Abstract
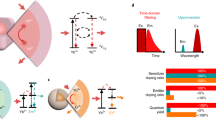
Characterizing sources and targets of illumination in living tissue is challenging. Here we show that spatial distributions of light in tissue can be mapped by using magnetic resonance imaging (MRI) in the presence of photosensitive nanoparticle probes. Each probe consists of a reservoir of paramagnetic molecules enclosed by a liposomal membrane incorporating photosensitive lipids. Incident light causes the photoisomerization of the lipids and alters hydrodynamic exchange across the membrane, thereby affecting longitudinal relaxation-weighted contrast in MRI. We injected the nanoparticles into the brains of live rats and used MRI to map responses to illumination profiles characteristic of widely used applications of photostimulation, photometry and phototherapy. The responses deviated from simple photon propagation models and revealed signatures of light scattering and nonlinear responsiveness. Paramagnetic liposomal nanoparticles may enable MRI to map a broad range of optical phenomena in deep tissue and other opaque environments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. The raw data generated in this study are available from the corresponding author on reasonable request. Source data for the figures are provided with this paper.
References
Tu, C. & Louie, A. Y. Strategies for the development of gadolinium-based “q”-activatable MRI contrast agents. NMR Biomed. 26, 781–787 (2013).
Lux, J. & Sherry, A. D. Advances in gadolinium-based MRI contrast agent designs for monitoring biological processes in vivo. Curr. Opin. Chem. Biol. 45, 121–130 (2018).
Wei, H., Frey, A. M. & Jasanoff, A. Molecular fMRI of neurochemical signaling. J. Neurosci. Methods 364, 109372 (2021).
Angelovski, G. What we can really do with bioresponsive MRI contrast agents. Angew. Chem. Int. Ed. Engl. 55, 7038–7046 (2016).
Boros, E., Gale, E. M. & Caravan, P. MR imaging probes: design and applications. Dalton Trans. 44, 4804–4818 (2015).
Li, H. & Meade, T. J. Molecular magnetic resonance imaging with Gd(III)-based contrast agents: challenges and key advances. J. Am. Chem. Soc. 141, 17025–17041 (2019).
Lauffer, R. B. Paramagnetic metal complexes as water proton relaxation agents for NMR imaging: theory and design. Chem. Rev. 87, 901–927 (1987).
Wahsner, J., Gale, E. M., Rodriguez-Rodriguez, A. & Caravan, P. Chemistry of MRI contrast agents: current challenges and new frontiers. Chem. Rev. 119, 957–1057 (2019).
Venkataramani, S. et al. Magnetic bistability of molecules in homogeneous solution at room temperature. Science 331, 445–448 (2011).
Tu, C. & Louie, A. Y. Photochromically-controlled, reversibly-activated MRI and optical contrast agent. Chem. Commun. https://doi.org/10.1039/B616991K (2007).
Gao, M. et al. Synthesis and comparative evaluation of photoswitchable magnetic resonance imaging contrast agents. ACS Omega 5, 14759–14766 (2020).
Reessing, F. et al. A light-responsive liposomal agent for MRI contrast enhancement and monitoring of cargo delivery. Chem. Commun. 55, 10784–10787 (2019).
Reessing, F. et al. A photocleavable contrast agent for light-responsive MRI. Pharmaceuticals 13, 296 (2020).
Koenig, S. H. et al. Permeability of liposomal membranes to water: results from the magnetic field dependence of T1 of solvent protons in suspensions of vesicles with entrapped paramagnetic ions. Magn. Reson. Med. 23, 275–286 (1992).
Terreno, E. et al. Determination of water permeability of paramagnetic liposomes of interest in MRI field. J. Inorg. Biochem. 102, 1112–1119 (2008).
Morgan, C. G., Sandhu, S. S., Yianni, Y. P. & Dodd, N. J. The phase behaviour of dispersions of Bis-Azo PC: photoregulation of bilayer dynamics via lipid photochromism. Biochim. Biophys. Acta 903, 495–503 (1987).
Pernpeintner, C. et al. Light-controlled membrane mechanics and shape transitions of photoswitchable lipid vesicles. Langmuir 33, 4083–4089 (2017).
Morstein, J., Impastato, A. C. & Trauner, D. Photoswitchable lipids. ChemBioChem 22, 73–83 (2021).
Osborne, E. A., Jarrett, B. R., Tu, C. & Louie, A. Y. Modulation of T2 relaxation time by light-induced, reversible aggregation of magnetic nanoparticles. J. Am. Chem. Soc. 132, 5934–5935 (2010).
Kruttwig, K. et al. Reversible low-light induced photoswitching of crowned spiropyran-DO3A complexed with gadolinium(III) ions. Molecules 17, 6605–6624 (2012).
Dommaschk, M. et al. Photoswitchable magnetic resonance imaging contrast by improved light-driven coordination-induced spin state switch. J. Am. Chem. Soc. 137, 7552–7555 (2015).
Dong, M., Babalhavaeji, A., Samanta, S., Beharry, A. A. & Woolley, G. A. Red-shifting azobenzene photoswitches for in vivo use. Acc. Chem. Res. 48, 2662–2670 (2015).
Chander, N. et al. Optimized photoactivatable lipid nanoparticles enable red light triggered drug release. Small 17, e2008198 (2021).
Thorne, R. G. & Nicholson, C. In vivo diffusion analysis with quantum dots and dextrans predicts the width of brain extracellular space. Proc. Natl Acad. Sci. USA 103, 5567–5572 (2006).
Yona, G., Meitav, N., Kahn, I. & Shoham, S. Realistic numerical and analytical modeling of light scattering in brain tissue for optogenetic applications. eNeuro 3, ENEURO.0059-15.2015 (2016).
Aravanis, A. M. et al. An optical neural interface: in vivo control of rodent motor cortex with integrated fiberoptic and optogenetic technology. J. Neural Eng. 4, S143–S156 (2007).
Acker, L., Pino, E. N., Boyden, E. S. & Desimone, R. FEF inactivation with improved optogenetic methods. Proc. Natl Acad. Sci. USA 113, E7297–E7306 (2016).
Wang, L. V. & Wu, H.-I. Biomedical Optics: Principles and Imaging (Wiley, 2007).
Canales, A., Park, S., Kilias, A. & Anikeeva, P. Multifunctional fibers as tools for neuroscience and neuroengineering. Acc. Chem. Res. 51, 829–838 (2018).
Ghosh, S., Harvey, P., Simon, J. C. & Jasanoff, A. Probing the brain with molecular fMRI. Curr. Opin. Neurobiol. 50, 201–210 (2018).
Cabre, G. et al. Rationally designed azobenzene photoswitches for efficient two-photon neuronal excitation. Nat. Commun. 10, 907 (2019).
Hai, A., Cai, L. X., Lee, T., Lelyveld, V. S. & Jasanoff, A. Molecular fMRI of serotonin transport. Neuron 92, 754–765 (2016).
Acknowledgements
This research was funded by NIH grants R21 DA044748 and R01 NS120592 and a grant from the G. Harold and Leila Y. Mathers Foundation to A.J. J.S. was supported by a Friends of the McGovern Fellowship from the McGovern Institute for Brain Research and by the MIT Neurobiological Engineering Training Program (NIH T32 EB019940). M.S. was the recipient of a Marie Curie Individual Fellowship from the European Commission. We thank G. Yona and S. Shoham for advice about light modelling and for sharing code for computing beam-spreading profiles.
Author information
Authors and Affiliations
Contributions
J.S. and A.J. developed the LisNR concept. J.S., M.S. and A.J. designed the research. J.S. performed and analysed the in vitro experiments. M.S. performed the in vivo experiments. M.S. and A.J. analysed the in vivo data. J.M. and D.T. synthesized and characterized AzoPC and advised on the in vitro light-response experiments. J.S., M.S. and A.J. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Jeff Bulte and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Light-LisNR response as a function of stimulus duration.
a, MRI signal in response to UV irradiation of Light-LisNRs (orange) over varying durations: 0.5 min (left), 2 min (middle), and 6 min (right). Corresponding unilluminated control data shown in black. Shaded margins denote s.e.m. of n = 3 animals. b, Equivalent time course data for blue illumination. c, Signal differences observed following UV irradiation (orange) or control treatment (black) of LisNRs over the durations indicated. Error bars denote s.e.m. of n = 3 animals. d, Equivalent signal difference data for blue illumination.
Extended Data Fig. 2 Dissociation between light response rates and amplitudes.
a, Top: Light-LisNR response amplitudes to blue and UV light are highly correlated over a 25 ×25 voxel region around fibre optic tips in six animals (R = 0.89, p < 10−5). Bottom: in contrast, blue light response amplitudes and response rates are statistically independent over the same voxels (R = –0.013, p = 0.66), indicating that the LisNR response rates are not strongly affected by variations in probe concentration, which largely determine the signal change amplitude distribution. b, Response amplitudes and rates can also be dissociated from one another as a function of distance from the fibre tip: amplitudes decrease monotonically (top), while response rates peak and then diminish (bottom). Error bars denote SEM over 6 (top) or 3 to 6 (bottom) biological replicates. c, Histograms of blue light response amplitudes (top) and response rates (bottom) over three cycles of illumination (B1-B3), showing stability over time (n = 2 animals). d, Box plots corresponding to the distributions in panel c. Central line = median, box limits = first and third quartiles, whiskers = limiting values. Consistency of the distributions indicates that diffusion and convection over the ~1 hour experimental time period do not strongly affect the results.
Supplementary information
Supplementary Information
Supplementary figures.
Source data
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Simon, J., Schwalm, M., Morstein, J. et al. Mapping light distribution in tissue by using MRI-detectable photosensitive liposomes. Nat. Biomed. Eng 7, 313–322 (2023). https://doi.org/10.1038/s41551-022-00982-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-022-00982-3
This article is cited by
-
Superwettable interface towards biodetection in confined space
Nano Research (2024)
-
Wireless agents for brain recording and stimulation modalities
Bioelectronic Medicine (2023)
-
Nanomechanical action opens endo-lysosomal compartments
Nature Communications (2023)
-
Mapping light distribution in the brain via MRI
Nature Biomedical Engineering (2022)