Abstract
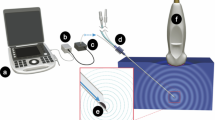
On-demand relief of local pain would allow patients to control the timing, intensity and duration of nerve blocks in a safe and non-invasive manner. Ultrasound would be a suitable trigger for such a system, as it is in common clinical use and can penetrate deeply into the body. Here, we demonstrate that ultrasound-triggered delivery of an anaesthetic from liposomes allows the timing, intensity and duration of nerve blocks to be controlled by ultrasound parameters. On insonation, the encapsulated sonosensitizer protoporphyrin IX produced reactive oxygen species that reacted with the liposomal membrane, leading to the release of the potent local anaesthetic tetrodotoxin. Repeatable ultrasound-triggered nerve blocks were achieved in vivo, with the nerve-block duration depending on the extent and intensity of insonation. There was no detectable systemic toxicity and tissue reaction was benign in all groups. On-demand, personalized local anaesthesia could be beneficial for the management of relatively localized pain states and could potentially minimize opioid use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Epstein-Barash, H. et al. Prolonged duration local anesthesia with minimal toxicity. Proc. Natl Acad. Sci. USA 106, 7125–7130 (2009).
McAlvin, J. B. & Kohane, D. S. in Focal Controlled Drug Delivery (eds. Domb, A. J. & Khan, W.) 653–677 (Springer US, New York, USA, 2014).
Rwei, A. Y. et al. Repeatable and adjustable on-demand sciatic nerve block with phototriggerable liposomes. Proc. Natl Acad. Sci. USA 112, 15719–15724 (2015).
Zhan, C. et al. Phototriggered local anesthesia. Nano Lett. 16, 177–181 (2016).
Stolik, S., Delgado, J. A., Pérez, A. & Anasagasti, L. Measurement of the penetration depths of red and near infrared light in human “ex vivo” tissues. J. Photochem. Photobiol. B 57, 90–93 (2000).
Rwei, A. Y., Wang, W. & Kohane, D. S. Photoresponsive nanoparticles for drug delivery. Nano Today 10, 451–467 (2015).
Smalley, P. J. Laser safety: risks, hazards, and control measures. Laser Ther. 20, 95–106 (2011).
Wood, A. K. & Sehgal, C. M. A review of low-intensity ultrasound for cancer therapy. Ultrasound Med. Biol. 41, 905–928 (2015).
Sirsi, S. R. & Borden, M. A. State-of-the-art materials for ultrasound-triggered drug delivery. Adv. Drug Deliv. Rev. 72, 3–14 (2014).
Marhofer, P. & Chan, V. W. S. Ultrasound-guided regional anesthesia: current concepts and future trends. Anesth. Analg. 104, 1265–1269 (2007).
Abrahams, M. S., Aziz, M. F., Fu, R. F. & Horn, J. L. Ultrasound guidance compared with electrical neurostimulation for peripheral nerve block: a systematic review and meta-analysis of randomized controlled trials. Br. J. Anaesth. 102, 408–417 (2009).
Li, F., Xie, C., Cheng, Z. & Xia, H. Ultrasound responsive block copolymer micelle of poly (ethylene glycol)–poly (propylene glycol) obtained through click reaction. Ultrason. Sonochem. 30, 9–17 (2016).
Lin, C.-Y., Javadi, M., Belnap, D. M., Barrow, J. R. & Pitt, W. G. Ultrasound sensitive eLiposomes containing doxorubicin for drug targeting therapy. Nanomedicine 10, 67–76 (2014).
Kim, H. J., Matsuda, H., Zhou, H. & Honma, I. Ultrasound‐triggered smart drug release from a poly (dimethylsiloxane)–mesoporous silica composite. Adv. Mater. 18, 3083–3088 (2006).
Paris, J. L., Cabañas, M. V., Manzano, M. & Vallet-Regí, M. Polymer-grafted mesoporous silica nanoparticles as ultrasound-responsive drug carriers. ACS Nano 9, 11023–11033 (2015).
Cintas, P., Tagliapietra, S., Caporaso, M., Tabasso, S. & Cravotto, G. Enabling technologies built on a sonochemical platform: challenges and opportunities. Ultrason. Sonochem. 25, 8–16 (2015).
Shi, J. et al. Reactive oxygen species—manipulated drug release from a smart envelope-type mesoporous titanium nanovehicle for tumor sonodynamic-chemotherapy. ACS Appl. Mater. Interfaces 7, 28554–28565 (2015).
Kuroki, M. et al. Sonodynamic therapy of cancer using novel sonosensitizers. Anticancer Res. 27, 3673–3677 (2007).
Kennedy, J. C. & Pottier, R. H. Endogenous protoporphyrin IX, a clinically useful photosensitizer for photodynamic therapy. J. Photochem. Photobiol. B 14, 275–292 (1992).
Jeffes, E. W. B. Levulan®: the first approved topical photosensitizer for the treatment of actinic keratosis. J. Dermatol. Treat. 13, S19–S23 (2002).
Padera, R. F., Tse, J. Y., Bellas, E. & Kohane, D. S. Tetrodotoxin for prolonged local anesthesia with minimal myotoxicity. Muscle Nerve 34, 747–753 (2006).
Sakura, S., Bollen, A. W., Ciriales, R. & Drasner, K. Local anesthetic neurotoxicity does not result from blockade of voltage-gated sodium channels. Anesth. Analg. 81, 338–346 (1995).
Hagen, N. A. et al. Tetrodotoxin for moderate to severe cancer-related pain: a multicentre, randomized, double-blind, placebo-controlled, parallel-design trial. Pain Res. Manag. 2017, 7212713 (2017).
Hagen, N. A. et al. A multicentre open-label safety and efficacy study of tetrodotoxin for cancer pain. Curr. Oncol. 18, E109–E116 (2011).
Carter, K. A. et al. Porphyrin–phospholipid liposomes permeabilized by near-infrared light. Nat. Commun. 5, 3546 (2014).
Ericson, M. B., Wennberg, A.-M. & Larkö, O. Review of photodynamic therapy in actinic keratosis and basal cell carcinoma. Ther. Clin. Risk Manag. 4, 1–9 (2008).
Kohane, D. S. et al. The local anesthetic properties and toxicity of saxitonin homologues for rat sciatic nerve block in vivo. Reg. Anesth. Pain Med. 25, 52–59 (2000).
Kohane, D. S. et al. A re-examination of tetrodotoxin for prolonged duration local anesthesia. Anesthesiology 89, 119–131 (1998).
McAlvin, J. B. et al. Corneal anesthesia with site 1 sodium channel blockers and dexmedetomidine. Invest. Ophthalmol. Vis. Sci. 56, 3820–3826 (2015).
Kohane, D. S. et al. Biocompatibility of lipid-protein-sugar particles containing bupivacaine in the epineurium. J. Biomed. Mater. Res. 59, 450–459 (2002).
Marhofer, P., Harrop-Griffiths, W., Willschke, H. & Kirchmair, L. Fifteen years of ultrasound guidance in regional anaesthesia: part 2-recent developments in block techniques. Br. J. Anaesth 104, 673–683 (2010).
Hayes, B. T., Merrick, M. A., Sandrey, M. A. & Cordova, M. L. Three-MHz ultrasound heats deeper into the tissues than originally theorized. J. Athl. Train. 39, 230–234 (2004).
Rosenthal, I., Sostaric, J. Z. & Riesz, P. Sonodynamic therapy—a review of the synergistic effects of drugs and ultrasound. Ultrason. Sonochem. 11, 349–363 (2004).
Mišík, V. & Riesz, P. Free radical intermediates in sonodynamic therapy. Ann. NY Acad. Sci. 899, 335–348 (2000).
Leighton, T. G., Pickworth, M. J. W., Walton, A. J. & Dendy, P. P. Studies of the cavitational effects of clinical ultrasound by sonoluminescence: 1. Correlation of sonoluminescence with the standing wave pattern in an acoustic field produced by a therapeutic unit. Phys. Med. Biol. 33, 1239 (1988).
Pong, M. et al. In vitro ultrasound-mediated leakage from phospholipid vesicles. Ultrasonics 45, 133–145 (2006).
Schroeder, A., Kost, J. & Barenholz, Y. Ultrasound, liposomes, and drug delivery: principles for using ultrasound to control the release of drugs from liposomes. Chem. Phys. Lipids 162, 1–16 (2009).
Lin, H.-Y. & Thomas, J. L. Factors affecting responsivity of unilamellar liposomes to 20 kHz ultrasound. Langmuir 20, 6100–6106 (2004).
Voszka, I. et al. Interaction of photosensitizers with liposomes containing unsaturated lipid. Chem. Phys. Lipids 145, 63–71 (2007).
Lyubimtsev, A. et al. Aggregation behavior and UV-vis spectra of tetra-and octaglycosylated zinc phthalocyanines. J. Porphyr. Phthalocyanines 15, 39–46 (2011).
Rokitskaya, T. I., Block, M., Antonenko, Y. N., Kotova, E. A. & Pohl, P. Photosensitizer binding to lipid bilayers as a precondition for the photoinactivation of membrane channels. Biophys. J. 78, 2572–2580 (2000).
Adams, H. J., Blair, M. R. J. & Takman, B. H. The local anesthetic activity of tetrodotoxin alone and in combination with vasoconstrictors and local anesthetics. Anesth. Analg. 55, 568–573 (1976).
Lobo, K. et al. A phase 1, dose-escalation, double-blind, block-randomized, controlled trial of safety and efficacy of neosaxitoxin alone and in combination with 0.2% bupivacaine, with and without epinephrine, for cutaneous anesthesia. Anesthesiology 123, 873–885 (2015).
Kohane, D. S. Microparticles and nanoparticles for drug delivery. Biotechnol. Bioeng. 96, 203–209 (2007).
Naor, O., Krupa, S. & Shy, S. Ultrasonic neuromodulation. J. Neural Eng. 13, 031003 (2016).
Brummett, C. M., Hong, E. K., Janda, A. M., Amodeo, F. S. & Lydic, R. Perineural dexmedetomidine added to ropivacaine for sciatic nerve block in rats prolongs the duration of analgesia by blocking the hyperpolarization-activated cation current. Anesthesiology 115, 836–843 (2011).
Yoshitomi, T. et al. Dexmedetomidine enhances the local anesthetic action of lidocaine via an α-2A adrenoceptor. Anesth. Analg. 107, 96–101 (2008).
Yabuki, A. et al. Locally injected dexmedetomidine induces vasoconstriction via peripheral α-2A adrenoceptor subtype in guinea pigs. Reg. Anesth. Pain Med. 39, 133–136 (2014).
Curley, J. et al. Prolonged regional nerve blockade. Injectable biodegradable bupivacaine/polyester microspheres. Anesthesiology 84, 1401–1410 (1996).
Kohane, D. S., Lipp, M., Kinney, R. C., Lotan, N. & Langer, R. Sciatic nerve blockade with lipid-protein-sugar particles containing bupivacaine. Pharm. Res. 17, 1243–1249 (2000).
Castillo, J. et al. Glucocorticoids prolong rat sciatic nerve blockade in vivo from bupivacaine microspheres. Anesthesiology 85, 1157–1166 (1996).
Kohane, D. S. et al. Prolonged duration local anesthesia from tetrodotoxin-enhanced local anesthetic microspheres. Pain 104, 415–421 (2003).
Lago, J., Rodriguez, L. P., Blanco, L., Vieites, J. M. & Cabado, A. G. Tetrodotoxin, an extremely potent marine neurotoxin: distribution, toxicity, origin and therapeutical uses. Mar. Drugs 13, 6384–6406 (2015).
Kohane, D. S., Lu, N. T., Cairns, B. E. & Berde, C. B. Effects of adrenergic agonists and antagonists on tetrodotoxin-induced nerve block. Reg. Anesth. Pain Med. 26, 239–245 (2001).
Richard, B. M. et al. The safety of EXPAREL® (bupivacaine liposome injectable suspension) administered by peripheral nerve block in rabbits and dogs. J. Drug Deliv. 2012, 962101 (2012).
Vanrooijen, N. & Vannieuwmegen, R. Liposomes in immunology—multilamllar phosphatidylcholine liposomes as a simple, biodegradable and harmless adjuvant without any immunogenic activity of its own. Immunol. Commun. 9, 243–256 (1980).
Rosenberg, G. J. & Cabrera, R. C. External ultrasonic lipoplasty: an effective method of fat removal and skin shrinkage. Plast. Reconstr. Surg. 105, 785–791 (2000).
Pudroma, X., Moan, J., Ma, L.-W., Iani, V. & Juzeniene, A. A comparison of 5-aminolaevulinic acid- and its heptyl ester: dark cytotoxicity and protoporphyrin IX synthesis in human adenocarcinoma WiDr cells and in athymic nude mice healthy skin. Exp. Dermatol. 18, 985–987 (2009).
Roots, R. & Okada, S. Estimation of life times and diffusion distances of radicals involved in X-ray-induced DNA strand breaks or killing of mammalian-cells. Radiat. Res. 64, 306–320 (1975).
Pryor, W. A. Oxyradicals and related species—their formation, lifetimes, and reactions. Annu. Rev. Physiol. 48, 657–667 (1986).
Skovsen, E., Snyder, J. W., Lambert, J. D. C. & Ogilby, P. R. Lifetime and diffusion of singlet oxygen in a cell. J. Phys. Chem. B 109, 8570–8573 (2005).
Kohane, D. S. et al. Sciatic nerve blockade in infant, adolescent, and adult rats: a comparison of ropivacaine with bupivacaine. Anesthesiology 89, 1199–1208 (1998).
Rodriguez-Navarro, A. J. et al. Potentiation of local anesthetic activity of neosaxitoxin with bupivacaine or epinephrine: development of a long-acting pain blocker. Neurotox. Res. 16, 408–415 (2009).
Rodríguez-Navarro, A. J. et al. Comparison of neosaxitoxin versus bupivacaine via port infiltration for postoperative analgesia following laparoscopic cholecystectomy: a randomized, double-blind trial. Reg. Anesth. Pain Med. 36, 103–109 (2011).
Alkan-Onyuksel, H. et al. Development of inherently echogenic liposomes as an ultrasonic contrast agent. J. Pharm. Sci. 85, 486–490 (1996).
Shung, K. K. High frequency ultrasonic imaging. J. Med. Ultrasound 17, 25–30 (2009).
Jaafar-Maalej, C., Diab, R., Andrieu, V., Elaissari, A. & Fessi, H. Ethanol injection method for hydrophilic and lipophilic drug-loaded liposome preparation. J. Liposome Res. 20, 228–243 (2010).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Da Costa, M. M. J. et al. A new zebrafish model produced by TILLING of SOD1-related amyotrophic lateral sclerosis replicates key features of the disease and represents a tool for in vivo therapeutic screening. Dis. Model. Mech. 7, 73–81 (2014).
Wu, D. & Yotnda, P. Production and detection of reactive oxygen species (ROS) in cancers. J. Vis. Exp. 3357 (2011).
Jiang, Z.-Y., Woollard, A. C. & Wolff, S. P. Lipid hydroperoxide measurement by oxidation of Fe2+ in the presence of xylenol orange. Comparison with the TBA assay and an iodometric method. Lipids 26, 853–856 (1991).
Rouhi, N., Jain, D., Zand, K. & Burke, P. J. Carbon nanotube field effect transistors using printed semiconducting tubes. Nanotechnology 1, 180–182 (2010).
Liang, X., Mao, G. & Ng, K. Y. S. Mechanical properties and stability measurement of cholesterol-containing liposome on mica by atomic force microscopy. J. Colloid Interface Sci. 278, 53–62 (2004).
Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16, 109–110 (1983).
Rwei, A. Y., Zhan, C., Wang, B. & Kohane, D. S. Multiply repeatable and adjustable on-demand phototriggered local anesthesia. J. Control. Release 251, 68–74 (2017).
Thalhammer, J., Vladimirova, M., Bershadsky, B. & Strichartz, G. Neurologic evaluation of the rat during sciatic nerve block with lidocaine. Anesthesiology 82, 1013–1025 (1995).
McAlvin, J. B. et al. Multivesicular liposomal bupivacaine at the sciatic nerve. Biomaterials 35, 4557–4564 (2014).
Acknowledgements
This work was supported by a National Institutes of Health grant (GM073626 to D.S.K.). J.L.P. acknowledges the Ministerio de Economía y Competitividad, Spain, for PhD grants (BES-2013-064182 and EEBB-I-16-11313) associated with MAT2012-35556. We thank A. Schwartzman and the Massachusetts Institute of Technology NanoMechanical Technology Laboratory for assistance with the AFM measurements.
Author information
Authors and Affiliations
Contributions
A.Y.R., J.L.P., W.W. and D.S.K. designed the experiments. A.Y.R., J.L.P., B.W. and C.D.A. performed the experiments. A.Y.R., J.L.P., W.W., M.V.-R., R.L. and D.S.K. analysed the data. A.Y.R., J.L.P., R.L. and D.S.K. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
Two provisional patent applications (U.S.S.N. 62/239,164 and U.S.S.N. 62/329,721) have been filed concerning the technology presented in this work.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Rwei, A.Y., Paris, J.L., Wang, B. et al. Ultrasound-triggered local anaesthesia. Nat Biomed Eng 1, 644–653 (2017). https://doi.org/10.1038/s41551-017-0117-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-017-0117-6
This article is cited by
-
Aromatized liposomes for sustained drug delivery
Nature Communications (2023)
-
Ultrasound-responsive low-dose doxorubicin liposomes trigger mitochondrial DNA release and activate cGAS-STING-mediated antitumour immunity
Nature Communications (2023)
-
New opportunities and old challenges in the clinical translation of nanotheranostics
Nature Reviews Materials (2023)
-
An aptamer-based depot system for sustained release of small molecule therapeutics
Nature Communications (2023)
-
Biodegradable reduce expenditure bioreactor for augmented sonodynamic therapy via regulating tumor hypoxia and inducing pro-death autophagy
Journal of Nanobiotechnology (2021)