Abstract
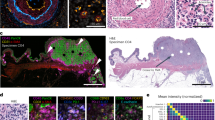
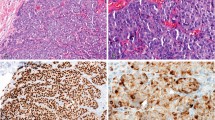
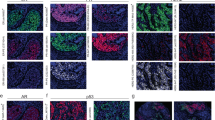
Immunohistochemistry is the gold-standard method for cancer-biomarker identification and patient stratification. Yet, owing to signal saturation, its use as a quantitative assay is limited as it cannot distinguish tumours with similar biomarker-expression levels. Here, we introduce a quantitative microimmunochemistry assay that enables the acquisition of dynamic information, via a metric of the evolution of the immunohistochemistry signal during tissue staining, for the quantification of relative antigen density on tissue surfaces. We used the assay to stratify 30 patient-derived breast-cancer samples into conventional classes and to determine the proximity of each sample to the other classes. We also show that the assay enables the quantification of multiple biomarkers (human epidermal growth factor receptor, oestrogen receptor and progesterone receptor) in a standard breast-cancer panel. The integration of quantitative microimmunohistochemistry into current pathology workflows may lead to improvements in the precision of biomarker quantification.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information. The raw data generated in this study are available from the corresponding author on reasonable request.
Code availability
The custom Python code for the described algorithms is available on request.
References
Coons, A. H., Creech, H. J. & Jones, R. N. Immunological properties of an antibody containing a fluorescent group. Exp. Biol. Med. 47, 200–202 (1941).
Coons, A. H. & Kaplan, M. H. Localization of antigen in tissue cells: II. Improvements in a method for the detection of antigen by means of fluorescent antibody. J. Exp. Med. 91, 1–13 (1949).
Nakane, P. K. & Pierce, G. B. Enzyme-labeled antibodies: preparation and application for the localization of antigens. J. Histochem. Cytochem. 14, 929–931 (1966).
Nakane, P. Simultaneous localization of multiple tissue antigens using the peroxidase labeled antibody method: a study of pituitary glands of the rat. J. Histochem. Cytochem. 16, 557–560 (1968).
de Matos, L. L., Trufelli, D. C., de Matos, M. G. L. & da Silva Pinhal, M. A. Immunohistochemistry as an important tool in biomarkers detection and clinical practice. Biomark. Insights 5, 9–20 (2010).
de Gramont, A. et al. Pragmatic issues in biomarker evaluation for targeted therapies in cancer. Nat. Rev. Clin. Oncol. 12, 197–212 (2014).
Smith, I. et al. 2-year follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer: a randomised controlled trial. Lancet 369, 29–36 (2007).
Gámez-Pozo, A. et al. The Long-HER study: clinical and molecular analysis of patients with HER2+ advanced breast cancer who become long-term survivors with trastuzumab-based therapy. PLoS ONE 9, e109611 (2014).
Zacharakis, N. et al. Immune recognition of somatic mutations leading to complete durable regression in metastatic breast cancer. Nat. Med. 24, 724–730 (2018).
Mason, J. T., Fowler, C. B. & O’leary, T. J. In Antigen Retrieval Immunohistochemistry Based Research and Diagnostics (eds Shi, S.-R. & Taylor, C. R.) 251–285 (John Wiley & Sons, Inc., 2010).
Kunz, P. et al. Osteosarcoma microenvironment: whole-slide imaging and optimized antigen detection overcome major limitations in immunohistochemical quantification. PLoS ONE 9, e90727 (2014).
Sabattini, E. et al. The EnVision++system: a new immunohistochemical method for diagnostics and research. Critical comparison with the APAAP, ChemMate, CSA, LABC, and SABC techniques. J. Clin. Pathol. 51, 506–511 (1998).
Wu, X. et al. Immunofluorescent labeling of cancer marker Her2 and other cellular targets with semiconductor quantum dots. Nat. Biotechnol. 21, 41–46 (2003).
Barrow, E., Evans, D. G., McMahon, R., Hill, J. & Byers, R. A comparative study of quantitative immunohistochemistry and quantum dot immunohistochemistry for mutation carrier identification in Lynch syndrome. J. Clin. Pathol. 64, 208–214 (2011).
Kwon, S., Cho, C. H., Lee, E. S. & Park, J.-K. Automated measurement of multiple cancer biomarkers using quantum-dot-based microfluidic immunohistochemistry. Anal. Chem. 87, 4177–4183 (2015).
Zaha, D. C. Significance of immunohistochemistry in breast cancer. World J. Clin. Oncol. 5, 382–392 (2014).
Wolff, A. C. et al. American society of clinical oncology/college of American pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J. Clin. Oncol. 25, 118–145 (2006).
Allred, D. et al. Immunocytochemical analysis of estrogen receptors in human breast carcinomas. Evaluation of 130 cases and review of the literature regarding concordance with biochemical assay and clinical relevance. Arch. Surg. 125, 107–113 (1990).
Carlson, R. W. et al. HER2 testing in breast cancer: NCCN Task Force report and recommendations. J. Natl Compr. Canc. Netw. 4, S1–S22 (2006).
Rhodes, A. et al. A formalin-fixed, paraffin-processed cell line standard for quality control of immunohistochemical assay of HER-2/neu expression in breast cancer. Am. J. Clin. Pathol. 117, 81–89 (2002).
Vyberg, M. & Nielsen, S. Proficiency testing in immunohistochemistry—experiences from Nordic Immunohistochemical Quality Control (NordiQC). Virchows Arch. 468, 19–29 (2016).
Grube, D. Constants and variables in immunohistochemistry. Arch. Histol. Cytol. 67, 115–134 (2004).
Taylor, C. R. & Levenson, R. M. Quantification of immunohistochemistry—issues concerning methods, utility and semiquantitative assessment II. Histopathology 49, 411–424 (2006).
Taylor, C. R. Predictive biomarkers and companion diagnostics. The future of immunohistochemistry. Appl. Immunohistochem. Mol. Morphol. 22, 555–561 (2014).
Rizzardi, A. E. et al. Quantitative comparison of immunohistochemical staining measured by digital image analysis versus pathologist visual scoring. Diagn. Pathol. 7, 42 (2012).
Hall, B. H. et al. Computer-assisted assessment of the human epidermal growth factor receptor 2 immunohistochemical assay in imaged histologic sections using a membrane isolation algorithm and quantitative analysis of positive controls. BMC Med. Imaging 8, 11 (2008).
Arar, N. M. et al. Computational immunohistochemistry: recipes for standardization of immunostaining. In Int. Conf. Medical Image Computing and Computer-Assisted Intervention (eds Descoteaux, M. et al.) 48–55 (2017).
O’Hurley, G. et al. Garbage in, garbage out: a critical evaluation of strategies used for validation of immunohistochemical biomarkers. Mol. Oncol. 8, 783–798 (2014).
Hammond, M. E. H. et al. American society of clinical oncology/college of American pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer (unabridged version). Arch. Pathol. Lab. Med. 134, e48–e72 (2010).
Goldstein, N. S. et al. Recommendations for improved standardization of immunohistochemistry. Appl. Immunohistochem. Mol. Morphol. 15, 124–133 (2007).
Brügmann, A. et al. Digital image analysis of membrane connectivity is a robust measure of HER2 immunostains. Breast Cancer Res. Treat. 132, 41–49 (2012).
Masmoudi, H., Hewitt, S. M., Petrick, N., Myers, K. J. & Gavrielides, M. A. Automated quantitative assessment of HER-2/neu immunohistochemical expression in breast cancer. IEEE Trans. Med. Imaging 28, 916–925 (2009).
Camp, R. L., Chung, G. G. & Rimm, D. L. Automated subcellular localization and quantification of protein expression in tissue microarrays. Nat. Med. 8, 1323–1328 (2002).
Camp, R. L. X-Tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin. Cancer Res. 10, 7252–7259 (2004).
Veta, M., Pluim, J. P. W., van Diest, P. J. & Viergever, M. A. Breast cancer histopathology image analysis: a review. IEEE Trans. Biomed. Eng. 61, 1400–1411 (2014).
Zhang, B. et al. Proteogenomic characterization of human colon and rectal cancer. Nature 513, 382–387 (2014).
Dong, F. et al. Computational pathology to discriminate benign from malignant intraductal proliferations of the breast. PLoS ONE 9, e114885 (2014).
Djuric, U., Zadeh, G., Aldape, K. & Diamandis, P. Precision histology: how deep learning is poised to revitalize histomorphology for personalized cancer care. NPJ Precis. Oncol. 1, 22 (2017).
Harigopal, M. et al. Multiplexed assessment of the southwest oncology group-directed intergroup breast cancer trial S9313 by AQUA shows that both high and low levels of HER2 are associated with poor outcome. Am. J. Pathol. 176, 1639–1647 (2010).
Potts, S. J. et al. Evaluating tumour heterogeneity in immunohistochemistry-stained breast cancer tissue. Lab. Invest. 92, 1342–1357 (2012).
Zrazhevskiy, P., True, L. D. & Gao, X. Multicolor multicycle molecular profiling with quantum dots for single-cell analysis. Nat. Protoc. 8, 1852–1869 (2013).
Vu, T. Q., Lam, W. Y., Hatch, E. W. & Lidke, D. S. Quantum dots for quantitative imaging: from single molecules to tissue. Cell Tissue Res. 360, 71–86 (2015).
Giesen, C. et al. Highly multiplexed imaging of tumour tissues with subcellular resolution by mass cytometry. Nat. Methods 11, 417–422 (2014).
Ciftlik, A. T., Lehr, H.-A. & Gijs, M. A. M. Microfluidic processor allows rapid HER2 immunohistochemistry of breast carcinomas and significantly reduces ambiguous (2+) read-outs. Proc. Natl Acad. Sci. USA 110, 5363–5368 (2013).
Juncker, D., Schmid, H. & Delamarche, E. Multipurpose microfluidic probe. Nat. Mater. 4, 622–628 (2005).
Kaigala, G. V., Lovchik, R. D., Drechsler, U. & Delamarche, E. A vertical microfluidic probe. Langmuir 27, 5686–5693 (2011).
Lovchik, R. D., Kaigala, G. V., Georgiadis, M. & Delamarche, E. Micro-immunohistochemistry using a microfluidic probe. Lab Chip 12, 1040–1043 (2012).
Kashyap, A., Autebert, J., Delamarche, E. & Kaigala, G. V. Selective local lysis and sampling of live cells for nucleic acid analysis using a microfluidic probe. Sci. Rep. 6, 29579 (2016).
Huber, D., Autebert, J. & Kaigala, G. V. Micro fluorescence in situ hybridization (μFISH) for spatially multiplexed analysis of a cell monolayer. Biomed. Microdevices 18, 40 (2016).
Sarkar, A., Kolitz, S., Lauffenburger, D. A. & Han, J. Microfluidic probe for single-cell analysis in adherent tissue culture. Nat. Commun. 5, 3421 (2014).
Ainla, A., Jansson, E. T., Stepanyants, N., Orwar, O. & Jesorka, A. A microfluidic pipette for single-cell pharmacology. Anal. Chem. 82, 4529–4536 (2010).
Ainla, A., Xu, S., Sanchez, N., Jeffries, G. D. M. & Jesorka, A. Single-cell electroporation using a multifunctional pipette. Lab Chip 12, 4605–4609 (2012).
Kaigala, G. V., Lovchik, R. D. & Delamarche, E. Microfluidics in the ‘Open Space’ for performing localized chemistry on biological interfaces. Angew. Chem. Int. Ed. 51, 11224–11240 (2012).
Delamarche, E. & Kaigala, G. V. (eds) Open-Space Microfluidics: Concepts, Implementations, Applications (Wiley, 2018).
Squires, T. M., Messinger, R. J. & Manalis, S. R. Making it stick: convection, reaction and diffusion in surface-based biosensors. Nat. Biotechnol. 26, 417–426 (2008).
Autebert, J., Cors, J., Taylor, D. & Kaigala, G. V. Convection-enhanced biopatterning with hydrodynamically confined nanoliter volumes of reagents. Anal. Chem. 88, 3235–3242 (2016).
De Michele, C., De Los Rios, P., Foffi, G. & Piazza, F. Simulation and theory of antibody binding to crowded antigen-covered surfaces. PLoS Comput. Biol. 12, e1004752 (2016).
Thurber, G. M., Schmidt, M. M. & Wittrup, K. D. Antibody tumour penetration: transport opposed by systemic and antigen-mediated clearance. Adv. Drug Deliv. Rev. 60, 1421–1434 (2008).
Worthylake, R., Opresko, L. K. & Wiley, H. S. ErbB-2 amplification inhibits down-regulation and induces constitutive activation of both ErbB-2 and epidermal growth factor receptors. J. Biol. Chem. 274, 8865–8874 (1999).
Van Der Loos, C. M. Chromogens in multiple immunohistochemical staining used for visual assessment and spectral imaging: the colorful future. J. Histochem. 33, 31–40 (2010).
Andersson, E., Nie, Y., Roessler, C. & Grimm, O. Color deconvolution method with DAB scatter correction for bright field image analysis. In Medical Imaging 2018: Digital Pathology (eds Gurcan, M. N. & Tomaszewski, J. E.) 19 (SPIE, 2018).
Autebert, J., Cors, J. F., Taylor, D. P. & Kaigala, G. V. Convection-enhanced biopatterning with recirculation of hydrodynamically confined nanoliter volumes of reagents. Anal. Chem. 88, 3235–3242 (2016).
Dunnwald, L. K., Rossing, M. A. & Li, C. I. Hormone receptor status, tumour characteristics, and prognosis: a prospective cohort of breast cancer patients. Breast Cancer Res. 9, R6 (2007).
Weigelt, B., Peterse, J. L. & van’t Veer, L. J. Breast cancer metastasis: markers and models. Nat. Rev. Cancer 5, 591–602 (2005).
Kim, T. J. et al. Prognostic significance of high expression of ER-beta in surgically treated ER-positive breast cancer following endocrine therapy. J. Breast Cancer 15, 79–86 (2012).
Sompuram, S. R., Vani, K., Tracey, B., Kamstock, D. A. & Bogen, S. A. Standardizing immunohistochemistry. J. Histochem. Cytochem. 63, 681–690 (2015).
Taylor, C. R. & Shi, S.-R. Quantifiable internal reference standards for immunohistochemistry and uses thereof. US patent 2007/015417 (2008).
Acknowledgements
A.K., A.F.K. and G.V.K. are partly supported by a European Research Council Starting Grant, under the 7th Framework Program (project no. 311122, BioProbe). We thank L. Von Voithenberg, N. M. Arar, J. Cors, R. Lovchik, D. Taylor, I. Pereiro, J. Autebert and U. Drechsler for technical assistance and discussions. We thank Z. Varga (Pathology, University Hospital Zurich) for selecting anonymized breast-cancer samples and S. Dettwiler (Tissue Biobank, University Hospital Zurich) for technical assistance. P. Dittrich (ETH Zurich), A. deMello (ETH Zurich), O. Goksel (ETH Zurich), E. Delamarche and W. Riess are acknowledged for their continuous support.
Author information
Authors and Affiliations
Contributions
A.K., A.F.K., P.S. and G.V.K. designed the research project. A.F.K and A.K. performed the experiments. A.K., P.P. and M.G. designed and developed the analytical pipeline. A.K., A.F.K, P.P. and G.V.K. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, figures, tables and references.
Rights and permissions
About this article
Cite this article
Kashyap, A., Fomitcheva Khartchenko, A., Pati, P. et al. Quantitative microimmunohistochemistry for the grading of immunostains on tumour tissues. Nat Biomed Eng 3, 478–490 (2019). https://doi.org/10.1038/s41551-019-0386-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0386-3
This article is cited by
-
Rapid micro-immunohistochemistry
Microsystems & Nanoengineering (2020)