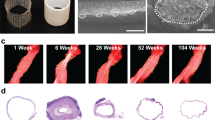
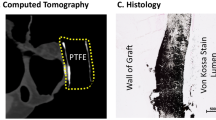
Abstract
Medical implants of fixed size cannot accommodate normal tissue growth in children and often require eventual replacement or—in some cases—removal, leading to repeated interventions, increased complication rates and worse outcomes. Implants that can correct anatomical deformities and accommodate tissue growth remain an unmet need. Here, we report the design and use of a growth-accommodating device for paediatric applications that consists of a biodegradable core and a tubular braided sleeve, with inversely related sleeve length and diameter. The biodegradable core constrains the diameter of the sleeve, and gradual core degradation following implantation enables sleeve and overall device elongation to accommodate tissue growth. By means of mathematical modelling and ex vivo experiments using harvested swine hearts, we demonstrate the predictability and tunability of the behaviour of the device for disease- and patient-specific needs. We also used the rat tibia and the piglet heart valve as two models of tissue growth to demonstrate that polymer degradation enables device expansion and growth accommodation in vivo.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
HCUP Kids’ Inpatient Database. Healthcare Cost and Utilization Project (Agency for Healthcare Research and Quality, 2012); https://hcupnet.ahrq.gov
Lam, S., Kuether, J., Fong, A. & Reid, R. Cranioplasty for large-sized calvarial defects in the pediatric population: a review. Craniomaxillofac. Trauma Reconstr. 8, 159–170 (2015).
Kaza, E. et al. Changes in left atrioventricular valve geometry after surgical repair of complete atrioventricular canal. J. Thorac. Cardiovasc. Surg. 143, 1117–1124 (2012).
Honjo, O., Mertens, L. & Van Arsdell, G. S. Atrioventricular valve repair in patients with single-ventricle physiology: mechanisms, techniques of repair, and clinical outcomes. Semin. Thorac. Cardiovasc. Surg. Pediatr. Card. Surg. Annu. 14, 75–84 (2011).
Chavaud, S. et al. Reconstructive surgery in congenital mitral valve insufficiency (Carpentier’s techniques): long-term results. J. Thorac. Cardiovasc. Surg. 115, 84–93 (1998).
Dearani, J. A. et al. Anatomic repair of Ebstein’s malformation: lessons learned with Cone reconstruction. Ann. Thorac. Surg. 95, 220–228 (2013).
Doty, D. B. & Doty, J. R. Cardiac Surgery: Operative Technique (Elsevier, Philadelphia, PA, 2012).
Parolari, A., Barili, F., Pilozzi, A. & Pacini, D. Ring or suture annuloplasty for tricuspid regurgitation? A meta-analysis review. Ann. Thorac. Surg. 98, 2255–2263 (2014).
Tang, G. H. et al. Tricuspid valve repair with an annuloplasty ring results in improved long-term outcomes. Circulation 114, 1577–1581 (2006).
Choi, J. B., Kim, K. H., Kim, M. H. & Kim, W. H. Mitral valve re-repair in an adolescent patient with prosthetic ring endocarditis: posterior leaflet augmentation and posterior strip annuloplasty. J. Card. Surg. 27, 560–562 (2012).
Jonas, R. A. Comprehensive Surgical Management of Congenital Heart Disease (CRC Press, New York, NY, 2014).
Kalangos, A. et al. Annuloplasty for valve repair with a new biodegradable ring: an experimental study. J. Heart Valve Dis. 15, 783–790 (2006).
Bautista-Hernandez, V. et al. Atrioventricular valve annular remodeling with a bioabsorbable ring in young children. J. Am. Coll. Cardiol. 60, 2255–2260 (2012).
Lee, T. M. et al. Risk factor analysis for second-stage palliation of single ventricle anatomy. Ann. Thorac. Surg. 93, 614–619 (2012).
Friend, L. & Widmann, R. F. Advances in management of limb length discrepancy and lower limb deformity. Curr. Opin. Pediatr. 20, 46–51 (2008).
Goldman, V. & Green, D. W. Advances in growth plate modulation for lower extremity malalignment (knock knees and bow legs). Curr. Opin. Pediatr. 22, 47–53 (2010).
Boero, S., Michelis, M. B. & Riganti, S. Use of the eight-plate for angular correction of knee deformities due to idiopathic and pathologic physis: initiating treatment according to etiology. J. Child. Orthop. 5, 209–216 (2011).
Stevens, P. M. Guided growth: 1933 to the present. Strat. Traum. Limb Recon. 1, 29–35 (2006).
Burnette, J. B. et al. Incidence of inpatient surgeries in children and young adults with childhood orthopedic diagnoses. J. Pediatr. Orthop. 24, 738–741 (2004).
Sturm, P. F., Anadio, J. M. & Dede, O. Recent advances in the management of early onset scoliosis. Orhop. Clin. N. Am. 45, 501–514 (2014).
Betz, R. R. et al. Vertebral body stapling: a fusionless treatment option for a growing child with moderate idiopathic scoliosis. Spine 35, 169–176 (2010).
Samdani, A. F. et al. Anterior vertebral body tethering for immature adolescent scoliosis: on year results on the first 32 patients. Eur. Spine J. 24, 1533–1539 (2015).
Daerden, F. & Lefeber, D. Pneumatic artificial muscles: actuators for robotics and automation. Eur. J. Mech. Environ. Eng. 47, 11–21 (2002).
Wang, Y., Ameer, G. A., Sheppard, B. J. & Langer, R. A tough biodegradable elastomer. Nat. Biotechnol. 20, 602–606 (2002).
Doumit, M., Fahim, A. & Munro, M. Analytical modeling and experimental validation of the braided pneumatic muscle. IEEE Trans. Robot. 25, 1282–1291 (2009).
Wang, Y., Kim, Y. M. & Langer, R. Technical note: in vivo degradation characteristics of poly(glycerol sebacate). J. Biomed. Mater. Res. 66A, 192–197 (2003).
Pomerantseva, I. et al. Degradation behavior of poly(glycerol sebacate). J. Biomed. Mater. Res. 91A, 1038–1047 (2009).
Stokes, I. A. F., Aronsson, D. D., Dimock, A. N., Cortright, V. & Beck, S. Endochondral growth in growth plates of three species at two anatomical locations modulated by mechanical compression and tension. J. Orthop. Res. 24, 1327–1334 (2006).
Sundback, C. A. et al. Biocompatibility analysis of poly(glycerol sebacate) as a nerve guide material. Biomaterials 26, 5454–5464 (2005).
Greenwald, D., Shumway, S., Albear, P. & Gottlieb, L. Mechanical comparison of 10 suture materials before and after in vivo incubation. J. Surg. Res. 56, 372–377 (1995).
Bylski-Austrow, D. I., Wall, E. J., Rupert, M. P., Roy, D. R. & Crawford, A. H. Growth plate forces in the adolescent human knee: a radiographic and mechanical study of epiphyseal staples. J. Pediatr. Orthop. 21, 817–823 (2001).
Siefert, A. W. et al. In-vivo mitral annuloplasty ring transducer: implications for implantation and annular downsizing. J. Biomech. 46, 2550–2553 (2013).
Sun, Z. et al. Glycolic acid modulates the mechanical property and degradation of poly(glycerol, sebacate, glycolic acid). J. Biomed. Mater. Res. A 92A, 332–339 (2010).
Colan, S. D. The why and how of Z scores. J. Am. Soc. Echocardiogr. 25, 1–2 (2013).
Dimeglio, A. & Canavese, F. The growing spine: how spinal deformities influence normal spine and thoracic cage growth. Eur. Spine J. 21, 64–70 (2012).
Wolford, L. M., Movahed, R. & Perez, D. E. A classification system for conditions causing condylar hyperplasia. J. Oral Maxillofac. Surg. 72, 567–595 (2014).
Acknowledgements
The authors are grateful to the Animal Research Children’s Hospital staff (A. Nedder (head veterinarian) and veterinary technicians) and the Boston Children’s Hospital perfusion team for their overwhelming support and assistance in this project. This work was also supported by the National Institutes of Health (grant GM086433 to J.M.K.) and the Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Education of Korea (2012R1A6A3A03041166) and the Korea Institute for Advancement of Technology (N0002123) to Y.L.
Author information
Authors and Affiliations
Contributions
E.N.F and Y.L. designed and performed the experiments, analysed the data and wrote the manuscript. E.D.O. contributed to the design of the experiments. N.V.V., S.S. and I.F. contributed to the design and performance of the experiments and to the analysis of the data. D.P., P.E.H., H.Y., A.G. and V.A. contributed to the design of the experiments. G.M. contributed to the analysis of the data. P.J.d.N. and J.M.K. contributed to the experimental design and manuscript preparation and supervised the overall project. All authors read and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
E.N.F., Y.L., E.D.O., N.V.V., D.P., P.E.H., H.Y., V.A., J.M.K. and P.J.d.N. have a provisional patent application entitled ‘Autonomously growing implantable device’ (USSN 62/295,768).
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary figures, tables and methods.
Supplementary Video 1
Representative video of a growth-accommodating annuloplasty ring in an ex vivo swine-heart preparation. The video is en face view of the tricuspid valve, with the ring in place. 100× normal speed
Rights and permissions
About this article
Cite this article
Feins, E.N., Lee, Y., O’Cearbhaill, E.D. et al. A growth-accommodating implant for paediatric applications. Nat Biomed Eng 1, 818–825 (2017). https://doi.org/10.1038/s41551-017-0142-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-017-0142-5
This article is cited by
-
Materials design for bone-tissue engineering
Nature Reviews Materials (2020)
-
Paediatric devices that grow up
Nature Biomedical Engineering (2017)