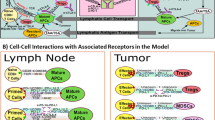
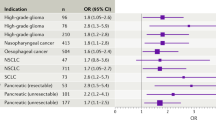
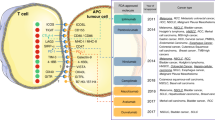
Abstract
A large proportion of patients with cancer are unresponsive to treatment with immune checkpoint blockade and other immunotherapies. Here, we report a mathematical model of the time course of tumour responses to immune checkpoint inhibitors. The model takes into account intrinsic tumour growth rates, the rates of immune activation and of tumour–immune cell interactions, and the efficacy of immune-mediated tumour killing. For 124 patients, four cancer types and two immunotherapy agents, the model reliably described the immune responses and final tumour burden across all different cancers and drug combinations examined. In validation cohorts from four clinical trials of checkpoint inhibitors (with a total of 177 patients), the model accurately stratified the patients according to reduced or increased long-term tumour burden. We also provide model-derived quantitative measures of treatment sensitivity for specific drug–cancer combinations. The model can be used to predict responses to therapy and to quantify specific drug–cancer sensitivities in individual patients.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All of the data supporting the results in this study are available within the paper and its Supplementary Information. The raw, de-identified patient data are available from the corresponding authors upon reasonable request and subject to Institutional Review Board approval.
References
Disis, M. L. Mechanism of action of immunotherapy. Semin Oncol. 41, S3–S13 (2014).
Farkona, S., Diamandis, E. P. & Blasutig, I. M. Cancer immunotherapy: the beginning of the end of cancer? BMC Med. 14, 73 (2016).
Topalian, S. L. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 366, 2443–2454 (2012).
Rosenberg, J. E. et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet 387, 1909–1920 (2016).
Schumacher, T. N., Kesmir, C. & van Buuren, M. M. Biomarkers in cancer immunotherapy. Cancer Cell 27, 12–14 (2015).
Chiou, V. L. & Burotto, M. Pseudoprogression and immune-related response in solid tumors. J. Clin. Oncol. 33, 3541–3543 (2015).
Carthon, B. C. et al. Preoperative CTLA-4 blockade: tolerability and immune monitoring in the setting of a presurgical clinical trial. Clin. Cancer Res. 16, 2861–2871 (2010).
Ahmadzadeh, M. et al. Tumor antigen-specific CD8 T cells infiltrating the tumor express high levels of PD-1 and are functionally impaired. Blood 114, 1537–1544 (2009).
Schmidt, H. et al. Pretreatment levels of peripheral neutrophils and leukocytes as independent predictors of overall survival in patients with American Joint Committee on Cancer Stage IV Melanoma: results of the EORTC 18951 Biochemotherapy Trial. J. Clin. Oncol. 25, 1562–1569 (2007).
Phan, G. Q., Attia, P., Steinberg, S. M., White, D. E. & Rosenberg, S. A. Factors associated with response to high-dose interleukin-2 in patients with metastatic melanoma. J. Clin. Oncol. 19, 3477–3482 (2001).
Yuan, J. et al. Integrated NY-ESO-1 antibody and CD8+ T-cell responses correlate with clinical benefit in advanced melanoma patients treated with ipilimumab. Proc. Natl Acad. Sci. USA 108, 16723–16728 (2011).
Yuan, J. et al. Pretreatment serum VEGF is associated with clinical response and overall survival in advanced melanoma patients treated with ipilimumab. Cancer Immunol. Res. 2, 127–132 (2014).
Blay, J. Y. et al. Serum level of interleukin 6 as a prognosis factor in metastatic renal cell carcinoma. Cancer Res. 52, 3317–3322 (1992).
Kelderman, S. et al. Lactate dehydrogenase as a selection criterion for ipilimumab treatment in metastatic melanoma. Cancer Immunol. Immunother. 63, 449–458 (2014).
Rizvi, N. A. et al. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348, 124–128 (2015).
Van Allen, E. M. et al. Genomic correlates of response to CTLA-4 blockade in metastatic melanoma. Science 350, 207–211 (2015).
Meng, X., Huang, Z., Teng, F., Xing, L. & Yu, J. Predictive biomarkers in PD-1/PD-L1 checkpoint blockade immunotherapy. Cancer Treat. Rev. 41, 868–876 (2015).
Madore, J. et al. PD-L1 expression in melanoma shows marked heterogeneity within and between patients: implications for anti-PD-1/PD-L1 clinical trials. Pigment Cell Melanoma Res. 28, 245–253 (2015).
Sabatino, M. et al. Serum vascular endothelial growth factor and fibronectin predict clinical response to high-dose interleukin-2 therapy. J. Clin. Oncol. 27, 2645–2652 (2009).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Deisboeck, T. S., Wang, Z., Macklin, P. & Cristini, V. Multiscale cancer modeling. Annu. Rev. Biomed. Eng. 13, 127–155 (2011).
Wang, Z., Butner, J. D., Kerketta, R., Cristini, V. & Deisboeck, T. S. Simulating cancer growth with multiscale agent-based modeling. Semin Cancer Biol. 30, 70–78 (2015).
Cristini, V., Koay, E. & Wang, Z. An Introduction to Physical Oncology: How Mechanistic Mathematical Modeling Can Improve Cancer Therapy Outcomes (CRC Press, 2017).
Wang, Z., Butner, J. D., Cristini, V. & Deisboeck, T. S. Integrated PK-PD and agent-based modeling in oncology. J. Pharmacokinet. Pharmacodyn. 42, 179–189 (2015).
Gillies, R. J., Brown, J. S., Anderson, A. R. A. & Gatenby, R. A. Eco-evolutionary causes and consequences of temporal changes in intratumoural blood flow. Nat. Rev. Cancer 18, 576–585 (2018).
Wang, Z. & Deisboeck, T. S. Dynamic targeting in cancer treatment. Front. Physiol. 10, 96 (2019).
Dogra, P. et al. Mathematical modeling in cancer nanomedicine: a review. Biomed. Microdevices 21, 40 (2019).
Dogra, P. et al. Image-guided mathematical modeling for pharmacological evaluation of nanomaterials and monoclonal antibodies. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 12, e1628 (2020).
Kronik, N., Kogan, Y., Vainstein, V. & Agur, Z. Improving alloreactive CTL immunotherapy for malignant gliomas using a simulation model of their interactive dynamics. Cancer Immunol. Immunother. 57, 425–439 (2008).
Eikenberry, S., Thalhauser, C. & Kuang, Y. Tumor-immune interaction, surgical treatment, and cancer recurrence in a mathematical model of melanoma. PLoS Comput. Biol. 5, e1000362 (2009).
Wells, D. K. et al. Spatial and functional heterogeneities shape collective behavior of tumor-immune networks. PLoS Comput. Biol. 11, e1004181 (2015).
Palladini, A. et al. In silico modeling and in vivo efficacy of cancer-preventive vaccinations. Cancer Res. 70, 7755–7763 (2010).
Pappalardo, F., Lollini, P. L., Castiglione, F. & Motta, S. Modeling and simulation of cancer immunoprevention vaccine. Bioinformatics 21, 2891–2897 (2005).
Walker, R. & Enderling, H. From concept to clinic: mathematically informed immunotherapy. Curr. Prob. Cancer 40, 68–83 (2016).
Konstorum, A., Vella, A. T., Adler, A. J. & Laubenbacher, R. C. Addressing current challenges in cancer immunotherapy with mathematical and computational modelling. J. R. Soc. Interface 14, 20170150 (2017).
Luksza, M. et al. A neoantigen fitness model predicts tumour response to checkpoint blockade immunotherapy. Nature 551, 517–520 (2017).
Kim, E., Rebecca, V. W., Smalley, K. S. & Anderson, A. R. Phase I trials in melanoma: a framework to translate preclinical findings to the clinic. Eur. J. Cancer 67, 213–222 (2016).
Cyll, K. et al. Tumour heterogeneity poses a significant challenge to cancer biomarker research. Br. J. Cancer 117, 367–375 (2017).
Powles, T. et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 515, 558–562 (2014).
Motzer, R. J. et al. Nivolumab for metastatic renal cell carcinoma: results of a randomized phase II trial. J. Clin. Oncol. 33, 1430–1437 (2015).
Borghaei, H. et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N. Engl. J. Med. 373, 1627–1639 (2015).
Plodinec, M. et al. The nanomechanical signature of breast cancer. Nat. Nanotechnol. 7, 757–765 (2012).
Pascal, J. et al. Mechanistic patient-specific predictive correlation of tumor drug response with microenvironment and perfusion measurements. Proc. Natl Acad. Sci. USA 110, 14266–14271 (2013).
Earle, C. C. et al. Aggressiveness of cancer care near the end of life: is it a quality-of-care issue? J. Clin. Oncol. 26, 3860–3866 (2008).
De Larco, J. E., Wuertz, B. R., Manivel, J. C. & Furcht, L. T. Progression and enhancement of metastatic potential after exposure of tumor cells to chemotherapeutic agents. Cancer Res. 61, 2857–2861 (2001).
Van Meir, H. et al. Impact of (chemo)radiotherapy on immune cell composition and function in cervical cancer patients. Oncoimmunology 6, e1267095 (2017).
Pascal, J. et al. Mechanistic modeling identifies drug-uptake history as predictor of tumor drug resistance and nano-carrier-mediated response. ACS Nano 7, 11174–11182 (2013).
Das, H. et al. Impact of diffusion barriers to small cytotoxic molecules on the efficacy of immunotherapy in breast cancer. PLoS ONE 8, e61398 (2013).
Koay, E. J. et al. Transport properties of pancreatic cancer describe gemcitabine delivery and response. J. Clin. Invest. 124, 1525–1536 (2014).
Frieboes, H. B., Wu, M., Lowengrub, J., Decuzzi, P. & Cristini, V. A computational model for predicting nanoparticle accumulation in tumor vasculature. PLoS ONE 8, e56876 (2013).
Wang, Z. et al. Theory and experimental validation of a spatio-temporal model of chemotherapy transport to enhance tumor cell kill. PLoS Comput. Biol. 12, e1004969 (2016).
Brocato, T. A. et al. Understanding the connection between nanoparticle uptake and cancer treatment efficacy using mathematical modeling. Sci. Rep. 8, 7538 (2018).
Koay, E. J. et al. A visually apparent and quantifiable CT imaging feature identifies biophysical subtypes of pancreatic ductal adenocarcinoma. Clin. Cancer Res. 24, 5883–5894 (2018).
Dogra, P. et al. Establishing the effects of mesoporous silica nanoparticle properties on in vivo disposition using imaging-based pharmacokinetics. Nat. Commun. 9, 4551 (2018).
Brocato, T. A. et al. Predicting breast cancer response to neoadjuvant chemotherapy based on tumor vascular features in needle biopsies.JCI Insight 5, e126518 (2019).
Goel, S. et al. Size-optimized ultrasmall porous silica nanoparticles depict vasculature-based differential targeting in triple negative breast cancer. Small 15, e1903747 (2019).
Pichler, M. et al. Therapeutic potential of FLANC, a novel primate-specific long non-coding RNA in colorectal cancer.Gut 69, 1818–1831 (2020).
Butner, J. D. et al. A multiscale agent-based model of ductal carcinoma in situ. IEEE Trans. Biomed. Eng. 67, 1450–1461 (2020).
Butner, J. D. et al. Mathematical prediction of clinical outcomes in advanced cancer patients treated with checkpoint inhibitor immunotherapy.Sci. Adv. 6, eaay6298 (2020).
Dogra, P. et al. A mathematical model to predict nanomedicine pharmacokinetics and tumor delivery. Comput. Struct. Biotechnol. J. 18, 518–531 (2020).
Rohatgi, A. WebPlotDigitizer v.4.0 (2017).
Mathematica v.11.2 (Wolfram Research, 2017).
Acknowledgements
The research reported in this manuscript was supported by the National Science Foundation grant DMS-1930583 (to Z.W. and V.C.), National Institutes of Health grants 1U01CA196403 (to Z.W., E.J.K. and V.C.), 1U01CA213759 (to Z.W. and V.C.), 1R01CA226537 (to Z.W. and V.C.), 1R01CA222007 (to Z.W., G.A.C. and V.C.) and U54CA210181 (to Z.W., E.J.K. and V.C.) and the University of Texas System STARs Award (to V.C.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
J.D.B. implemented the model and collected the data. Z.W. and V.C. initiated the project, participated in data collection and supervised the model development and analysis. J.D.B., Z.W., M.P., P.D. and V.C. performed the model analysis. D.E., K.A.A.F., G.V.M., C.C., E.J.K., J.W.W. and D.S.H. participated in data collection. J.D.B., Z.W., D.E., K.A.A.F., M.P., G.A.C., P.D., S.N., J.R.-R., G.V.M., H.A.T., C.C., E.J.K., J.W.W., D.S.H. and V.C. interpreted the data and model results. J.D.B., Z.W., M.P., E.J.K. and V.C. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.W.W. reports research support from GlaxoSmithKline, Bristol Myers Squibb, Merck, Nanobiotix, Mavupharma and Checkmate Pharmaceuticals. J.W.W. serves on the scientific advisory board for RefleXion Medical, MolecularMatch, OncoResponse, Checkmate, Mavupharmaceuticals and Alpine Immune Sciences. J.W.W. is co-founder of Healios Oncology, MolecularMatch and OncoResponse and serves as an advisor to AstraZeneca, Merck, MolecularMatch, Incyte, Aileron and Nanobiotix. J.W.W. has the following patents: MP470 (amuvatinib), MRX34 regulation of PD-L1, and the X-ray tomography technique to overcome immune resistance. The University of Texas MD Anderson Cancer Center has a trademark for RadScopal.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Discussion, Figs. 1–10, Tables 1–5 and references.
Rights and permissions
About this article
Cite this article
Butner, J.D., Wang, Z., Elganainy, D. et al. A mathematical model for the quantification of a patient’s sensitivity to checkpoint inhibitors and long-term tumour burden. Nat Biomed Eng 5, 297–308 (2021). https://doi.org/10.1038/s41551-020-00662-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-00662-0
This article is cited by
-
Global stability and parameter analysis reinforce therapeutic targets of PD-L1-PD-1 and MDSCs for glioblastoma
Journal of Mathematical Biology (2024)
-
On fast simulation of dynamical system with neural vector enhanced numerical solver
Scientific Reports (2023)
-
Harnessing progress in radiotherapy for global cancer control
Nature Cancer (2023)
-
Mathematical modeling of cancer immunotherapy for personalized clinical translation
Nature Computational Science (2022)
-
Dedifferentiation-mediated stem cell niche maintenance in early-stage ductal carcinoma in situ progression: insights from a multiscale modeling study
Cell Death & Disease (2022)