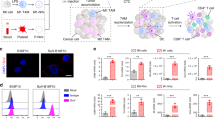
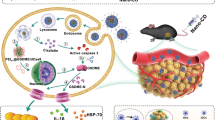
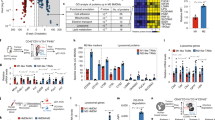
Abstract
Effectively activating macrophages that can ‘eat’ cancer cells is challenging. In particular, cancer cells secrete macrophage colony stimulating factor (MCSF), which polarizes tumour-associated macrophages from an antitumour M1 phenotype to a pro-tumorigenic M2 phenotype. Also, cancer cells can express CD47, a ‘don’t eat me’ signal that ligates with the signal regulatory protein alpha (SIRPα) receptor on macrophages to prevent phagocytosis. Here, we show that a supramolecular assembly consisting of amphiphiles inhibiting the colony stimulating factor 1 receptor (CSF-1R) and displaying SIRPα-blocking antibodies with a drug-to-antibody ratio of 17,000 can disable both mechanisms. The supramolecule homes onto SIRPα on macrophages, blocking the CD47–SIRPα signalling axis while sustainedly inhibiting CSF-1R. The supramolecule enhances M2-to-M1 repolarization within the tumour microenvironment, and significantly improves antitumour and antimetastatic efficacies in two aggressive animal models of melanoma and breast cancer, with respect to clinically available small-molecule and biologic inhibitors of CSF-1R signalling. Simultaneously blocking the CD47–SIRPα and MCSF–CSF-1R signalling axes may constitute a promising immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mantovani, A., Marchesi, F., Malesci, A., Laghi, L. & Allavena, P. Tumour-associated macrophages as treatment targets in oncology. Nat. Rev. Clin. Oncol. 14, 399–416 (2017).
Engblom, C., Pfirschke, C., & Pittet, M. J. The role of myeloid cells in cancer therapies. Nat. Rev. Cancer 16, 447–462 (2016).
Noy, R., & Pollard, J. W. Tumor-associated macrophages: from mechanisms to therapy. Immunity 41, 49–61 (2014).
Condeelis, J., & Pollard, J. W. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124, 263–266 (2006).
Gabrilovich, D. I., Ostrand-Rosenberg, S. & Bronte, V. Coordinated regulation of myeloid cells by tumours. Nat. Rev. Immunol. 12, 253–268 (2012).
Grivennikov, S. I., Greten, F. R., & Karin, M. Immunity, inflammation, and cancer. Cell 140, 883–899 (2010).
Biswas, S. K. & Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat. Immunol. 11, 889–896 (2010).
Mosser, D. M. & Edwards, J. P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 8, 958–969 (2008).
Sica, A. et al. Macrophage polarization in tumour progression. Sem. Cancer Biol. 18, 349–355 (2008).
Qian, B. Z. & Pollard, J. W. Macrophage diversity enhances tumor progression and metastasis. Cell 141, 39–51 (2010).
Ruffell, B. & Coussens, L. M. Macrophages and therapeutic resistance in cancer. Cancer Cell 27, 462–472 (2015).
Ries, C. H. et al. Targeting tumor-associated macrophages with anti-CSF-1R antibody reveals a strategy for cancer therapy. Cancer Cell 25, 846–859 (2014).
Pyonteck, S. M. et al. CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat. Med. 19, 1264–1272 (2013).
Chao, M. P., Weissman, I. L. & Majeti, R. The CD47-SIRPα pathway in cancer immune evasion and potential therapeutic implications. Curr. Opin. Immunol. 24, 225–232 (2012).
McCracken, M. N., Cha, A. C. & Weissman, I. L. Molecular pathways: activating T cells after cancer cell phagocytosis from blockade of CD47 “don’t eat me” signals. Clin. Cancer Res. 21, 3597–3601 (2015).
Beck, A., Goetsch, L., Dumontet, C. & Corvaia, N. Strategies and challenges for the next generation of antibody–drug conjugates. Nat. Rev. Drug Discov. 16, 315–337 (2017).
Garber, K. Bispecific antibodies rise again. Nat. Rev. Drug Discov. 13, 799–801 (2014).
Kulkarni, A. et al. Algorithm for designing nanoscale supramolecular therapeutics with increased anticancer efficacy. ACS Nano 10, 8154–8168 (2016).
Kulkarni, A., Natarajan, S. K., Chandrasekar, V., Pandey, P. R. & Sengupta, S. Combining immune checkpoint inhibitors and kinase-inhibiting supramolecular therapeutics for enhanced anticancer efficacy. ACS Nano 10, 9227–9242 (2016).
Kulkarni, A. A. et al. Supramolecular nanoparticles that target phosphoinositide-3-kinase overcome insulin resistance and exert pronounced antitumor efficacy. Cancer Res. 73, 6987–6997 (2013).
Zhou, D. et al. Macrophage polarization and function with emphasis on the evolving roles of coordinated regulation of cellular signaling pathways. Cell. Signal. 26, 192–197 (2014).
Wilson, H. M. SOCS proteins in macrophage polarization and function. Front. Immunol. 5, 357, (2014).
Ruffell, B., Affara, N. I. & Coussens, L. M. Differential macrophage programming in the tumor microenvironment. Trends Immunol. 33, 119–126 (2012).
Whyte, C. S. et al. Suppressor of cytokine signaling (SOCS)1 is a key determinant of differential macrophage activation and function. J. Leukoc. Biol. 90, 845–854 (2011).
Genin, M., Clement, F., Fattaccioli, A., Raes, M. & Michiels, C. M1 and M2 macrophages derived from THP-1 cells differentially modulate the response of cancer cells to etoposide. BMC Cancer 15, 577 (2015).
Gajewski, T. F., Schreiber, H. & Fu, Y. X. Innate and adaptive immune cells in the tumor microenvironment. Nat. Immunol. 14, 1014–1022 (2013).
Ruffell, B. et al. Macrophage IL-10 blocks CD8+ T cell-dependent responses to chemotherapy by suppressing IL-12 expression in intratumoral dendritic cells. Cancer Cell 26, 623–637 (2014).
Diamantis, N. & Banerji, U. Antibody-drug conjugates–an emerging class of cancer treatment. Br. J. Cancer 114, 362–367 (2016).
Goldman, A. et al. Rationally designed 2-in-1 nanoparticles can overcome adaptive resistance in cancer. ACS Nano 10, 5823–5834 (2016).
Sengupta, S. Cancer nanomedicine: lessons for immuno-oncology. Trends Cancer 3, 551–560 (2017).
Gholamin, S. et al. Disrupting the CD47-SIRPà anti-phagocytic axis by a humanized anti-CD47 antibody is an efficacious treatment for malignant pediatric brain tumors. Sci. Transl. Med. 9, eaaf2968 (2017).
Long, G. V. et al. Standard-dose pembrolizumab in combination with reduced-dose ipilimumab for patients with advanced melanoma (KEYNOTE-029): an open-label, phase 1b trial. Lancet Oncol. 18, 1202–1210 (2017).
Acknowledgements
This work was supported by a DoD Breakthrough Award (BC132168), an American Lung Association Innovation Award (LCD-259932-N), and an NCI UO1 (CA214411) to S.S. and a National Cancer Institute of the National Institutes of Health (P50CA168504) and Hearst Foundation/Brigham and Women’s Hospital Young Investigator Award to A.K. The authors would like to thank the Dana Farber Cancer Institute Flow Cytometry Core Facility for their expertise, consulting and assistance with flow cytometry experiments. The authors would like to thank the Mass Spectrometry Core Facility and Biophysical Characterization Core Facility at the Institute for Applied Life Sciences (IALS), University of Massachusetts Amherst for consultation and assistance with mass spectrometry experiments.
Author information
Authors and Affiliations
Contributions
A.K. conceived the idea, designed the experiments and mentored the research. P.P. performed the molecular dynamics simulation studies. V.C., S.K.N. and A.R. performed the supramolecule synthesis and characterization. V.C., S.K.N., A.R., J.N., H.B. and D.A. performed in vitro experiments. A.K.A. helped with confocal imaging. A.K., V.C., S.K.N. and A.R. performed in vivo experiments. A.K. and S.S. wrote the paper and received comments and edits from all the authors.
Corresponding authors
Ethics declarations
Competing interests
S.S. is a cofounder and holds equity in Akamara Therapeutics, which is developing supramolecular therapeutics, and holds equity in Mitra Biotech, which is developing cancer diagnostics.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplementary methods, figures and tables.
Rights and permissions
About this article
Cite this article
Kulkarni, A., Chandrasekar, V., Natarajan, S.K. et al. A designer self-assembled supramolecule amplifies macrophage immune responses against aggressive cancer. Nat Biomed Eng 2, 589–599 (2018). https://doi.org/10.1038/s41551-018-0254-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-018-0254-6
This article is cited by
-
Targeted modulation of immune cells and tissues using engineered biomaterials
Nature Reviews Bioengineering (2023)
-
Dual-ratiometric magnetic resonance tunable nanoprobe with acidic-microenvironment-responsive property to enhance the visualization of early tumor pathological changes
Nano Research (2023)
-
Tumor-associated macrophages as a potential therapeutic target in thyroid cancers
Cancer Immunology, Immunotherapy (2023)
-
Sequential acid/reduction response of triblock copolymeric nanomicelles to release camptothecin and toll-like receptor 7/8 agonist for orchestrated chemoimmunotherapy
Journal of Nanobiotechnology (2022)
-
Intercellular nanotubes mediate mitochondrial trafficking between cancer and immune cells
Nature Nanotechnology (2022)