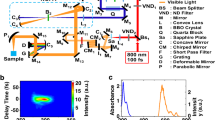
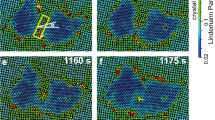
Abstract
Control over the nucleation of new phases is highly desirable but elusive. Even though there is a long history of crystallization engineering by varying physicochemical parameters, controlling which polymorph crystallizes or whether a molecule crystallizes or forms an amorphous precipitate is still a poorly understood practice. Although there are now numerous examples of control using laser-induced nucleation, the absence of physical understanding is preventing progress. Here we show that the proximity of a liquid–liquid critical point or the corresponding binodal line can be used by a laser-tweezing potential to induce concentration gradients. A simple theoretical model shows that the stored electromagnetic energy of the laser beam produces a free-energy potential that forces phase separation or triggers the nucleation of a new phase. Experiments in a liquid mixture using a low-power laser diode confirm the effect. Phase separation and nucleation using a laser-tweezing potential explains the physics behind non-photochemical laser-induced nucleation and suggests new ways of manipulating matter.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bučar, D.-K., Lancaster, R. W. & Bernstein, J. Disappearing polymorphs revisited. Angew. Chem. Int. Ed. 54, 6972–6993 (2015).
Garetz, B., Aber, J., Goddard, N., Young, R. & Myerson, A. Nonphotochemical, polarization-dependent, laser-induced nucleation in supersaturated aqueous urea solutions. Phys. Rev. Lett. 77, 3475–3476 (1996).
Garetz, B., Matic, J. & Myerson, A. Polarization switching of crystal structure in the nonphotochemical light-induced nucleation of supersaturated aqueous glycine solutions. Phys. Rev. Lett. 89, 175501 (2002).
Ward, M. R., Mchugh, S. & Alexander, A. J. Non-photochemical laser-induced nucleation of supercooled glacial acetic acid. Phys. Chem. Chem. Phys. 14, 90–93 (2012).
Iefuji, N. et al. Laser-induced nucleation in protein crystallization: local increase in protein concentration induced by femtosecond laser irradiation. J. Cryst. Growth 318, 741–744 (2011).
Usman, A., Uwada, T. & Masuhara, H. Optical reorientation and trapping of nematic liquid crystals leading to the formation of micrometer-sized domain. J. Phys. Chem. C. 115, 11906–11913 (2011).
Kosa, T. et al. Light-induced liquid crystallinity. Nature 485, 347–349 (2012).
Knott, B. C., Larue, J. L., Wodtke, A. M., Doherty, M. F. & Peters, B. Communication: bubbles, crystals, and laser-induced nucleation. J. Chem. Phys. 134, 171102 (2011).
Knott, B. C., Doherty, M. F. & Peters, B. A simulation test of the optical Kerr mechanism for laser-induced nucleation. J. Chem. Phys. 134, 154501 (2011).
Liu, Y., Van Den Berg, M. H. & Alexander, A. J. Supersaturation dependence of glycine polymorphism using laser-induced nucleation, sonocrystallization and nucleation by mechanical shock. Phys. Chem. Chem. Phys. 19, 19386–19392 (2017).
Yuyama, K.-I., Rungsimanon, T., Sugiyama, T. & Masuhara, H. Selective fabrication of α- and γ-polymorphs of glycine by intense polarized continuous wave laser beams. Cryst. Growth Des. 12, 2427–2434 (2012).
Tenwolde, P. & Frenkel, D. Enhancement of protein crystal nucleation by critical density fluctuations. Science 277, 1975–1978 (1997).
Gebauer, D., Voelkel, A. & Coelfen, H. Stable prenucleation calcium carbonate clusters. Science 322, 1819–1822 (2008).
Gebauer, D., Kellermeier, M., Gale, J. D., Bergström, L. & Cölfen, H. Pre-nucleation clusters as solute precursors in crystallisation. Chem. Soc. Rev. 43, 2348–2371 (2014).
Radu, M. & Kremer, K. Enhanced crystal growth in binary Lennard–Jones mixtures. Phys. Rev. Lett. 118, 055702–055706 (2017).
Wedekind, J. et al. Optimization of crystal nucleation close to a metastable fluid–fluid phase transition. Sci. Rep. 5, 11260 (2015).
Jones, R. A. L. Soft Condensed Matter (Oxford Univ. Press, Oxford, 2002).
Bowman, R. W. & Padgett, M. J. Optical trapping and binding. Rep. Prog. Phys. 76, 026401 (2013).
Yuyama, K.-I., George, J., Thomas, K. G., Sugiyama, T. & Masuhara, H. Two-dimensional growth rate control of l-phenylalanine crystal by laser trapping in unsaturated aqueous solution. Cryst. Growth Des. 16, 953–960 (2016).
Méndez-Castro, P., Troncoso, J., Peleteiro, J. & Romaní, L. Heat capacity singularity of binary liquid mixtures at the liquid–liquid critical point. Phys. Rev. E 88, 042107 (2013).
Gao, P., Yao, B., Harder, I., Lindlein, N. & Torcal-Milla, F. J. Phase-shifting Zernike phase contrast microscopy for quantitative phase measurement. Opt. Lett. 36, 4305–4307 (2011).
Liu, Y., Ward, M. R. & Alexander, A. J. Polarization independence of laser-induced nucleation in supersaturated aqueous urea solutions. Phys. Chem. Chem. Phys. 19, 3464–3467 (2017).
Duffus, C., Camp, P. J. & Alexander, A. J. Spatial control of crystal nucleation in agarose gel. J. Am. Chem. Soc. 131, 11676–11677 (2009).
Gutierrez, J. M. P., Hinkley, T., Taylor, J. W., Yanev, K. & Cronin, L. Evolution of oil droplets in a chemorobotic platform. Nat. Commun. 5, 5571 (2014).
Peterman, E. J. G., Gittes, F. & Schmidt, C. F. Laser-induced heating in optical traps. Biophys. J. 84, 1308–1316 (2003).
Hofkens, J., Hotta, J., Sasaki, K., Masuhara, H. & Iwai, K. Molecular assembling by the radiation pressure of a focused laser beam: poly(-isopropylacrylamide) in aqueous solution. Langmuir 13, 414–419 (1997).
Oana, H. et al. Spontaneous formation of giant unilamellar vesicles from microdroplets of a polyion complex by thermally induced phase separation. Angew. Chem. Int. Ed. 48, 4613–4616 (2009).
Kitamura, N., Yamada, M., Ishizaka, S. & Konno, K. Laser-induced liquid-to-droplet extraction of chlorophenol: photothermal phase separation of aqueous triethylamine solutions. Anal. Chem. 77, 6055–6061 (2005).
Bunkin, N. F., Lobeev, A. V., Lyakhov, G. A. & Svirko, Y. P. Local light-induced phase separation of binary liquid solutions. Quantum Electron. 26, 60–64 (1996).
Osborne, M. A., Balasubramanian, S., Furey, W. S. & Klenerman, D. Optically biased diffusion of single molecules studied by confocal fluorescence microscopy. J. Phys. Chem. B 102, 3160–3167 (1998).
Yuyama, K.-I., Sugiyama, T. & Masuhara, H. Laser trapping and crystallization dynamics of l-phenylalanine at solution surface. J. Phys. Chem. Lett. 4, 2436–2440 (2013).
Wallace, A. F. et al. Microscopic evidence for liquid–liquid separation in supersaturated CaCO3 solutions. Science 341, 885–889 (2013).
De Yoreo, J. J. et al. Crystallization by particle attachment in synthetic, biogenic, and geologic environments. Science 349, aaa6760 (2015).
Masuhara, H., Sugiyama, T., Yuyama, K.-I. & Usman, A. Optical trapping assembling of clusters and nanoparticles in solution by CW and femtosecond lasers. Opt. Rev. 22, 143–148 (2015).
Mosses, J., Syme, C. D. & Wynne, K. Order parameter of the liquid–liquid transition in a molecular liquid. J. Phys. Chem. Lett. 6, 38–43 (2015).
Syme, C. D. et al. Frustration of crystallisation by a liquid–crystal phase. Sci. Rep. 7, 42439 (2017).
Acknowledgements
We thank the Engineering and Physical Sciences Research Council (EPSRC) for support through grants EP/J004790/1, EP/J009733/1 and EP/N007417/1. We gratefully acknowledge discussions in 2010 with C. Bain that planted the seed for this work.
Author information
Authors and Affiliations
Contributions
The experiments and data analysis were conducted by F.W. Theory and simulations were conducted by K.W. Both authors contributed to writing the paper. K.W. conceived the overall project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6, Supplementary Results
Rights and permissions
About this article
Cite this article
Walton, F., Wynne, K. Control over phase separation and nucleation using a laser-tweezing potential. Nature Chem 10, 506–510 (2018). https://doi.org/10.1038/s41557-018-0009-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-018-0009-8
This article is cited by
-
Mixed-halide perovskite synthesis by chemical reaction and crystal nucleation under an optical potential
NPG Asia Materials (2019)