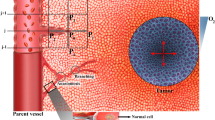
Abstract
Understanding the uptake of a drug by diseased tissue, and the drug’s subsequent spatiotemporal distribution, are central factors in the development of effective targeted therapies. However, the interaction between the pathophysiology of diseased tissue and individual therapeutic agents can be complex, and can vary across tissue types and across subjects. Here, we show that the combination of mathematical modelling, high-resolution optical imaging of intact and optically cleared tumour tissue from animal models, and in vivo imaging of vascular perfusion predicts the heterogeneous uptake, by large tissue samples, of specific therapeutic agents, as well as their spatiotemporal distribution. In particular, by using murine models of colorectal cancer and glioma, we report and validate predictions of steady-state blood flow and intravascular and interstitial fluid pressure in tumours, of the spatially heterogeneous uptake of chelated gadolinium by tumours, and of the effect of a vascular disrupting agent on tumour vasculature.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and its Supplementary Information. Raw data generated from this study can be found at https://doi.org/10.17605/OSF.IO/ZH9EU.
References
Brocato, T. et al. Understanding drug resistance in breast cancer with mathematical oncology. Curr. Breast Cancer Rep. 6, 110–120 (2014).
Dobosz, M., Ntziachristos, V., Scheuer, W. & Strobel, S. Multispectral fluorescence ultramicroscopy: three-dimensional visualization and automatic quantification of tumor morphology, drug penetration, and antiangiogenic treatment response. Neoplasia 16, 1–13 (2014).
Jahrling, N., Becker, K. & Dodt, H. U. 3D-reconstruction of blood vessels by ultramicroscopy. Organogenesis 5, 227–230 (2009).
Walls, J. R., Sled, J. G., Sharpe, J. & Henkelman, R. M. Resolution improvement in emission optical projection tomography. Phys. Med. Biol. 52, 2775–2790 (2007).
Alizadeh, A. A. et al. Toward understanding and exploiting tumor heterogeneity. Nat. Med. 21, 846–853 (2015).
Jain, R. K. Transport of molecules in the tumor interstitium: a review. Cancer Res. 47, 3039–3051 (1987).
Housman, G. et al. Drug resistance in cancer: an overview. Cancers 6, 1769–1792 (2014).
Gaya, A., Akle, C. A., Mudan, S. & Grange, J. The concept of hormesis in cancer therapy - is less more? Cureus 7, e261 (2015).
Johnson, S. P.et al. Acute changes in liver tumour perfusion measured non-invasively with arterial spin labelling. Br. J. Cancer 114, 897–904 2016).
El Emir, E. et al. Predicting response to radioimmunotherapy from the tumor microenvironment of colorectal carcinomas. Cancer Res. 67, 11896–11905 (2007).
Folarin, A. A., Konerding, M. A., Timonen, J., Nagl, S. & Pedley, R. B. Three-dimensional analysis of tumour vascular corrosion casts using stereoimaging and micro-computed tomography. Microvasc. Res. 80, 89–98 2010).
Walker-Samuel, S. et al. In vivo imaging of glucose uptake and metabolism in tumors. Nat. Med. 19, 1067–1072 (2013).
Rajkumar, V. S. et al. A comparative study of PDGFR inhibition with imatinib on radiolabeled antibody targeting and clearance in two pathologically distinct models of colon adenocarcinoma. Tumour Biol. 33, 2019–2029 (2012).
El-Emir, E. et al. Characterisation and radioimmunotherapy of L19-SIP, an anti-angiogenic antibody against the extra domain B of fibronectin, in colorectal tumour models. Br. J. Cancer 96, 1862–1870 (2007).
Lankester, K. J. et al. Combretastatin A-4-phosphate effectively increases tumor retention of the therapeutic antibody, 131I-A5B7, even at doses that are sub-optimal for vascular shut-down. Int. J. Oncol. 30, 453–460 (2007).
d’Esposito, A., Nikitichev, D., Desjardins, A., Walker-Samuel, S. & Lythgoe, M. F. Quantification of light attenuation in optically cleared mouse brains. J. Biomed. Opt. 20, 80503 (2015).
Frangi, A. F., Niessen, W. J., Vincken, K. L. & Viergever, M. A. in Medical Image Computing and Computer-Assisted Intervention—MICCAI’98 (eds Wells, W. M., Colchester A. & Delp, S.) 130–137 (Springer, Berlin, Heidelberg, 1998).
Thomas, D. L., Lythgoe, M. F., Pell, G. S., Calamante, F. & Ordidge, R. J. The measurement of diffusion and perfusion in biological systems using magnetic resonance imaging. Phys. Med. Biol. 45, R97–R138 (2000).
Konerding, M. A. et al. Evidence for characteristic vascular patterns in solid tumours: quantitative studies using corrosion casts. Br. J. Cancer 80, 724–732 (1999).
Walker-Samuel, S. et al. Investigating low-velocity fluid flow in tumours using convection-MRI. Cancer Res. 78, 1859–1872 (2018).
Walker-Samuel, S. et al. Investigating low-velocity fluid flow in tumors with convection-MRI. Cancer Res. 78, 1859–1872 (2018).
Yuan, F. et al. Vascular permeability in a human tumor xenograft: molecular size dependence and cutoff size. Cancer Res. 55, 3752–3756 (1995).
Reyes-Aldasoro, C. C. et al. Estimation of apparent tumor vascular permeability from multiphoton fluorescence microscopic images of P22 rat sarcomas in vivo. Microcirculation 15, 65–79 (2008).
Panagiotaki, E. et al. Noninvasive quantification of solid tumor microstructure using VERDICT MRI. Cancer Res. 74, 1902–1912 (2014).
Tofts, P. S. Modeling tracer kinetics in dynamic Gd-DTPA MR imaging. J. Magn. Reson. Imaging 7, 91–101 (1997).
Koh, T. S. et al. In vivo measurement of gadolinium diffusivity by dynamic contrast-enhanced MRI: a preclinical study of human xenografts. Magn. Reson. Med. 69, 269–276 (2013).
Benjaminsen, I. C., Graff, B. A., Brurberg, K. G. & Rofstad, E. K. Assessment of tumor blood perfusion by high-resolution dynamic contrast-enhanced MRI: a preclinical study of human melanoma xenografts. Magn. Reson. Med. 52, 269–276 (2004).
Sheng, Y. et al. Combretastatin family member OXi4503 induces tumor vascular collapse through the induction of endothelial apoptosis. Int. J. Cancer 111, 604–610 (2004).
Chan, L. S., Malcontenti-Wilson, C., Muralidharan, V. & Christophi, C. Alterations in vascular architecture and permeability following OXi4503 treatment. Anti-Cancer Drugs 19, 17–22 (2008).
Salmon, H. W., Mladinich, C. & Siemann, D. W. Evaluations of vascular disrupting agents CA4P and OXi4503 in renal cell carcinoma (Caki-1) using a silicon based microvascular casting technique. Eur. J. Cancer 42, 3073–3078 2006).
Salmon, H. W. & Siemann, D. W. Effect of the second-generation vascular disrupting agent OXi4503 on tumor vascularity. Clin. Cancer Res. 12, 4090–4094 (2006).
Wankhede, M., Dedeugd, C., Siemann, D. W. & Sorg, B. S. In vivo functional differences in microvascular response of 4T1 and Caki-1 tumors after treatment with OXi4503. Oncol. Rep. 23, 685–692 (2010).
El-Emir, E. et al. Tumour parameters affected by combretastatin A-4 phosphate therapy in a human colorectal xenograft model in nude mice. Eur. J. Cancer 41, 799–806 (2005).
Kirwan, I. G. et al. Comparative preclinical pharmacokinetic and metabolic studies of the combretastatin prodrugs combretastatin A4 phosphate and A1 phosphate. Clin. Cancer Res. 10, 1446–1453 (2004).
Byrne, H. M., Alarcon, T., Owen, M. R., Webb, S. D. & Maini, P. K. Modelling aspects of cancer dynamics: a review. Philos. Trans. A Math. Phys. Eng. Sci. 364, 1563–1578 2006).
Baxter, L. T. & Jain, R. K. Transport of fluid and macromolecules in tumors. I. Role of interstitial pressure and convection. Microvasc. Res. 37, 77–104 (1989).
Baxter, L. T. & Jain, R. K. Transport of fluid and macromolecules in tumors. II. Role of heterogeneous perfusion and lymphatics. Microvasc. Res. 40, 246–263 (1990).
Baxter, L. T. & Jain, R. K. Transport of fluid and macromolecules in tumors. III. Role of binding and metabolism. Microvasc. Res. 41, 5–23 (1991).
Altrock, P. M., Liu, L. L. & Michor, F. The mathematics of cancer: integrating quantitative models. Nat. Rev. Cancer 15, 730–745 (2015).
Beerenwinkel, N., Schwarz, R. F., Gerstung, M. & Markowetz, F. Cancer evolution: mathematical models and computational inference. Syst. Biol. 64, e1–e25 (2015).
Masoudi-Nejad, A. et al. Cancer systems biology and modeling: microscopic scale and multiscale approaches. Semin. Cancer Biol. 30, 60–69 (2015).
Boucher, Y., Baxter, L. T. & Jain, R. K. Interstitial pressure gradients in tissue-isolated and subcutaneous tumors: implications for therapy. Cancer Res. 50, 4478–4484 (1990).
Senthebane, D. A. et al. The role of tumor microenvironment in chemoresistance: to survive, keep your enemies closer. Int. J. Mol. Sci. 18, E1586 (2017).
Rubin, E. H. & Gilliland, D. G. Drug development and clinical trials—the path to an approved cancer drug. Nat. Rev. Clin. Oncol. 9, 215–222 (2012).
Jain, R. K. & Stylianopoulos, T. Delivering nanomedicine to solid tumors. Nat. Rev. Clin. Oncol. 7, 653–664 (2010).
Renier, N. et al. iDISCO: a simple, rapid method to immunolabel large tissue samples for volume imaging. Cell 159, 896–910 (2014).
Chung, K. et al. Structural and molecular interrogation of intact biological systems. Nature 497, 332–337 (2013).
Jain, R. K., Martin, J. D. & Stylianopoulos, T. The role of mechanical forces in tumor growth and therapy. Annu. Rev. Biomed. Eng. 16, 321–346 (2014).
Soltani, M. & Chen, P. Numerical modeling of interstitial fluid flow coupled with blood flow through a remodeled solid tumor microvascular network. PLoS ONE 8, e67025 (2013).
Sefidgar, M. et al. Numerical modeling of drug delivery in a dynamic solid tumor microvasculature. Microvasc. Res. 99, 43–56 (2015).
Mohammadi, M. & Chen, P. Effect of microvascular distribution and its density on interstitial fluid pressure in solid tumors: A computational model. Microvasc. Res. 101, 26–32 (2015).
Zhao, J., Salmon, H. & Sarntinoranont, M. Effect of heterogeneous vasculature on interstitial transport within a solid tumor. Microvasc. Res. 73, 224–236 (2007).
Baish, J. W. et al. Role of tumor vascular architecture in nutrient and drug delivery: an invasion percolation-based network model. Microvasc. Res. 51, 327–346 (1996).
Stamatelos, S. K., Kim, E., Pathak, A. P. & Popel, A. S. A bioimage informatics based reconstruction of breast tumor microvasculature with computational blood flow predictions. Microvasc. Res. 91, 8–21 (2014).
Boujelben, A. et al. Multimodality imaging and mathematical modelling of drug delivery to glioblastomas. Interface Focus 6, 20160039 (2016).
Thurber, G. M. et al. Single-cell and subcellular pharmacokinetic imaging allows insight into drug action in vivo. Nat. Commun. 4, 1504 (2013).
van de Ven, A. L. et al. Integrated intravital microscopy and mathematical modeling to optimize nanotherapeutics delivery to tumors. AIP Adv. 2, 011208 (2012).
Stéphanou, A., McDougall, S. R., Anderson, A. R. A. & Chaplain, M. A. J. Mathematical modelling of flow in 2D and 3D vascular networks: Applications to anti-angiogenic and chemotherapeutic drug strategies. Math. Comput. Model. 41, 1137–1156 (2005).
Sinek, J. P. et al. Predicting drug pharmacokinetics and effect in vascularized tumors using computer simulation. J. Math. Biol. 58, 485–510 (2008).
Steuperaert, M. et al. Mathematical modeling of intraperitoneal drug delivery: simulation of drug distribution in a single tumor nodule. Drug Deliv. 24, 491–501 (2017).
McDougall, S. R., Anderson, A. R. A., Chaplain, M. A. J. & Sherratt, J. A. Mathematical modelling of flow through vascular networks: Implications for tumour-induced angiogenesis and chemotherapy strategies. Bull. Math. Biol. 64, 673–702 (2002).
Mitchell, M. J., Jain, R. K. & Langer, R. Engineering and physical sciences in oncology: challenges and opportunities. Nat. Rev. Cancer 17, 659–675 (2017).
Baronzio, G., Parmar, G. & Baronzio, M. Overview of methods for overcoming hindrance to drug delivery to tumors, with special attention to tumor interstitial fluid. Front. Oncol. 8, 165 (2015).
Olive, K. P. et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 324, 1457–1461 (2009).
Workman, P. et al. Guidelines for the welfare and use of animals in cancer research. Br. J. Cancer 102, 1555–1577 (2010).
Janssen, F. J. A study of the absorption and scattering factors of light in whole blood. Med. Biol. Eng. 10, 231–240 (1972).
Dodt, H. U. et al. Ultramicroscopy: three-dimensional visualization of neuronal networks in the whole mouse brain. Nat. Methods 4, 331–336 (2007).
Jonkman, J. E., Swoger, J., Kress, H., Rohrbach, A. & Stelzer, E. H. Resolution in optical microscopy. Methods Enzymol. 360, 416–446 (2003).
Pries, A. R. et al. Resistance to blood flow in microvessels in vivo. Circ. Res. 75, 904–915 (1994).
Pries, A. R., Secomb, T. W. & Gaehtgens, P. The endothelial surface layer. Pflugers Arch. 440, 653–666 (2000).
Pries, A. R. & Secomb, T. W. Microvascular blood viscosity in vivo and the endothelial surface layer. Am. J. Physiol. Heart Circ. Physiol. 289, H2657–H2664 (2005).
Fry, B. C., Lee, J., Smith, N. P. & Secomb, T. W. Estimation of blood flow rates in large microvascular networks. Microcirculation 19, 530–538 (2012).
Fry, B. C., Roy, T. K. & Secomb, T. W. Capillary recruitment in a theoretical model for blood flow regulation in heterogeneous microvessel networks. Physiol. Rep. 1, e00050 (2013).
Sweeney, P. W., Walker-Samuel, S. & Shipley, R. J. Insights into cerebral haemodynamics and oxygenation utilising in vivo mural cell imaging and mathematical modelling. Sci. Rep. 8, 1373 (2018).
Pries, A. R. et al. Structural adaptation and heterogeneity of normal and tumor microvascular networks. PLoS. Comput. Biol. 5, e1000394 (2009).
Morikawa, S. et al. Abnormalities in pericytes on blood vessels and endothelial sprouts in tumors. Am. J. Pathol. 160, 985–1000 (2002).
Stamatelos, S. K., Androulakis, I. P., Kong, A. N. & Georgopoulos, P. G. A semi-mechanistic integrated toxicokinetic-toxicodynamic (TK/TD) model for arsenic(iii) in hepatocytes. J. Theor. Biol. 317, 244–256 (2013).
Secomb, T. W., Hsu, R., Park, E. Y. & Dewhirst, M. W. Green’s function methods for analysis of oxygen delivery to tissue by microvascular networks. Ann. Biomed. Eng. 32, 1519–1529 (2004).
Xu, X., Chen, A., Jansuwan, S., Heaslip, K. & Yang, C. Modeling transportation network redundancy. Transport. Res. Procedia 9, 283–302 (2015).
Schabel, M. C. & Parker, D. L. Uncertainty and bias in contrast concentration measurements using spoiled gradient echo pulse sequences. Phys. Med. Biol. 53, 2345–2373 (2008).
Ramasawmy, R. et al. Hepatic arterial spin labelling MRI: an initial evaluation in mice. NMR Biomed. 28, 272–280 (2015).
Acknowledgements
The authors acknowledge support received for the Kings College London & UCL CR-UK and EPSRC Comprehensive Cancer Imaging Centre, in association with the MRC and Department of Health (England) (C1519/A10331), Wellcome Trust (WT100247MA) and Rosetrees Trust/Stoneygate Trust (M135-F1 and M601). The authors thank OXiGENE for supplying OXi4503.
Author information
Authors and Affiliations
Contributions
A.d’E. designed and performed optical imaging experiments, analysed and interpreted results and wrote the first drafts of the manuscript. P.W.S. and R.S. developed software to perform mathematical and computational analysis. P.W.S. analysed and interpreted data and wrote and edited the paper. M.A. performed a subset of the optical imaging experiments. M.S. and S.W.-S. developed software for performing time-dependent simulations. R.R. and T.A.R. designed and performed ASL-MRI measurements, and developed software for quantifying the data. G.A. provided murine brain tumour models. A.D. assisted with student supervision and with the design of optical imaging experiments. M.F.L. provided access to imaging resources and student supervision. R.B.P. provided murine xenograft models and interpreted results. S.W.-S. developed software for segmenting optical imaging data. R.S. and S.W.-S. co-led the project, secured funding, supervised the design of experiments and simulations, developed the main concepts, interpreted results and contributed to the writing and editing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1–8, Supplementary Tables 1–3, Supplementary Results, Supplementary References 1–4 and Supplementary Video Captions 1–4.
Supplementary Video 1
Three-dimensional rendering of REANIMATE simulation results, describing the passage of a bolus of the commonly used MRI contrast agent Gd‐DTPA through tumour vasculature.
Supplementary Video 2
REANIMATE simulation of the delivery of the MRI contrast agent Gd‐DTPA using the vascular network from the LS174T colorectal tumour-xenograft model.
Supplementary Video 3
Dual-fluorescence labelling of the vasculature in an LS174T colorectal xenograft model, following treatment with the vascular targeting agent OXi4503.
Supplementary Video 4
REANIMATE simulation of the delivery of the vascular targeting agent OXi4503 to an LS174T colorectal xenograft model.
Rights and permissions
About this article
Cite this article
d’Esposito, A., Sweeney, P.W., Ali, M. et al. Computational fluid dynamics with imaging of cleared tissue and of in vivo perfusion predicts drug uptake and treatment responses in tumours. Nat Biomed Eng 2, 773–787 (2018). https://doi.org/10.1038/s41551-018-0306-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-018-0306-y
This article is cited by
-
Effect of vessel compression on blood flow in microvascular networks and its implications for tumour tissue hypoxia
Communications Physics (2024)
-
Development of a multiphase perfusion model for biomimetic reduced-order dense tumors
Experimental and Computational Multiphase Flow (2023)
-
Imaging intact human organs with local resolution of cellular structures using hierarchical phase-contrast tomography
Nature Methods (2021)
-
Tissue clearing to examine tumour complexity in three dimensions
Nature Reviews Cancer (2021)
-
Kinetic aspects of virus targeting by nanoparticles in vivo
Journal of Biological Physics (2021)