Abstract
Despite advances in the differentiation of insulin-producing cells from human embryonic stem cells, the generation of mature functional β cells in vitro has remained elusive. To accomplish this goal, we have developed cell culture conditions to closely mimic events occurring during pancreatic islet organogenesis and β cell maturation. In particular, we have focused on recapitulating endocrine cell clustering by isolating and reaggregating immature β-like cells to form islet-sized enriched β-clusters (eBCs). eBCs display physiological properties analogous to primary human β cells, including robust dynamic insulin secretion, increased calcium signalling in response to secretagogues, and improved mitochondrial energization. Notably, endocrine cell clustering induces metabolic maturation by driving mitochondrial oxidative respiration, a process central to stimulus–secretion coupling in mature β cells. eBCs display glucose-stimulated insulin secretion as early as three days after transplantation in mice. In summary, replicating aspects of endocrine cell clustering permits the generation of stem-cell-derived β cells that resemble their endogenous counterparts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Materials and protocols that were used in this study are available from the corresponding author upon request. RNA-seq data that support the findings of this study have been deposited in the Gene Expression Omnibus (GEO) under accession code GSE109795. Source data for Figs. 1–3 and 5–7 and Supplementary Figs. 1, 3, 4 and 6 have been provided in Supplementary Table 6. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
Change history
26 March 2019
In the version of this article originally published, the Gene Expression Omnibus (GEO) accession number listed in the data availability section was incorrectly given as GSE10979 instead of GSE109795. The sentence should read “RNA-seq data that support the findings of this study have been deposited in the Gene Expression Omnibus (GEO) under accession code GSE109795,” and the code should link to https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE109795. The error has been corrected in the HTML and PDF versions of the paper.
References
Barton, F. B. et al. Improvement in outcomes of clinical islet transplantation: 1999–2010. Diabetes Care 35, 1436–1445 (2012).
Pagliuca, F. W. et al. Generation of functional human pancreatic β cells in vitro. Cell. 159, 428–439 (2014).
Rezania, A. et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat. Biotech. 32, 1121–1133 (2014).
Russ, H. A. et al. Controlled induction of human pancreatic progenitors produces functional β-like cells in vitro. EMBO J. 34, 1759–1772 (2015).
Yoshihara, E. et al. ERRɣ is required for the metabolic maturation of therapeutically functional glucose-responsive β cells. Cell. Metab. 23, 622–634 (2016).
Blum, B. et al. Functional β-cell maturation is marked by an increased glucose threshold and by expression of urocortin 3. Nat. Biotech. 30, 261–264 (2012).
Aguayo-Mazzucato, C. et al. Mafa expression enhances glucose-responsive insulin secretion in neonatal rat β cells. Diabetologia 54, 583–593 (2011).
Jermendy, A. et al. Rat neonatal β cells lack the specialised metabolic phenotype of mature β cells. Diabetologia 54, 594–604 (2011).
Dhawan, S. et al. DNA methylation directs functional maturation of pancreatic β cells. J. Clin. Invest. 125, 2851–2860 (2015).
Arda, H. E. et al. Age-dependent pancreatic gene regulation reveals mechanisms governing human β cell function. Cell. Metab. 23, 909–920 (2016).
Gregg, B. E. et al. Formation of a human β-cell population within pancreatic islets is set early in life. J. Clin. Endocrinol. Metab. 97, 3197–3206 (2012).
Gu, C. et al. Pancreatic β cells require neurod to achieve and maintain functional maturity. Cell. Metab. 11, 298–310 (2010).
Gosmain, Y. et al. Pax6 is crucial for β-cell function, insulin biosynthesis, and glucose-induced insulin secretion. Mol. Endocrinol. 26, 696–709 (2012).
Dai, C. et al. Islet-enriched gene expression and glucose-induced insulin secretion in human and mouse islets. Diabetologia 55, 707–718 (2012).
Scoville, D. W. et al. MLL3 and MLL4 methyltransferases bind to the mafa and mafb transcription factors to regulate islet β-cell function. Diabetes 64, 3772–3783 (2015).
Lemaire, K., Thorrez, L. & Schuit, F. Disallowed and allowed gene expression: two faces of mature islet beta cells. Annu. Rev. Nutr. 36, 45–71 (2016).
Nair, G. & Hebrok, M. Islet formation in mice and men: lessons for the generation of functional insulin-producing β-cells from human pluripotent stem cells. Curr. Op. Genet. Dev. 32, 171–180 (2015).
Jeon, J. et al. Endocrine cell clustering during human pancreas development. J. Histochem. Cytochem. 57, 811–824 (2009).
Rahier, J., Wallon, J. & Henquin, J. C. Cell populations in the endocrine pancreas of human neonates and infants. Diabetologia 20, 540–546 (1981).
Borden, P. et al. Sympathetic innervation during development is necessary for pancreatic islet architecture and functional maturation. Cell Rep. 4, 287–301 (2013).
Pisania, A. et al. Quantitative analysis of cell composition and purity of human pancreatic islet preparations. Lab. Invest. 90, 1661–1675 (2010).
Taylor, B. L., Liu, F.-F. & Sander, M. Nkx6.1 is essential for maintaining the functional state of pancreatic β cells. Cell Rep. 4, 1262–1275 (2013).
Du, A. et al. Islet-1 is required for the maturation, proliferation, and survival of the endocrine pancreas. Diabetes 58, 2059–2069 (2009).
Portela-Gomes, G. M. et al. The importance of chromogranin A in the development and function of endocrine pancreas. Regul. Pept. 151, 19–25 (2008).
Jacovetti, C. et al. Postnatal β-cell maturation is associated with islet-specific microRNA changes induced by nutrient shifts at weaning. Nat. Commun. 6, 8084 (2015).
Stolovich-Rain, M. et al. Weaning triggers a maturation step of pancreatic β cells. Dev. Cell. 32, 535–545 (2015).
Huang, C. et al. Synaptotagmin 4 regulates pancreatic β cell maturation by modulating the Ca2+sensitivity of insulin secretion vesicles. Dev. Cell. 45, 347–361.e5 (2018).
Qiu, W.-L. et al. Deciphering pancreatic islet β cell and ɑ cell maturation pathways and characteristic features at the single-cell level. Cell. Metab. 25, 1194–1205 (2017).
Hrvatin, S. et al. Differentiated human stem cells resemble fetal, not adult, β cells. Proc. Natl Acad. Sci. USA 111, 3038–3043 (2014).
Dorrell, C. et al. Transcriptomes of the major human pancreatic cell types. Diabetologia 54, 2832 (2011).
Cogliati, S., Enriquez, J. A. & Scorrano, L. Mitochondrial cristae: where beauty meets functionality. Trends. Biochem. Sci. 41, 261–2273 (2016).
Lehmann, R. et al. Superiority of small islets in human islet transplantation. Diabetes 56, 594–603 (2007).
Riedel, M. J. et al. Immunohistochemical characterisation of cells co-producing insulin and glucagon in the developing human pancreas. Diabetologia 55, 372–381 (2012).
Sosa-Pineda, B. et al. The Pax4 gene is essential for differentiation of insulin-producing β cells in the mammalian pancreas. Nature 386, 399 (1997).
Hauge-Evans, A. C. et al. Pancreatic beta-cell-to-beta-cell interactions are required for integrated responses to nutrient stimuli: enhanced Ca2+and insulin secretory responses of MIN6 pseudoislets. Diabetes 48, 1402 (1999).
Luther, M. J. et al. MIN6 β-cell–β-cell interactions influence insulin secretory responses to nutrients and non-nutrients. Biochem. Biophys. Res. Commun. 343, 99–104 (2006).
Persaud, S. et al. Pseudoislets as primary islet replacements for research: report on a symposium at King’s College London. Islets 2, 236–239 (2010).
Henquin, J.-C. & Nenquin, M. Dynamics and regulation of insulin secretion in pancreatic islets from normal young children. PLoS ONE 11, e0165961 (2016).
Hawdon, J. M. et al. The role of pancreatic insulin secretion in neonatal glucoregulation. I. Healthy term and preterm infants. Arch. Dis. Child 68, 274–279 (1993).
Kaye, R. et al. The response of blood glucose, ketones, and plasma nonesterified fatty acids to fasting and epinephrine injection in infants and children. J. Pediatr. 59, 836–847 (1961).
Trapnell, C. et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 7, 562–578 (2012).
Nair, G. G., Holger, R. & Hebrok, M. Rapid generation of mature functional pancreatic islet-beta cells from human pluripotent stem cells. Prot. Exchange https://doi.org/10.1038/protex.2018.140 (2019).
Subramanian, A. et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proceedings of the National Academy of Sciences 102, 15545–15550 (2005).
Adewola, A. F. et al. Microfluidic perifusion and imaging device for multi-parametric islet function assessment. Biomed. Microdevices. 12, 409–417 (2010).
Acknowledgements
We would like to thank the members of the Hebrok laboratory for advice and insightful comments during the preparation of the manuscript. G.G.N. was supported by a Kraft Family Fellowship and a JDRF postdoctoral fellowship (1-PNF-2016-320-S-B). Work in the laboratory of M.H. was supported by grants from the NIH (DK105831, DK108666). Imaging and flow cytometry experiments were supported by resources from the UCSF Diabetes and Endocrinology Research Center (DRC) and UCSF Flow Cytometry Core. Image acquisition was supported by the University of California, San Francisco Diabetes and Endocrinology Research Center (DRC) microscopy core P30 DK63720.
Author information
Authors and Affiliations
Contributions
Conception and study design: G.G.N. and M.H. Execution of experiments: G.G.N., J.S.L., H.A.R., S.T., M.S., R.C., C.J., M.L., Y.X., Y.W., V.Q.N. and G.S. Data analysis and interpretation: G.G.N., J.S.L., H.A.R., S.T., R.C., Y.X., Y.W., S.G., A.B. and M.H. Manuscript writing: G.G.N., J.S.L., S.P. and M.H. Final approval of manuscript: G.G.N., J.S.L., H.A.R., S.T., M.S., R.C., M.L., C.J., Y.X., Y.W., S.G., S.P., V.Q.N., G.S., J.O., A.B. and M.H.
Corresponding author
Ethics declarations
Competing interests
M.H. is affiliated with Semma Therapeutics (consultant, SAB member and stock holder) and Encellin Inc. (SAB member and stock holder). He holds stocks from Viacyte Inc.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Quantitative and temporal analysis of the appearance of key β cell and endocrine markers over the course of endocrine differentiation (d11-d27).
(a) Representative flow cytometry plots of cell populations co-stained with C-peptide and markers for the endocrine lineage, such as Chromogranin A, NEUROD1, NKX2.2, ISL1, and PAX6, as well as β cell-enriched markers including PDX1 and NKX6.1. Also, note the low numbers of α and δ cells that co-express C-peptide and Glucagon or Somatostatin during the differentiation process. These experiments were repeated with ten independent hPSC differentiations yielding similar results (b) Quantification of median fluorescence intensity of C-peptide+ cells of eBCs and d20 immature β-like cells from flow cytometry data. Data are presented as median ±SEM. eBC-β, n = 12 independent samples; d20 immature β-like, n = 8 independent samples ****P = <0.0001 determined by two-tailed unpaired t-test with Welch’s correction. Refer Supplementary Table 6 for source data.
Supplementary Figure 2 Immunofluorescence staining characterization of d26-28 eBCs.
Confocal images of eBCs stained for C-peptide (C-PEP) and the hallmark β cell transcription factors, PDX1 and NKX6.1 (Upper panel). Co-staining for C-peptide, NKX2.2 and SOX9 (marker for progenitors and duct cells) (middle panel) in eBCs. Co-staining for synaptophysin (endocrine), amylase (acinar) and CK19 (ducts) (lower panel) in eBCs. All panels were stained for nuclei with DAPI. Scale bar, 100 μm. Images are representative of three independent experiments yielding similar results.
Supplementary Figure 3 Functional profiling of d20 clusters, eBCs, NECs, human islets (Related to
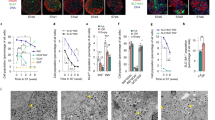
Fig. 2). (a) AUC for the experiment in Fig. 2 (full profile), during the period of low glucose (0–9 min), high glucose (10–31 min) and during first phase of secretion with high glucose (10–15 min). Error bars represent standard error. *P < 0.05, **P < 0.01, ***P < 0.001, ns = not significant determined by one-way ANOVA with Tukey test for multiple comparisons versus eBCs. †P<0.05 determined by two-tailed t-test with Welch’s correction versus eBCs. Refer Supplementary Table 6 for individual p values (b) C-peptide secreted during dynamic perfusion of Non-enriched clusters (NECs) (brown) with 20mM glucose, 10nM exendin-4 and 30mM KCl in an in vitro perifusion assay with a starting basal glucose concentration of 2.8mM. n=3 independent samples. Data=Mean+/- SEM. (c) GSIS on clusters harvested following the 48-hour period of reaggregation of sorted GFP+ cells and cultured for two additional days. eBCs were either incubated in successive 2.8mM glucose KRB (2.8mM-2.8mM glucose) (n = 2 independent experiments) or in 2.8mM and then 20mM glucose condition (2.8mM-20mM glucose)(n = 3 independent experiments). eBCs are non-functional immediately post-aggregation. (d) C-peptide secreted during dynamic perfusion of eBCs with successive rounds of KRB containing alternating 2.8mM and 20mM glucose. n = 2 independent samples. (e) Average of calcium signalling traces of d20 clusters (four independent samples), eBCs (three independent samples) and human islets (three independent samples). Data are presented as Mean+/-SEM for d20 clusters and eBCs; as Mean+/-SD for human islets. eBCs rapidly increase their cytosolic calcium ion concentrations in response to glucose. (f) Bottom-left and bottom-right traces are average of traces from preparations of human islets and eBC preparations (three independent samples) with mediocre function, respectively. Note, eBCs still raise their calcium flux upon glucose stimulation unlike islets but it does not come back to basal flux upon removal of glucose. Refer Supplementary Table 6 for source data.
Supplementary Figure 4 β cell markers are enriched in eBCs compared to d20 immature β-like cells, and similarly expressed as in human islets.
(a)Schematic depicting populations (in boxes) at different stages of differentiation used for qPCR analysis of key β cell markers. d11: pancreatic progenitors (pre-endocrine induction, blue diamond), d20 sorted GFP-high cells (black triangle, n = 3 independent samples, d26-27 eBCs (green circle, n = 4 independent samples) and human islets (red square, n = 6 independent samples). Data are expressed as fold change relative to d11 cells (mean+/-SEM). Pairwise comparison between d20 GFP-high cells and eBCs. p values determined by two-tailed unpaired t-tests without or with Welch’s correction if SD was unequal. (b) QPCR data comparing expression of GCK, ABCC8, PCSK1, KCNJ11, and KCNK3 between eBCs and human islets. Data are expressed as fold-change relative to d11 cells (mean+/-SEM) eBCs, n=4 independent samples; human islets, n = 6 independent samples. P values determined by two-tailed unpaired t-tests without or with Welch’s correction if SD was unequal. Refer Supplementary Table 6 for source data (c) FPKM values from RNA-seq of eBC-β cells and human islet-β cells for genes GLIS3, PCSK2 and GLUT-1. p values and FDR corrected q values are indicated on the graphs. Cuffdiff uses a theoretic metric derived from the Jensen-Shannon divergence to calculate p values41.
Supplementary Figure 5 eBC β cells have reduced expression of disallowed genes.
Schematic depicting the isolation of INSGFP-high + cells via FACS from d20 immature clusters, d26-27 NECs, and d26-27 eBCs for RNA-seq. Note that only INSGFP-high+ cells were analysed for all d20 immature, d26-27 NECs, and eBC populations. Legend: INS-GFP+ (GFP positive insulin-producing cells, green), Panc-Prog (Pancreatic progenitors, blue cells) and Non-Panc (Non-pancreatic cells, purple cells). RNA-seq FPKM values of disallowed genes HK1, HK2, LDHA and MCT1 in d20 Immature β-like cells (Black bar), NEC-β cells (Brown bar) and eBC-β cells (Green bar). n = 2 independent samples.
Supplementary Figure 6 Mitochondrial function and structure of eBCs are better than that of NECs and d20 immature β-like cells.
(a) NECs do not display a significant rise in OCR on glucose stimulation. Mitochondrial respiration assessed by a Cell Mito Stress Test using a Seahorse XFe24 Bioanalyzer. Oxygen consumption rate (OCR) was first measured under basal conditions (2.8mM glucose) followed by sequential addition of either 2.8mM (blue line) or 20mM glucose (red line), 5 μM Oligomycin (Oligo), 1 μm FCCP, and 5 μm Rotenone + Antimycin A (Rot/Anti-M). SRC, Spare respiratory capacity. n = 4 independent samples. Data are expressed as mean±SEM. (b) Quenching of Rhodamine 123 is a reliable signal of inner mitochondrial membrane. To dismiss the possibility of signal decay due to photobleaching, Oligomycin and FCCP were added to the experiment. Addition of Oligomycin A, an inhibitor of ATP synthase, accelerated the decay in fluorescence and FCCP, an uncoupler of OXPHOS, collapsed the mitochondrial potential. Based on these data, we confirm that the results presented in the manuscript do not represent a photobleaching event. This experiment was conducted once as a positive control (c) MitoTracker Red CM-H2XRos specifically binds to the mitochondria. Confocal images of dispersed eBC cells stained live with MitoTracker Red CM-H2XRos followed by fixation and staining with a mitochondrial membrane marker Tomm20, insulin and DapI. Tomm20 and MitoTracker are co-localized whereas insulin granules are not. Images are representative of three independent experiments Scale bar, 30 um (d) Full resolution transmission electron microscopy images of mitochondria in immature β-like cells in d20 clusters and mature β cells of eBCs. Note well formed and densely packed cristae in eBC β cells compared to immature β-like cells (yellow arrows). Scale bar, 250 nm. Images are representative from two biologically independent samples. Refer Supplementary Table 6 for source data.
Supplementary Figure 7 ‘Per cell’ β cell function assessed by transplantation of equivalent numbers of “β cells” in eBCs, NECs and d20.
2000 eBCs (~2x10^6 cells), 4x10^6 cells each of d20 clusters or NECs were transplanted into non-diabetic NSG mice. Human islets were used as positive control. A lower dose of 500 IEQ (~2000cells/IEQ) of human islets was transplanted to approximately match the number of cells present in eBCs and accounting for one missing allele of insulin in the INSGFP/W cells. In vivo glucose challenge was performed three days following transplant. Human C-peptide levels in the serum were measured after an overnight fast (grey bar) and again 60 min after IP glucose injection (black bar).
Supplementary information
Supplementary Information
Supplementary Figures 1–7 and legends for Supplementary Tables 1–6
Rights and permissions
About this article
Cite this article
Nair, G.G., Liu, J.S., Russ, H.A. et al. Recapitulating endocrine cell clustering in culture promotes maturation of human stem-cell-derived β cells. Nat Cell Biol 21, 263–274 (2019). https://doi.org/10.1038/s41556-018-0271-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-018-0271-4
This article is cited by
-
Metabolic switching, growth kinetics and cell yields in the scalable manufacture of stem cell-derived insulin-producing cells
Stem Cell Research & Therapy (2024)
-
Sox9 regulates alternative splicing and pancreatic beta cell function
Nature Communications (2024)
-
From stem cells to pancreatic β-cells: strategies, applications, and potential treatments for diabetes
Molecular and Cellular Biochemistry (2024)
-
CDK8/19 inhibition plays an important role in pancreatic β-cell induction from human iPSCs
Stem Cell Research & Therapy (2023)
-
Islets in the body are never flat: transitioning from two-dimensional (2D) monolayer culture to three-dimensional (3D) spheroid for better efficiency in the generation of functional hPSC-derived pancreatic β cells in vitro
Cell Communication and Signaling (2023)