Abstract
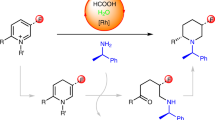
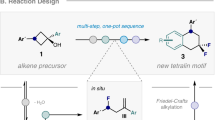
Piperidines and fluorine substituents are both independently indispensable components in pharmaceuticals, agrochemicals and materials. Logically, the incorporation of fluorine atoms into piperidine scaffolds is therefore an area of tremendous potential. However, synthetic approaches towards the formation of these architectures are often impractical. The diastereoselective synthesis of substituted monofluorinated piperidines often requires substrates with pre-defined stereochemistry. That of multifluorinated piperidines is even more challenging, and often needs to be carried out in multistep syntheses. In this report, we describe a straightforward process for the one-pot rhodium-catalysed dearomatization–hydrogenation of fluoropyridine precursors. This strategy enables the formation of a plethora of substituted all-cis-(multi)fluorinated piperidines in a highly diastereoselective fashion through pyridine dearomatization followed by complete saturation of the resulting intermediates by hydrogenation. Fluorinated piperidines with defined axial/equatorial orientation of fluorine substituents were successfully applied in the preparation of commercial drugs analogues. Additionally, fluorinated PipPhos as well as fluorinated ionic liquids were obtained by this dearomatization–hydrogenation process.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre under deposition numbers CCDC 1845054 (29) and 1845055 (60). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within the Article and its Supplementary Information, or from the corresponding author upon reasonable request.
References
Trost, B. M. Selectivity: a key to synthetic efficiency. Science 219, 245–250 (1983).
Baran, P. S., Maimone, T. J. & Richter, J. M. Total synthesis of marine natural products without using protecting groups. Nature 446, 404–408 (2007).
Young, I. S. & Baran, P. S. Protecting-group-free synthesis as an opportunity for invention. Nat. Chem. 1, 193–205 (2009).
Wender, P. A. & Miller, B. L. Synthesis at the molecular frontier. Nature 460, 197–201 (2009).
Zhao, D., Candish, L., Paul, D. & Glorius, F. N-Heterocyclic carbenes in asymmetric hydrogenation. ACS Catal. 6, 5978–5988 (2016).
Wei, Y., Rao, B., Cong, X. & Zeng, X. Highly selective hydrogenation of aromatic ketones and phenols enabled by cyclic (amino)(alkyl)carbene rhodium complexes. J. Am. Chem. Soc. 137, 9250–9253 (2015).
Wiesenfeldt, M. P., Nairoukh, Z., Li, W. & Glorius, F. Hydrogenation of fluoroarenes: direct access to all-cis-(multi)fluorinated cycloalkanes. Science 357, 908–912 (2017).
Wiesenfeldt, M. P., Knecht, T., Schlepphorst, C. & Glorius, F. Silylarene hydrogenation: a strategic approach enabling direct access to versatile silylated saturated carbo- and heterocycles. Angew. Chem. Int. Ed. 57, 8297–8300 (2018).
O’Hagan, D. Pyrrole, pyrrolidine, pyridine, piperidine and tropane alkaloids. Nat. Prod. Rep. 17, 435–446 (2000).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among US FDA approved pharmaceuticals. J. Med. Chem. 57, 10257–10274 (2014).
Müller, K., Faeh, C. & Diederich, F. Fluorine in pharmaceuticals: Looking beyond intuition. Science 317, 1881–1886 (2007).
Shah, P. & Westwell, A. D. The role of fluorine in medicinal chemistry. J. Enzyme Inhib. Med. Chem. 22, 527–540 (2007).
O’Hagan, D. Understanding organofluorine chemistry. An introduction to the C–F bond. Chem. Soc. Rev. 37, 308–319 (2008).
Thiehoff, C., Rey, Y. P. & Gilmour, R. The fluorine gauche effect: a brief history. Isr. J. Chem. 57, 92–100 (2017).
Wang, J. et al. Fluorine in pharmaceutical industry: fluorine-containing drugs introduced to the market in the last decade (2001−2011). Chem. Rev. 114, 2432–2506 (2014).
Gillis, E. P., Eastman, K. J., Hill, M. D., Donnelly, D. J. & Meanwell, N. A. Applications of fluorine in medicinal chemistry. J. Med. Chem. 58, 8315–8359 (2015).
Li, X. et al. Process development for scale-up of a novel 3,5-substituted thiazolidine-2,4-dione compound as a potent inhibitor for estrogen-related receptor 1. Org. Process Res. Dev. 18, 321–330 (2014).
Goldberg, N. W., Shen, X., Li, J. & Ritter, T. AlkylFluor: deoxyfluorination of alcohols. Org. Lett. 18, 6102–6104 (2016).
Liu, W. et al. Oxidative aliphatic C–H fluorination with fluoride ion catalyzed by a manganese porphyrin. Science 337, 1322–1325 (2012).
Ventre, S., Petronijevic, F. R. & MacMillan, D. W. C. Decarboxylative fluorination of aliphatic carboxylic acids via photoredox catalysis. J. Am. Chem. Soc. 137, 5654–5657 (2015).
Snyder, J. P., Chandrakumar, N. S., Sato, H. & Lankin, D. C. The unexpected diaxial orientation of cis-3,5-difluoropiperidine in water: a potent CF- - -NH charge-dipole effect. J. Am. Chem. Soc. 122, 544–545 (2000).
Glorius, F., Spielkamp, N., Holle, S., Goddard, R. & Lehmann, C. W. Efficient asymmetric hydrogenation of pyridines. Angew. Chem. Int. Ed. 43, 2850–2852 (2004).
Zhou, Y.-G. Asymmetric hydrogenation of heteroaromatic compounds. Acc. Chem. Res. 40, 1357–1366 (2007).
Whittlesey, M. K. & Peris, E. Catalytic hydrodefluorination with late transition metal complexes. ACS Catal. 4, 3152–3159 (2014).
Dyson, P. J. Arene hydrogenation by homogeneous catalysts: fact or fiction? Dalton Trans. 2003, 2964–2974 (2003).
Park, S. & Chang, S. Catalytic dearomatization of N-heteroarenes with silicon and boron compounds. Angew. Chem. Int. Ed. 56, 7720–7738 (2017).
Oshima, K., Ohmura, T. & Suginome, M. Regioselective synthesis of 1,2-dihydropyridines by rhodium-catalyzed hydroboration of pyridines. J. Am. Chem. Soc. 134, 3699–3702 (2012).
Jazzar, R. et al. Intramolecular ‘hydroiminiumation’ of alkenes: application to the synthesis of conjugate acids of cyclic alkyl amino carbenes (CAACs). Angew. Chem. Int. Ed. 46, 2899–2902 (2007).
Lankin, D. C., Chandrakumar, N. S., Rao, S. N., Spangler, D. P. & Snyder, J. P. Protonated 3-fluoropiperidines: an unusual fluoro directing effect and a test for quantitative theories of solvation. J. Am. Chem. Soc. 115, 3356–3357 (1993).
Silla, J. M. et al. Gauche preference of β-fluoroalkyl ammonium salts. J. Phys. Chem. A 118, 503–507 (2014).
Pereiro, A. B. et al. Fluorinated ionic liquids: properties and applications. ACS Sustain. Chem. Eng. 1, 427–439 (2013).
Acknowledgements
The authors acknowledge financial support from the Hans-Jensen-Minerva Foundation (Z.N.), the Deutsche Forschungsgemeinschaft IRTG 2027 (M.W.) and the European Research Council (ERC Advanced Grant Agreement no. 788558). The authors thank M.P. Wiesenfeldt, M. Teders, M.J. James and M. van Gemmeren for helpful discussions. C.G. Daniliuc is acknowledged for X-ray crystallographic analysis. 1-(cis-3,5-difluoropiperidin-1-yl)-2,2,2-trifluoroethan-1-one (3) is available from Sigma-Aldrich (product no. 903817).
Author information
Authors and Affiliations
Contributions
Z.N., M.W., C.S. and F.G. designed, performed and analysed experiments. K.B. performed and analysed NMR data. Z.N. and F.G. prepared the manuscript with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
Z.N., C.S. and F.G. are inventors on German patent application DE 10 2018 104 201.9 held by WWU Muenster, which covers the DAH process for the synthesis of all-cis-(multi)fluorinated aliphatic heterocycles.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Detailed experimental procedures, extensive optimization data, comprehensive NMR analysis and MS data of all new compounds, crystallographic reports and mechanistic studies
Crystallographic data
CIF for compound 29; CDCC reference 1845054
Crystallographic data
CIF for compound 60; CDCC reference 1845055
Rights and permissions
About this article
Cite this article
Nairoukh, Z., Wollenburg, M., Schlepphorst, C. et al. The formation of all-cis-(multi)fluorinated piperidines by a dearomatization–hydrogenation process. Nature Chem 11, 264–270 (2019). https://doi.org/10.1038/s41557-018-0197-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-018-0197-2
This article is cited by
-
Synthesis of polysubstituted azepanes by dearomative ring expansion of nitroarenes
Nature Chemistry (2024)
-
Construction and Characterization of Magnetic Fe3O4 Nanoparticles Supported Zn Complex: Research on Multicomponent Synthesis of Highly Functionalized Piperidines
Catalysis Letters (2024)
-
Copper complex immobilized on Fe3O4 nanoparticles: a novel and recoverable heterogenous catalyst for synthesis of heterocycles
Research on Chemical Intermediates (2024)
-
Chromium-catalyzed stereodivergent E- and Z-selective alkyne hydrogenation controlled by cyclic (alkyl)(amino)carbene ligands
Nature Communications (2023)
-
Synthesis of chiral piperidines from pyridinium salts via rhodium-catalysed transfer hydrogenation
Nature Catalysis (2022)