealed by in situ imaging. Nat. Commun. 7, 13256 (2016).
Wang, H. et al. Surface monocrystallization of copper foil for fast growth of large single-crystal graphene under free molecular flow. Adv. Mater. 28, 8968–8974 (2016).
Zhang, Z. H. et al. The way towards ultrafast growth of single-crystal graphene on copper. Adv. Sci. 4, 1700087 (2017).
Xu, X. Z. et al. Ultrafast epitaxial growth of metre-sized single-crystal graphene on industrial Cu foil. Sci. Bull. 62, 1074–1080 (2017).
Vlassiouk, I. V. et al. Evolutionary selection growth of two-dimensional materials on polycrystalline substrates. Nat. Mater. 17, 318–322 (2018).
Liu, Z. et al. In-plane heterostructures of graphene and hexagonal boron nitride with controlled domain sizes. Nat. Nanotechnol. 8, 119–124 (2013).
Liu, L. et al. Heteroepitaxial growth of two-dimensional hexagonal boron nitride templated by graphene edges. Science 343, 163–167 (2014).
Duan, X. D. et al. Lateral epitaxial growth of two-dimensional layered semiconductor heterojunctions. Nat. Nanotechnol. 9, 1024–1030 (2014).
Yin, J. et al. Large single-crystal hexagonal boron nitride monolayer domains with controlled morphology and straight merging boundaries. Small 11, 4497–4502 (2015).
Kang, K. et al. High-mobility three-atom-thick semiconducting films with wafer-scale homogeneity. Nature 520, 656–660 (2015).
Lu, G. Y. et al. Synthesis of large single-crystal hexagonal boron nitride grains on Cu–Ni alloy. Nat. Commun. 6, 6160 (2015).
Gao, Y. et al. Ultrafast growth of high-quality monolayer WSe2 on Au. Adv. Mater. 29, 1700990 (2017).
Zhang, Z. W. et al. Robust epitaxial growth of two-dimensional heterostructures, multiheterostructures and superlattices. Science 357, 788–792 (2017).
Wang, H. et al. High-quality monolayer superconductor NbSe2 grown by chemical vapour deposition. Nat. Commun. 8, 394 (2017).
Dong, J., Zhang, L. & Ding, F. Kinetics of graphene and 2D materials growth. Adv. Mater. 31, 1801583 (2019).
Li, X. S., Cai, W. W., Colombo, L. & Ruoff, R. S. Evolution of graphene growth on Ni and Cu by carbon isotope labeling. Nano Lett. 9, 4268–4272 (2009).
Moss, J. H., Ottie, R. & Wilford, J. B. The fluorination of methane and related compounds by copper(ii) fluoride and other metal fluorides. J. Fluor. Chem. 6, 393–416 (1975).
Kaneko, C. et al. Reaction of methane with molecular fluorine: an ab initio MO study. Chem. Pharm. Bull. 42, 745–747 (1994).
Zhou, J. D. et al. A library of atomically thin metal chalcogenides. Nature 556, 355–359 (2018).
Kresse, G. & Furthmuller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comp. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Hafner, J. Ab-initio molecular-dynamics for open-shell transition-metals. Phys. Rev. B. 48, 13115–13118 (1993).
Wu, P. et al. Carbon dimers as the dominant feeding species in epitaxial growth and morphological phase transition of graphene on different Cu substrates. Phys. Rev. Lett. 114, 216102 (2015).
Henkelman, G., Uberuaga, B. P. & Jonsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B. 59, 1758–1775 (1999).
Acknowledgements
This work was supported by the National Key R&D Program of China (2016YFA0300903, 2016YFA0300804 and 2015CB358600), the NSFC (51522201, 11474006 and 51722204), the National Equipment Program of China (ZDYZ2015-1), Beijing Municipal Science & Technology Commission (Z181100004218006), Beijing Graphene Innovation Program (Z181100004818003 and Z161100002116028), the Bureau of Industry and Information Technology of Shenzhen (Graphene Platform contract no. 201901161512), the Science–Technology and Innovation Commission of Shenzhen Municipality (ZDSYS20170303165926217 and JCYJ20170412152620376), the Economic–Trade and Information Commission of Shenzhen Municipality, Guangdong Innovative and Entrepreneurial Research Team Program (2016ZT06D348), the National Postdoctoral Program for Innovative Talents (BX201700014 and BX20190016), the Fundamental Research Funds for the Central Universities (ZYGX2016Z004), the Institute for Basic Science (IBS-R019-D1) of South Korea and the Outstanding Research Fund (1.180066.01) of UNIST (Ulsan National Institute of Science & Technology).
Author information
Authors and Affiliations
Contributions
K.L., E.W., D.Y. and C.L. conceived the experiment. K.L., F.D. and J.X. supervised the project. C.L., X.X. and M.W. conducted the growth experiment. C.L. performed Raman and XPS experiments. X.X. and Y.J. performed LEED and STM experiments. R.Q. and P.G. conducted the TEM experiments. J.N. and X.W. performed the electrical measurements. F.D. and L.Q. performed theoretical calculations. All of the authors discussed the results and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–18; Supplementary Tables 1–2; Supplementary Note
Rights and permissions
About this article
Cite this article
Liu, C., Xu, X., Qiu, L. et al. Kinetic modulation of graphene growth by fluorine through spatially confined decomposition of metal fluorides. Nat. Chem. 11, 730–736 (2019). https://doi.org/10.1038/s41557-019-0290-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0290-1
This article is cited by
-
Growth of millimeter-sized 2D metal iodide crystals induced by ion-specific preference at water-air interfaces
Nature Communications (2024)
-
General low-temperature growth of two-dimensional nanosheets from layered and nonlayered materials
Nature Communications (2023)
-
Controllable growth of two-dimensional quantum materials
Science China Physics, Mechanics & Astronomy (2023)
-
Fast scanning growth of high-quality graphene films on Cu foils fueled by dimeric carbon precursor
Nano Research (2023)
-
Recent developments in CVD growth and applications of 2D transition metal dichalcogenides
Frontiers of Physics (2023)