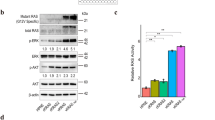
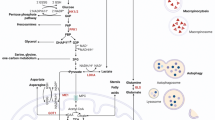
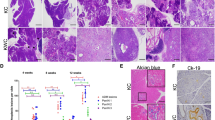
Abstract
G protein αs (GNAS) mediates receptor-stimulated cAMP signalling, which integrates diverse environmental cues with intracellular responses. GNAS is mutationally activated in multiple tumour types, although its oncogenic mechanisms remain elusive. We explored this question in pancreatic tumourigenesis where concurrent GNAS and KRAS mutations characterize pancreatic ductal adenocarcinomas (PDAs) arising from intraductal papillary mucinous neoplasms (IPMNs). By developing genetically engineered mouse models, we show that GnasR201C cooperates with KrasG12D to promote initiation of IPMN, which progress to invasive PDA following Tp53 loss. Mutant Gnas remains critical for tumour maintenance in vivo. This is driven by protein-kinase-A-mediated suppression of salt-inducible kinases (Sik1–3), associated with induction of lipid remodelling and fatty acid oxidation. Comparison of Kras-mutant pancreatic cancer cells with and without Gnas mutations reveals striking differences in the functions of this network. Thus, we uncover Gnas-driven oncogenic mechanisms, identify Siks as potent tumour suppressors, and demonstrate unanticipated metabolic heterogeneity among Kras-mutant pancreatic neoplasms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
O’Hayre, M. et al. The emerging mutational landscape of G proteins and G-protein-coupled receptors in cancer. Nat. Rev. Cancer 13, 412–424 (2013).
Sassone-Corsi, P. The cyclic AMP pathway. Cold Spring Harb. Perspect. Biol. 4, a011148 (2012).
Rosciglione, S., Theriault, C., Boily, M. O., Paquette, M. & Lavoie, C. Gαs regulates the post-endocytic sorting of G protein-coupled receptors. Nat. Commun. 5, 4556 (2014).
Drelon, C. et al. PKA inhibits WNT signalling in adrenal cortex zonation and prevents malignant tumour development. Nat. Commun. 7, 12751 (2016).
He, X. et al. The G protein α subunit Gαs is a tumor suppressor in Sonic hedgehog-driven medulloblastoma. Nat. Med. 20, 1035–1042 (2014).
Iglesias-Bartolome, R. et al. Inactivation of a Gαs-PKA tumour suppressor pathway in skin stem cells initiates basal-cell carcinogenesis. Nat. Cell Biol. 17, 793–803 (2015).
Pattabiraman, D. R. et al. Activation of PKA leads to mesenchymal-to-epithelial transition and loss of tumor-initiating ability. Science 351, aad3680 (2016).
Xing, F. et al. The anti-Warburg effect elicited by the cAMP-PGC1α pathway drives differentiation of glioblastoma cells into astrocytes. Cell Rep. 18, 468–481 (2017).
Wu, J. et al. Recurrent GNAS mutations define an unexpected pathway for pancreatic cyst development. Sci. Transl. Med. 3, 92ra66 (2011).
Amato, E. et al. Targeted next-generation sequencing of cancer genes dissects the molecular profiles of intraductal papillary neoplasms of the pancreas. J. Pathol. 233, 217–227 (2014).
Witkiewicz, A. K. et al. Whole-exome sequencing of pancreatic cancer defines genetic diversity and therapeutic targets. Nat. Commun. 6, 6744 (2015).
Bailey, P. et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 531, 47–52 (2016).
Zehir, A. et al. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat. Med. 23, 703–713 (2017).
Innamorati, G., Valenti, M. T., Giacomello, L., Carbonare, L. & Bassi, C. GNAS mutations: drivers or o-pilots? Yet, promising diagnostic biomarkers. Trends Cancer 2, 282–285 (2016).
Patra, K. C., Bardeesy, N. & Mizukami, Y. Diversity of precursor lesions for pancreatic cancer: the genetics and biology of intraductal papillary mucinous neoplasm. Clin. Transl. Gastroenterol. 8, e86 (2017).
Cancer Genome Atlas Research Network. Integrated genomic characterization of pancreatic ductal adenocarcinoma. Cancer Cell 32 , 185–203 (2017).
Aguirre, A. J. et al. Activated Kras and Ink4a/Arf deficiency cooperate to produce metastatic pancreatic ductal adenocarcinoma. Genes Dev. 17, 3112–3126 (2003).
Farrell, J. J. & Fernandez-del Castillo, C. Pancreatic cystic neoplasms: management and unanswered questions. Gastroenterology 144, 1303–1315 (2013).
Castellone, M. D., Teramoto, H., Williams, B. O., Druey, K. M. & Gutkind, J. S. Prostaglandin E2 promotes colon cancer cell growth through a Gs-axin-beta-catenin signaling axis. Science 310, 1504–1510 (2005).
Wu, J. et al. Inhibition of the EGF-activated MAP kinase signaling pathway by adenosine 3’,5’-monophosphate. Science 262, 1065–1069 (1993).
Altarejos, J. Y. & Montminy, M. CREB and the CRTC co-activators: sensors for hormonal and metabolic signals. Nat. Rev. Mol. Cell Biol. 12, 141–151 (2011).
Yu, F. X. et al. Regulation of the Hippo-YAP pathway by G-protein-coupled receptor signaling. Cell 150, 780–791 (2012).
Cheng, H. et al. SIK1 couples LKB1 to p53-dependent anoikis and suppresses metastasis. Sci. Signal. 2, ra35 (2009).
Wehr, M. C. et al. Salt-inducible kinases regulate growth through the Hippo signalling pathway in Drosophila. Nat. Cell Biol. 15, 61–71 (2013).
Miranda, F. et al. Salt-inducible kinase 2 couples ovarian cancer cell metabolism with survival at the adipocyte-rich metastatic niche. Cancer Cell 30, 273–289 (2016).
Patel, K. et al. The LKB1-salt-inducible kinase pathway functions as a key gluconeogenic suppressor in the liver. Nat. Commun. 5, 4535 (2014).
Sonntag, T., Vaughan, J. M. & Montminy, M. 14-3-3 proteins mediate inhibitory effects of cAMP on salt-inducible kinases (SIKs). FEBS J. 285, 467–480 (2017).
Clark, K. et al. Phosphorylation of CRTC3 by the salt-inducible kinases controls the interconversion of classically activated and regulatory macrophages. Proc. Natl Acad. Sci. USA 109, 16986–16991 (2012).
Zechner, R. et al. FAT SIGNALS—lipases and lipolysis in lipid metabolism and signaling. Cell Metab. 15, 279–291 (2012).
Park, J. et al. SIK2 is critical in the regulation of lipid homeostasis and adipogenesis in vivo. Diabetes 63, 3659–3673 (2014).
Wang, B. et al. A hormone-dependent module regulating energy balance. Cell 145, 596–606 (2011).
Bricambert, J. et al. Salt-inducible kinase 2 links transcriptional coactivator p300 phosphorylation to the prevention of ChREBP-dependent hepatic steatosis in mice. J. Clin. Invest. 120, 4316–4331 (2010).
Benjamin, D. I. et al. Ether lipid generating enzyme AGPS alters the balance of structural and signaling lipids to fuel cancer pathogenicity. Proc. Natl Acad. Sci. USA 110, 14912–14917 (2013).
Lodhi, I. J. & Semenkovich, C. F. Peroxisomes: a nexus for lipid metabolism and cellular signaling. Cell Metab. 19, 380–392 (2014).
Li, Y. Q. et al. Gsα deficiency in adipose tissue improves glucose metabolism and insulin sensitivity without an effect on body weight. Proc. Natl Acad. Sci. USA 113, 446–451 (2016).
Louie, S. M., Roberts, L. S., Mulvihill, M. M., Luo, K. & Nomura, D. K. Cancer cells incorporate and remodel exogenous palmitate into structural and oncogenic signaling lipids. Biochim Biophys Acta 1831, 1566–1572 (2013).
Menendez, J. A. & Lupu, R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat. Rev. Cancer 7, 763–777 (2007).
Currie, E., Schulze, A., Zechner, R., Walther, T. C. & Farese Jr., R. V. Cellular fatty acid metabolism and cancer. Cell Metab. 18, 153–161 (2013).
Padanad, M. S. et al. Fatty acid oxidation mediated by acyl-CoA synthetase long chain 3 is required for mutant KRAS lung tumorigenesis. Cell Rep. 16, 1614–1628 (2016).
Nomura, D. K. et al. Monoacylglycerol lipase regulates a fatty acid network that promotes cancer pathogenesis. Cell 140, 49–61 (2010).
Viale, A. et al. Oncogene ablation-resistant pancreatic cancer cells depend on mitochondrial function. Nature 514, 628–632 (2014).
Lim, J. H. et al. Oleic acid stimulates complete oxidation of fatty acids through protein kinase A-dependent activation of SIRT1-PGC1alpha complex. J. Biol. Chem. 288, 7117–7126 (2013).
Gerhart-Hines, Z. et al. The cAMP/PKA pathway rapidly activates SIRT1 to promote fatty acid oxidation independently of changes in NAD+. Mol. Cell 44, 851–863 (2011).
Wilderman, A. et al. Proteomic and metabolic analyses of S49 lymphoma cells reveal novel regulation of mitochondria by cAMP and protein kinase A. J. Biol. Chem. 290, 22274–22286 (2015).
Ying, H. et al. Oncogenic Kras maintains pancreatic tumors through regulation of anabolic glucose metabolism. Cell 149, 656–670 (2012).
Dominguez, E. et al. Integrated phenotypic and activity-based profiling links Ces3 to obesity and diabetes. Nat. Chem. Biol. 10, 113–121 (2014).
Mino-Kenudson, M. et al. Prognosis of invasive intraductal papillary mucinous neoplasm depends on histological and precursor epithelial subtypes. Gut 60, 1712–1720 (2011).
Honeyman, J. N. et al. Detection of a recurrent DNAJB1-PRKACA chimeric transcript in fibrolamellar hepatocellular carcinoma. Science 343, 1010–1014 (2014).
Shackelford, D. B. & Shaw, R. J. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nat. Rev. Cancer 9, 563–575 (2009).
Lodish, M. & Stratakis, C. A. A genetic and molecular update on adrenocortical causes of Cushing syndrome. Nat. Rev. Endocrinol. 12, 255–262 (2016).
Gaujoux, S. et al. Hepatobiliary and pancreatic neoplasms in patients with McCune-Albright syndrome. J. Clin. Endocrinol. Metab. 99, E97–E101 (2014).
Bossis, I. et al. Protein kinase A and its role in human neoplasia: the Carney complex paradigm. Endocr. Relat. Cancer 11, 265–280 (2004).
Taki, K. et al. GNASR201H and KrasG12D cooperate to promote murine pancreatic tumorigenesis recapitulating human intraductal papillary mucinous neoplasm. Oncogene 35, 2407–2412 (2016).
von Figura, G. et al. The chromatin regulator Brg1 suppresses formation of intraductal papillary mucinous neoplasm and pancreatic ductal adenocarcinoma. Nat. Cell Biol. 16, 255–267 (2014).
Collins, M. A. et al. Oncogenic Kras is required for both the initiation and maintenance of pancreatic cancer in mice. J. Clin. Invest. 122, 639–653 (2012).
Wang, Y. Y. et al. Mammary adipocytes stimulate breast cancer invasion through metabolic remodeling of tumor cells. JCI Insight 2, e87489 (2017).
Caro, P. et al. Metabolic signatures uncover distinct targets in molecular subsets of diffuse large B cell lymphoma. Cancer Cell 22, 547–560 (2012).
Prentki, M. & Madiraju, S. R. Glycerolipid/free fatty acid cycle and islet beta-cell function in health, obesity and diabetes. Mol. Cell Endocrinol. 353, 88–100 (2012).
Schug, Z. T., Vande Voorde, J. & Gottlieb, E. The metabolic fate of acetate in cancer. Nat. Rev. Cancer 16, 708–717 (2016).
Hauser, A. S., Attwood, M. M., Rask-Andersen, M., Schioth, H. B. & Gloriam, D. E. Trends in GPCR drug discovery: new agents, targets and indications. Nat. Rev. Drug Discov. 16, 829–842 (2017).
Dickins, R. A. et al. Probing tumor phenotypes using stable and regulated synthetic microRNA precursors. Nat. Genet. 37, 1289–1295 (2005).
Kawaguchi, Y. et al. The role of the transcriptional regulator Ptf1a in converting intestinal to pancreatic progenitors. Nat. Genet. 32, 128–134 (2002).
Beard, C., Hochedlinger, K., Plath, K., Wutz, A. & Jaenisch, R. Efficient method to generate single-copy transgenic mice by site-specific integration in embryonic stem cells. Genesis 44, 23–28 (2006).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Reichert, M. et al. Isolation, culture and genetic manipulation of mouse pancreatic ductal cells. Nat. Protocols 8, 1354–1365 (2013).
Lapek, J. D. Jr. et al. Detection of dysregulated protein-association networks by high-throughput proteomics predicts cancer vulnerabilities. Nat. Biotechnol. 35, 983–989 (2017).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Smulan, L. J. et al. Cholesterol-independent SREBP-1 maturation is linked to ARF1 inactivation. Cell Rep. 16, 9–18 (2016).
Louie, S. M. et al. GSTP1 is a driver of triple-negative breast cancer cell metabolism and pathogenicity. Cell Chem. Biol. 23, 567–578 (2016).
Huynh, F. K., Green, M. F., Koves, T. R. & Hirschey, M. D. Measurement of fatty acid oxidation rates in animal tissues and cell lines. Methods Enzymol. 542, 391–405 (2014).
Acknowledgements
We thank L. Ellisen, R. Mostoslavsky, F. Kottakis and other members of the Bardeesy laboratory for helpful suggestions throughout the course of this project and critical reading of the manuscript. We thank C. Wright for Ptf1a-Cre and Ptf1a-CreER, D. Tuveson and T. Jacks for LSL-KrasG12D and A. Berns for Tp53Lox/Lox animal strains, N. Gray for SIK inhibitors, S. Boukhali and A. Escudier for experimental support and M. Keibler for initial help with metabolomics analysis. N.B. holds the Gallagher Endowed Chair in Gastrointestinal Cancer Research and received support from the Granara-Skerry Trust, the Linda J. Verville Foundation, the Begg Family, and grants from the Fibrolamellar Cancer Foundation and the NIH (P01 CA117969-07, R01 CA133557-05, P50CA1270003). K.C.P. is supported by a post-doctoral fellowship from Department of Defense, USA (W81XWH-16-1-0285). K.C.P., Y.M. and N.B. are fellows of the Andrew L. Warshaw Institute for Pancreatic Cancer at Massachusetts General Hospital.
Author information
Authors and Affiliations
Contributions
K.C.P., Y.M., and N.B conceived and designed the study. Y.M. generated the TetO-GnasR201C mouse strain. K.P. performed most of the animal and cell-based experiments, with assistance of Y.K., Y.M., S.W., and I.R. Y.K. and M.M-K performed histological analysis. M.B. and W.H. performed proteomics analysis and interpreted the data. F.J. and R.I.S. performed bioinformatics analysis. E.A.G. did LC-MS for polar metabolites, and D.K.N. analysed and interpreted the data. A.S.L., R.A.S., and K.S. provided essential resources. A.S.L., R.A.S., K.S., D.P.R. M.M-K. and C.F.C provided important intellectual input and data interpretation. K.C.P. and N.B. wrote the manuscript with feedback from all authors. N.B. supervised the studies.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Characterization of GnasR201C GEM model.
a, H&E stained sections of control and GC pancreata at 12 weeks of age. b, IHC staining for IPMN markers in pancreata from mice of the indicated genotypes. The insets show higher magnification views. Staining of different subtypes of human IPMN (right panels) is presented as a reference; pancreatobiliary (PB), intestinal (I) and gastric (G) type IPMN specimens are arranged as shown in the grid to the right. c, H&E images of end-stage KGC pancreata (12 weeks of age) revealing IPMNs with a range of histological grades. H&E and IHC data are representative of 3 mice/group. Scale bars: (a) 100 μm, (b) for KC and KGC (left) 200 μm, inset 40 μm and for human IPMN (right) 40 μm (c) 40 μm.
Supplementary Figure 2 Role of mutant GNAS in tumour maintenance.
a, Representative gross appearance of animals of indicated genotypes. Left panel: KC mouse, age 10 weeks; Middle panel: KGC mouse, age 10 weeks; Right panel: same KGC mice at age 30 weeks (following Dox withdrawal at age 10 weeks). b, IHC for cleaved caspase-3 in pancreatic tumours from 12-week old KGC mice provided Dox supplementation or following Dox withdrawal for the indicated number of days. Minimal staining was observed in each group (3 animals/group). Right panels: Duodenal tissue from a TNFα-treated mouse serves as a positive control. The boxed regions are shown as higher magnification views in the lower panels. Scale bars: (b) 200 μm upper, 40 μm lower.
Supplementary Figure 3 PKA is required for GnasR201C-mediated tumour maintenance.
a, Growth of KPCshp53 organoids ± Dox. b, cAMP levels in KGC cultures grown ± Dox and ± isobutylmethylxanthine (IBMX; 200 μM) or FSK (10 μM). IBMX is used to gauge cAMP production by preventing phosphodiesterase-mediated cAMP hydrolysis. c, KGC organoids grown ± Dox and ± 10 μM Forskolin (FSK), 100 μM Sp-8-Br-cAMPS, or 100 μM 8-pCPT-2’-O-Me-cAMP. Data are quantified in main text, Fig. 4d. N = 3 independent biological replicates. d, mRNA expression of PKA subunits and Rapgef3 and 4 (EPAC1 and 2) in KGC cells (FPKM: Fragments Per Kilobase of transcript per Million mapped reads). RNA-seq was performed using two KGC lines, each tested as two independent biological replicates. e, Immunoblot for PKA targets in lysates of KGC organoids grown ± Dox and ± the indicated agonists. f, g, Demonstration of Dox-dependent control of GnasR201C in vivo. Mice injected subcutaneously with KGC organoids were provided Dox until tumours reached ~500 mm3 volume, and then randomized into + Dox and -Dox groups. Tumours were isolated after 96 hrs and subjected to qRT- PCR (f) and immunoblot analyses (g). Each column/lane represents an independent tumour. Gnas mRNA levels and phosphorylation of the PKA target, VASP, decrease upon Dox withdrawal, whereas Gnas protein levels are relatively unchanged, indicating near physiological GnasR201C protein expression (consistent with the reported instability of mutant GNAS)1. h, Growth of KGC organoids expressing empty vector (V) or PKADN and cultured in ± Dox. Data are quantified in main text, Fig. 4g. i, Representative images of tumours formed by the indicated KGC cells. Data are quantified in main text, Fig. 4i. Scale bars: (a, c, and h) 750 μm, (i) 0.5 cm. a-c: N = 3, f: N = 3, h: N = 6 independent biological replicates and i: N = 8 tumours/group. Data in (b) derived from 2D culture. Error bars: (a, b, f: ± s.e.m.). Immunoblots in (e) and (g) were performed two times with similar results. Significance was analysed using two-tailed Student’s t-test. p < 0.05 was considered statistically significant. Source data are provided in Supplementary Table 2. Supplementary Fig. 8 shows original scans of the immunoblots.
Supplementary Figure 4 SIKs are key targets of GnasR201C-PKA signaling in pancreatic tumour maintenance.
a, KGC organoids grown ± Dox were tested by immunoblot for the indicated markers. pYap(Ser-127); pβ- catenin(Ser-33/37/Thr-41); pERK(Thr-202/Tyr-204). Immunoblots were performed two times with similar results. b, mRNA expression levels (FPKM) of SIK family genes in KGC organoid lines. RNA-seq was performed using two KGC lines, each tested as two independent biological replicates c, Upper: Schematic of SIK2-S4A. Lower and Right: Growth of KGC organoids ectopically expressing SIK2-WT or SIK2-S4A. d, qRT-PCR analysis showing relative expression of Sik1, 2, and 3 in organoids engineered with Cas9 and the indicated sgRNAs. Scale bars: (c) 750 μm. c: N = 4 and d: N = 3 independent biological replicates. Error bars: (b, d: ± s.d and c: ± s.e.m.). Significance was analysed using two-tailed Student’s t-test. p < 0.05 was considered statistically significant. Source data are provided in Supplementary Table 2. Original scans of the immunoblots are shown in Supplementary Fig. 8.
Supplementary Figure 5 Mutant Gnas induces lipid metabolism expression signatures in pancreatic tumour cells.
a, Schematic: integration of lipid metabolism. Blue text: selected enzymes activated/upregulated by GnasR201C. FA: Fatty acid; TG: triglyceride, DG: diglyceride, MG: monoglyceride; VLC/LC/SC-FA: very long-chain/long- chain/short-chain-fatty acid. b, c, qRT-PCR analysis of lipid metabolism enzymes in (b) KGC organoids grown in vitro ± Dox and ± KT5270 (30 μM) and (c) subcutaneous tumours formed from KGC organoids injected into SCID mice. Tumours were harvested from mice provided with Dox or following Dox withdrawal for 96 hrs. Starting tumour volume was ~500 mm3. d, RNA isolated from the indicated human PDA cell lines expressing empty vector or PKADN was examined by qRT-PCR analysis for the indicated genes. b: N = 3, d: N = 6 independent biological replicates, c: N = 8 tumours/group. Error bars: (b-d: ± s.e.m.). Significance was analysed using two-tailed Student’s t-test. p < 0.05 was considered statistically significant. p values for b-d are provided in the source data to avoid crowding in the figure panels. Source data are provided in Supplementary Table 2.
Supplementary Figure 6 Mutant Gnas controls lipid metabolism in pancreatic tumour cells.
a, Neutral lipid levels (BODIPY staining) in KGC 2D cultures grown ± Dox. b-e, LC-MS analysis of KGC organoids ± Dox for (b) total lipids, (c) relative levels of lipid classes (see Supplementary Table 2 source data for definition of the abbreviations and p values of the statistical test), (d) relative levels of alkyl and plasmalogen ether lipids versus total lipids, and (e) relative levels of alkyl and plasmalogen ether lipids. f, Relative abundance of lipases detected by quantitative proteomics in KGC organoids ± Dox. Data are from two independent organoid lines (A and B) and two separate experiments, with 2-3 replicates per condition as indicated. Protein levels are shown as log2 ratio of protein intensity over the average intensity across all samples of the studied cell line in each experiment. For colour coding, ratios were capped as indicated. g, Relative FAO rate assessed by 14CO2 trap in human PDA cell lines expressing empty vector or PKADN. h, Immunoblot of lysates from subcutaneous KGC tumours harvested from mice provided with Dox, or following Dox withdrawal for 96 hrs. Starting tumour volume was ~500 mm3. Each lane represents an independent tumour. Immunoblot was performed twice with similar results. i, Relative FAO rate in KGC cultures grown ± Dox and ± FSK (10 μM) or HG- 9-91-01 (500 nM). j, Relative FAO rate in KGC cultures engineered with sgRNAs to Sik1-3 or GFP with Cas9 and grown ± Dox. k, Relative acetyl-CoA levels in Gnas mutant (KGC) or Gnas-wt (KPC) cultures expressing shGFP or two different shRNAs against Cpt1a, measured by fluorometric assay. Scale bar in (a) 25 μm. a: N = 4, b, c, e: N = 3, g: N = 4, i: N = 5, j: N = 6, k: N = 5 (GnasMUT); N = 3 (GnasWT) independent biological replicates. LC-MS experiments were performed in triplicate for the + Dox and -Dox conditions. Error bars: (b, c, e: ± s.d.), (a, g i-k: ± s.e.m.). Significance was analysed using two-tailed Student’s t-test. p < 0.05 was considered statistically significant. Source data are provided in Supplementary Table 2. Original scans of the immunoblots are shown in Supplementary Fig. 8.
Supplementary Figure 7 Distinct metabolic circuitry of Gnas-mutant and WT pancreatic tumour cells.
a, Growth of murine Gnas mutant (MUT) and WT pancreatic tumour organoids expressing PKADN compared to empty vector. GnasMUT organoid data from a different line is also shown in Fig. 4h of the main text. b, Growth of organoids treated with increasing concentration of KT5720. c, Relative FAO rate in the indicated human PDA cell lines. d, Response (IC50) of organoids treated with the carboxyesterase/lipase inhibitor WWL113, which inactivates Ces3, Ces1f, Ces1 and Ces1c and partially inhibits Abdh6. e, IC50 measurements showing response of organoids to the FAO inhibitor, BrCA. f, qRT-PCR data validating knockdown of Cpt1a in the indicated organoids (corresponds with Fig. 7j and k in the main text). h, Response of organoids to increasing concentrations of the glycolysis inhibitors 2-deoxyglucose (2DG) and oxamate. a: N = 4, b, f-h: N = 3, c: N = 5 independent biological replicates. (d, e) Data pooled from N = 3 independent biological replicates. Error bars: (a-c, f-h: ± s.e.m). Significance was analysed using two-tailed Student’s t-test. p < 0.05 was considered statistically significant. Source data are provided in Supplementary Table 2.
Supplementary Figure 8
Scans of unprocessed immunoblots.
Supplementary information
Supplementary Information
Supplementary Figures 1–8, and legends for Supplementary Tables 1 and 2
Rights and permissions
About this article
Cite this article
Patra, K.C., Kato, Y., Mizukami, Y. et al. Mutant GNAS drives pancreatic tumourigenesis by inducing PKA-mediated SIK suppression and reprogramming lipid metabolism. Nat Cell Biol 20, 811–822 (2018). https://doi.org/10.1038/s41556-018-0122-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-018-0122-3
This article is cited by
-
cAMP-PKA/EPAC signaling and cancer: the interplay in tumor microenvironment
Journal of Hematology & Oncology (2024)
-
Adenosquamous carcinoma coexisting with intraductal papillary mucinous neoplasm of the pancreas: a case report
Journal of Medical Case Reports (2023)
-
Activation of melanocortin-1 receptor signaling in melanoma cells impairs T cell infiltration to dampen antitumor immunity
Nature Communications (2023)
-
Mechanisms of obesity- and diabetes mellitus-related pancreatic carcinogenesis: a comprehensive and systematic review
Signal Transduction and Targeted Therapy (2023)
-
Current status of molecular diagnostic approaches using liquid biopsy
Journal of Gastroenterology (2023)