Abstract
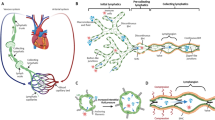
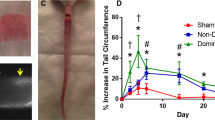
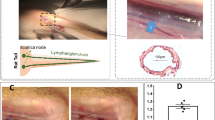
Contractile activity in the lymphatic vasculature is essential for maintaining fluid balance within organs and tissues. However, the mechanisms by which collecting lymphatics adapt to changes in fluid load and how these adaptations influence lymphatic contractile activity are unknown. Here we report a model of lymphatic injury based on the ligation of one of two parallel lymphatic vessels in the hind limb of sheep and the evaluation of structural and functional changes in the intact, remodelling lymphatic vessel over a 42-day period. We show that the remodelled lymphatic vessel displayed increasing intrinsic contractile frequency, force generation and vessel compliance, as well as decreasing flow-mediated contractile inhibition via the enzyme endothelial nitric oxide synthase. A computational model of a chain of lymphatic contractile segments incorporating these adaptations predicted increases in the flow-generation capacity of the remodelled vessel at the expense of normal mitochondrial function and elevated oxidative stress within the lymphatic muscle. Our findings may inform interventions for mitigating lymphatic muscle fatigue in patients with dysfunctional lymphatics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The authors declare that all data supporting the results in this study are available in the paper and Supplementary Information. Source data for the figures in this study are available on reasonable request. The mass spectrometry proteomics data have been deposited with the ProteomeXchange Consortium via the PRIDE partner repository64 with the dataset identifier PXD014343.
Code availability
Matlab code for the computational modelling and for the analysis of NIR videos is available at https://github.com/LLBB-GT/NBE-2019. Any additional custom codes (LabView and Matlab) used in this study are available from the authors on reasonable request.
References
Levick, J. R. & Michel, C. C. Microvascular fluid exchange and the revised Starling principle. Cardiovasc. Res. 87, 198–210 (2010).
Miteva, D. O. et al. Transmural flow modulates cell and fluid transport functions of lymphatic endothelium. Circ. Res. 106, 920–931 (2010).
Clement, C. C. et al. The dendritic cell major histocompatibility complex II (MHC II) peptidome derives from a variety of processing pathways and includes peptides with a broad spectrum of HLA-DM sensitivity. J. Biol. Chem. 291, 5576–5595 (2016).
Clement, C. C. & Santambrogio, L. The lymph self-antigen repertoire. Front. Immun. 4, 424 (2013).
Kassis, T. et al. Postprandial lymphatic pump function after a high-fat meal: a characterization of contractility, flow, and viscosity. Am. J. Physiol. Gastrointest. Liver Physiol. 310, G776–G789 (2016).
Zawieja, D. C. Contractile physiology of lymphatics. Lymphat. Res. Biol. 7, 87–96 (2009).
Mihara, M. et al. Pathological steps of cancer-related lymphedema: histological changes in the collecting lymphatic vessels after lymphadenectomy. PLoS ONE 7, e41126 (2012).
DiSipio, T., Rye, S., Newman, B. & Hayes, S. Incidence of unilateral arm lymphoedema after breast cancer: a systematic review and meta-analysis. Lancet Oncol. 14, 500–515 (2013).
Avraham, T. et al. Fibrosis is a key inhibitor of lymphatic regeneration. Plast. Reconstr. Surg. 124, 438–450 (2009).
Gousopoulos, E. et al. An important role of VEGF-C in promoting lymphedema development. J. Investig. Dermatol. 137, 1995–2004 (2017).
Rutkowski, J. M., Moya, M., Johannes, J., Goldman, J. & Swartz, M. A. Secondary lymphedema in the mouse tail: lymphatic hyperplasia, VEGF-C upregulation, and the protective role of MMP-9. Microvasc. Res. 72, 161–171 (2006).
Armer, J. M. et al. Lymphedema symptoms and limb measurement changes in breast cancer survivors treated with neoadjuvant chemotherapy and axillary dissection: results of American College of Surgeons Oncology Group (ACOSOG) Z1071 (Alliance) substudy. Support. Care Cancer 52, 370–379 (2018).
Kornuta, J. A. & Dixon, J. B. Ex vivo lymphatic perfusion system for independently controlling pressure gradient and transmural pressure in isolated vessels. Ann. Biomed. Eng. 42, 1691–1704 (2014).
Kornuta, J. A. et al. Effects of dynamic shear and transmural pressure on wall shear stress sensitivity in collecting lymphatic vessels. Am. J. Physiol. Regul. Integr. Comp. Physiol. 309, R1122–R1134 (2015).
Scuteri, A. et al. Metabolic syndrome amplifies the age-associated increases in vascular thickness and stiffness. J. Am. Coll. Cardiol. 43, 1388–1395 (2004).
Nichols, W. W. Clinical measurement of arterial stiffness obtained from noninvasive pressure waveforms. Am. J. Hypertens. 18, 3S–10S (2005).
Huang, G. K. & Maekawa, J. A new experimental model of lymphedema in rats’ hind lower legs. Eur. J. Plast. Surg. 13, 192–197 (1990).
Olszewski, W., Machowski, Z., Sokolowski, J. & Nielubowicz, J. Experimental lymphedema in dogs. J. Cardiovasc. Surg. 9, 178–183 (1968).
Slavin, S. A., Van den Abbeele, A. D., Losken, A., Swartz, M. A. & Jain, R. K. Return of lymphatic function after flap transfer for acute lymphedema. Ann. Surg. 229, 421–427 (1999).
Tobbia, D. et al. Lymphedema development and lymphatic function following lymph node excision in sheep. J. Vasc. Res. 46, 426–434 (2009).
Wiig, H. & Swartz, M. A. Interstitial fluid and lymph formation and transport: physiological regulation and roles in inflammation and cancer. Physiol. Rev. 92, 1005–1060 (2012).
Adair, T. H., Vance, G. A., Montani, J. P. & Guyton, A. C. Effect of skin concavity on subcutaneous tissue fluid pressure. Am. J. Physiol. Heart Circ. Physiol. 261, H349–H353 (1991).
Blatter, C. et al. In vivo label-free measurement of lymph flow velocity and volumetric flow rates using Doppler optical coherence tomography. Sci. Rep. 6, 29035 (2016).
Davis, M. J., Davis, A. M., Ku, C. W. & Gashev, A. A. Myogenic constriction and dilation of isolated lymphatic vessels. Am. J. Physiol. Heart Circ. Physiol. 296, H293–H302 (2009).
Davis, M. J., Davis, A. M., Lane, M. M., Ku, C. W. & Gashev, A. A. Rate-sensitive contractile responses of lymphatic vessels to circumferential stretch. J. Physiol. 587, 165–182 (2009).
Jones, D. et al. Methicillin-resistant Staphylococcus aureus causes sustained collecting lymphatic vessel dysfunction. Sci. Transl. Med. 10, eaam7964 (2018).
Weiler, M. J., Cribb, M. T., NepiyushchikhZ., Nelson, T. S. & Dixon, B. A novel mouse tail lymphedema model for observing lymphatic pump failure during lymphedema development. Sci. Rep. (2019).
Ballinger, S. W. et al. Hydrogen peroxide– and peroxynitrite-induced mitochondrial DNA damage and dysfunction in vascular endothelial and smooth muscle cells. Circ. Res. 86, 960–966 (2000).
Custodis, F. et al. Heart rate reduction by ivabradine improves aortic compliance in apolipoprotein E-deficient mice. J. Vasc. Res. 49, 432–440 (2012).
Thangaswamy, S., Bridenbaugh, E. A. & Gashev, A. A. Evidence of increased oxidative stress in aged mesenteric lymphatic vessels. Lymphat. Res. Biol. 10, 53–62 (2012).
Bohlen, H. G., Gasheva, O. Y. & Zawieja, D. C. Nitric oxide formation by lymphatic bulb and valves is a major regulatory component of lymphatic pumping. Am. J. Physiol. Heart Circ. Physiol. 301, H1897–H1906 (2011).
Gashev, A. A. & Zawieja, D. C. Hydrodynamic regulation of lymphatic transport and the impact of aging. Pathophysiology 17, 277–287 (2010).
Quick, C. M., Venugopal, A. M., Gashev, A. A., Zawieja, D. C. & Stewart, R. H. Intrinsic pump-conduit behavior of lymphangions. Am. J. Physiol. Regul. Integr. Comp. Physiol. 292, R1510–R1518 (2007).
Scallan, J. P. & Davis, M. J. Genetic removal of basal nitric oxide enhances contractile activity in isolated murine collecting lymphatic vessels. J. Physiol. 591, 2139–2156 (2013).
Griffith, O. W. & Kilbourn, R. G. Nitric oxide synthase inhibitors: amino acids. Meth. Enzymol. 268, 375–392 (1995).
Víteček, J., Lojek, A., Valacchi, G. & Kubala, L. Arginine-based inhibitors of nitric oxide synthase: therapeutic potential and challenges. Mediators Inflamm. 2012, 318087 (2012).
Gashev, A. A., Davis, M. J. & Zawieja, D. C. Inhibition of the active lymph pump by flow in rat mesenteric lymphatics and thoracic duct. J. Physiol. 540, 1023–1037 (2002).
Lu, D. & Kassab, G. S. Role of shear stress and stretch in vascular mechanobiology. J. R. Soc. Interface 8, 1379–1385 (2011).
Datar, S. A. et al. Altered reactivity and nitric oxide signaling in the isolated thoracic duct from an ovine model of congenital heart disease with increased pulmonary blood flow. Am. J. Physiol. Heart Circ. Physiol. 306, H954–H962 (2014).
Datar, S. A. et al. Disrupted NOS signaling in lymphatic endothelial cells exposed to chronically increased pulmonary lymph flow. Am. J. Physiol. Heart Circ. Physiol. 311, H137–H145 (2016).
Razavi, M., Nelson, T. S., Nepiyushchikh, Z., Gleason, R. L. & Dixon, B. The relationship between lymphangion chain length and maximum pressure generation established through in vivo imaging and computational modeling. Am. J. Physiol. Heart Circ. Physiol. 313, H1249–H1260 (2017).
Dongaonkar, R. M. et al. Adaptation of mesenteric lymphatic vessels to prolonged changes in transmural pressure. Am. J. Physiol. Heart Circ. Physiol. 305, H203–H210 (2013).
Quick, C. M., Ngo, B. L., Venugopal, A. M. & Stewart, R. H. Lymphatic pump-conduit duality: contraction of postnodal lymphatic vessels inhibits passive flow. Am. J. Physiol. Heart Circ. Physiol. 296, H662–H668 (2009).
Baish, J. W., Kunert, C., Padera, T. P. & Munn, L. L. Synchronization and random triggering of lymphatic vessel contractions. PLoS Comp. Biol. 12, e1005231 (2016).
Kunert, C., Baish, J. W., Liao, S., Padera, T. P. & Munn, L. L. Mechanobiological oscillators control lymph flow. Proc. Natl Acad. Sci. USA 112, 10938–10943 (2015).
Zawieja, S. D., Castorena-Gonzalez, J. A., Dixon, B. & Davis, M. J. Experimental models used to assess lymphatic contractile function. Lymphat. Res. Biol. 15, 331–342 (2017).
O’Mahony, S. et al. Imaging of lymphatic vessels in breast cancer–related lymphedema: intradermal versus subcutaneous injection of 99mTc-immunoglobulin. Am. J. Roentgenol. 186, 1349–1355 (2012).
Bains, S. K. et al. A constitutional predisposition to breast cancer-related lymphoedema and effect of axillary lymph node surgery on forearm muscle lymph flow. Breast 24, 68–74 (2014).
Cintolesi, V. et al. Constitutively enhanced lymphatic pumping in the upper limbs of women who later develop breast cancer-related lymphedema. Lymphat. Res. Biol. 14, 50–61 (2016).
Armer, J. M. & Stewart, B. R. Post-breast cancer lymphedema: incidence increases from 12 to 30 to 60 months. Lymphology 43, 118–127 (2010).
Pires, P. W., Deutsch, C., McClain, J. L., Rogers, C. T. & Dorrance, A. M. Tempol, a superoxide dismutase mimetic, prevents cerebral vessel remodeling in hypertensive rats. Microvasc. Res. 80, 445–452 (2010).
Rossman, M. J. et al. Chronic supplementation with a mitochondrial antioxidant (MitoQ) improves vascular function in healthy older adults. Hypertension 71, 1056–1063 (2018).
Santos-Parker, J. R. et al. Curcumin supplementation improves vascular endothelial function in healthy middle-aged and older adults by increasing nitric oxide bioavailability and reducing oxidative stress. Aging 9, 187–208 (2016).
Nelson, T. S. et al. Minimally invasive method for determining the effective lymphatic pumping pressure in rats using near-infrared imaging. Am. J. Physiol. Regul. Integr. Comp. Physiol. 306, R281–R290 (2014).
Weiler, M., Kassis, T. & Dixon, B. Sensitivity analysis of near infrared functional lymphatic imaging. J. Biomed. Opt. 17, 066019 (2012).
Weiler, M. & Dixon, B. Differential transport function of lymphatic vessels in the rat tail model and the long-term effects of Indocyanine Green as assessed with near-infrared imaging. Front. Physiol. 4, 215 (2013).
Blum, K. S., Proulx, S. T., Luciani, P., Leroux, J.-C. & Detmar, M. Dynamics of lymphatic regeneration and flow patterns after lymph node dissection. Breast Cancer Res. Treat. 139, 81–86 (2013).
Proulx, S. T. et al. Use of a PEG-conjugated bright near-infrared dye for functional imaging of rerouting of tumor lymphatic drainage after sentinel lymph node metastasis. Biomaterials 34, 5128–5137 (2013).
Chong, C. et al. In vivo visualization and quantification of collecting lymphatic vessel contractility using near-infrared imaging. Sci. Rep. 6, 22930 (2016).
Mukherjee, A., Hooks, J., Nepiyushchikh, Z. & Dixon, B. Entrainment of lymphatic contraction to oscillatory flow. Sci. Rep. 9, 5840 (2019).
Chelko, S. P., Schmiedt, C. W., Lewis, T. H., Lewis, S. J. & Robertson, T. P. Vasopressin-induced constriction of the isolated rat occipital artery is segment dependent. J. Vasc. Res. 50, 478–485 (2013).
Clement, C. C. et al. Protein expression profiles of human lymph and plasma mapped by 2D-DIGE and 1D SDS–PAGE coupled with nanoLC–ESI–MS/MS bottom-up proteomics. J. Proteom. 78, 172–187 (2013).
Ovis aries (SwissProt, accessed April 2017); https://www.uniprot.org/uniprot/?query=reviewed:yes
Perez-Riverol, Y. et al. The PRIDE database and related tools and resources in 2019: improving support for quantification data. Nucleic Acids Res. 47, D442–D450 (2019).
Gleason, R. L., Gray, S. P., Wilson, E. & Humphrey, J. D. A multiaxial computer-controlled organ culture and biomechanical device for mouse carotid arteries. J. Biomech. Eng. 126, 787–795 (2004).
Caulk, A. W., Nepiyushchikh, Z. V., Shaw, R., Dixon, B. & Gleason, R. L. Quantification of the passive and active biaxial mechanical behaviour and microstructural organization of rat thoracic ducts. J. R. Soc. Interface 12, 20150280 (2015).
Caulk, A. W., Dixon, B. & Gleason, R. L. A lumped parameter model of mechanically mediated acute and long-term adaptations of contractility and geometry in lymphatics for characterization of lymphedema. Biomech. Model. Mechanobiol. 15, 1601–1618 (2016).
Dixon, B. et al. Lymph flow, shear stress, and lymphocyte velocity in rat mesenteric prenodal lymphatics. Microcirculation 13, 597–610 (2006).
Bertram, C. D., Macaskill, C., Davis, M. J. & Moore, J. E. Development of a model of a multi-lymphangion lymphatic vessel incorporating realistic and measured parameter values. Biomech. Model. Mechanobiol. 13, 401–416 (2014).
Bertram, C. D., Macaskill, C., Davis, M. J. & Moore, J. E. Consequences of intravascular lymphatic valve properties: a study of contraction timing in a multi-lymphangion model. Am. J. Physiol. Heart Circ. Physiol. 310, H847–H860 (2016).
Bertram, C. D., Macaskill, C. & Moore, J. E. Simulation of a chain of collapsible contracting lymphangions with progressive valve closure. J. Biomech. Eng. 133, 011008 (2011).
Jamalian, S., Bertram, C. D., Richardson, W. J. & Moore, J. E. Parameter sensitivity analysis of a lumped-parameter model of a chain of lymphangions in series. Am. J. Physiol. Heart Circ. Physiol. 305, H1709–H1717 (2013).
Holzapfel, G. A., Gasser, T. C. & Ogden, R. W. A new constitutive framework for arterial wall mechanics and a comparative study of material models. J. Elast. 61, 1–48 (2000).
Acknowledgements
The authors acknowledge the following individuals at the at the University of Georgia for their contributions to this study: A. Bullington, for assistance with the care and handling of the sheep; M. Barletta, for conducting and developing the anaesthesia protocol; S. P. Holmes, for conducting the MRI scans. The authors acknowledge the following funding sources that supported this project: Regenerative Engineering and Medicine (REM) Seed Grant (to J.B.D. and J.P.), NIH RO1HL113061 (to J.B.D.) and American Heart Association (AHA) Predoctoral Fellowship 16PRE27390011 (to T.N.).
Author information
Authors and Affiliations
Contributions
T.S.N., Z.N., R.L.G.Jr, J.F.P. and J.B.D. conceived the concept and designed the experiments. T.S.N., Z.N., J.S.T.H., T.L., M.T.C. and M.R. developed the methods. T.S.N., Z.N., J.S.T.H., T.L., M.T. and J.F.P. performed the experiments. T.S.N., Z.N., J.S.T.H., M.R., C.C.C. and L.S. analysed the data. T.S.N., Z.N., J.S.T.H. and J.B.D. prepared and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary figures and tables.
Supplementary Video 1
Rotating movie of 3D MRI lymphangiography of the drainage pathways of a sheep prior to surgery. Five independent experiments were performed with similar results.
Supplementary Video 2
Movie demonstrating surgical excision of lymphatic vessel via NIR guidance. Five independent experiments were performed with similar results.
Rights and permissions
About this article
Cite this article
Nelson, T.S., Nepiyushchikh, Z., Hooks, J.S.T. et al. Lymphatic remodelling in response to lymphatic injury in the hind limbs of sheep. Nat Biomed Eng 4, 649–661 (2020). https://doi.org/10.1038/s41551-019-0493-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0493-1
This article is cited by
-
A 1D model characterizing the role of spatiotemporal contraction distributions on lymph transport
Scientific Reports (2023)